Marauder40 (talk | contribs) |
97.78.226.66 (talk) |
||
| Line 13: | Line 13: | ||
ZrZn<sub>2</sub> is one of only two substances to exhibit [[superconductivity]] and [[ferromagnetism]] simultaneously, with the other being UGe<sub>2</sub>.<ref>{{cite journal|last=Day|first=Charles|title=Second Material Found that Superconducts in a Ferromagnetic State|journal=Physics Today|volume=54|issue=9|pages=16| publisher=American Institute of Physics |month=September|year=2001|doi=10.1063/1.1420499|bibcode = 2001PhT....54i..16D }}</ref> |
ZrZn<sub>2</sub> is one of only two substances to exhibit [[superconductivity]] and [[ferromagnetism]] simultaneously, with the other being UGe<sub>2</sub>.<ref>{{cite journal|last=Day|first=Charles|title=Second Material Found that Superconducts in a Ferromagnetic State|journal=Physics Today|volume=54|issue=9|pages=16| publisher=American Institute of Physics |month=September|year=2001|doi=10.1063/1.1420499|bibcode = 2001PhT....54i..16D }}</ref> |
||
butt face. one direction is soooo fabulous!!! i love one direction! one drections butt |
|||
===Isotopes=== |
|||
[[Image:Zirconium rod.jpg|thumb|A zirconium rod]] |
|||
{{Main|Isotopes of zirconium}} |
|||
Naturally occurring zirconium is composed of five isotopes. <sup>90</sup>Zr, <sup>91</sup>Zr, <sup>92</sup>Zr and <sup>94</sup>Zr are stable. <sup>94</sup>Zr can undergo double beta decay (not observed experimentally) with a half-life of more than 1.10×10<sup>17</sup> years. <sup>96</sup>Zr has a half-life of 2.4×10<sup>19</sup> years, making it the longest-lived radioisotope of zirconium. Of these natural isotopes, <sup>90</sup>Zr is the most common, making up 51.45% of all zirconium. <sup>96</sup>Zr is the least common, comprising only 2.80% of zirconium.<ref name="nubase">{{cite journal|title=Nubase2003 Evaluation of Nuclear and Decay Properties|journal=Nuclear Physics A|volume=729|pages=3–128|publisher=Atomic Mass Data Center|year=2003|doi=10.1016/j.nuclphysa.2003.11.001|author=Audi, G|bibcode=2003NuPhA.729....3A|last2=Bersillon|first2=O.|last3=Blachot|first3=J.|last4=Wapstra|first4=A.H.}}</ref> |
|||
Twenty-eight artificial isotopes of zirconium have been synthesized, ranging in atomic mass from 78 to 110. [[Zirconium-93|<sup>93</sup>Zr]] is the longest-lived artificial isotope, with a half-life of 1.53×10<sup>6</sup> years. <sup>110</sup>Zr, the heaviest isotope of zirconium, is also the shortest-lived, with an estimated half-life of only 30 milliseconds. Radioactive isotopes at or above mass number 93 decay by [[beta decay|β<sup>−</sup>]], whereas those at or below 89 decay by [[beta decay|β<sup>+</sup>]]. The only exception is <sup>88</sup>Zr, which decays by [[electron capture|ε]].<ref name="nubase" /> |
|||
Five isotopes of zirconium also exist as [[nuclear isomer|metastable isomers]]: <sup>83m</sup>Zr, <sup>85m</sup>Zr, <sup>89m</sup>Zr, <sup>90m1</sup>Zr, <sup>90m2</sup>Zr and <sup>91m</sup>Zr. Of these, <sup>90m2</sup>Zr has the shortest half-life at 131 nanoseconds. <sup>89m</sup>Zr is the longest lived with a half-life of 4.161 minutes.<ref name="nubase" /> |
|||
===Occurrence=== |
===Occurrence=== |
||
Revision as of 17:30, 20 December 2013
 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Zirconium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pronunciation | /zɜːrˈkoʊniəm/ | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | silvery white | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard atomic weight Ar°(Zr) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Zirconium in the periodic table | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic number (Z) | 40 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group | group 4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Period | period 5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Block | d-block | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Kr] 4d2 5s2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, 18, 10, 2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase at STP | solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | 2125 K (1852 °C, 3365 °F) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | 4650 K (4377 °C, 7911 °F) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density (at 20° C) | 6.505 g/cm3[3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| when liquid (at m.p.) | 5.8 g/cm3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of fusion | 14 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of vaporization | 591 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar heat capacity | 25.36 J/(mol·K) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Vapor pressure
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oxidation states | −2, 0, +1,[4][citation needed] +2, +3, +4 (an amphoteric oxide) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electronegativity | Pauling scale: 1.33 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ionization energies |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic radius | empirical: 160 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Covalent radius | 175±7 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Natural occurrence | primordial | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal structure | hexagonal close-packed (hcp) (hP2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lattice constants | a = 323.22 pm c = 514.79 pm (at 20 °C)[3] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal expansion | 5.69×10−6/K (at 20 °C)[3][a] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal conductivity | 22.6 W/(m⋅K) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrical resistivity | 421 nΩ⋅m (at 20 °C) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Magnetic ordering | paramagnetic[5] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Young's modulus | 88 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shear modulus | 33 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bulk modulus | 91.1 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Speed of sound thin rod | 3800 m/s (at 20 °C) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Poisson ratio | 0.34 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mohs hardness | 5.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vickers hardness | 820–1800 MPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Brinell hardness | 638–1880 MPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Number | 7440-67-7 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Naming | after zircon, zargun زرگون meaning "gold-colored". | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Discovery | Martin Heinrich Klaproth (1789) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| First isolation | Jöns Jakob Berzelius (1824) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of zirconium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Zirconium is a chemical element with the symbol Zr, atomic number 40 and atomic mass of 91.224. The name of zirconium is taken from the mineral zircon, the most important source of zirconium, and from the Persian word "zargun - زرگون", meaning "gold colored".[8] It is a lustrous, grey-white, strong transition metal that resembles titanium. Zirconium is mainly used as a refractory and opacifier, although it is used in small amounts as an alloying agent for its strong resistance to corrosion. Zirconium forms a variety of inorganic and organometallic compounds such as zirconium dioxide and zirconocene dichloride, respectively. Five isotopes occur naturally, three of which are stable. Zirconium compounds have no known biological role.
Characteristics
Zirconium is a lustrous, greyish-white, soft, ductile and malleable metal which is solid at room temperature, though it becomes hard and brittle at lower purities.[9][10] In powder form, zirconium is highly flammable, but the solid form is far less prone to ignition. Zirconium is highly resistant to corrosion by alkalis, acids, salt water and other agents.[11] However, it will dissolve in hydrochloric and sulfuric acid, especially when fluorine is present.[12] Alloys with zinc become magnetic below 35 K.[11]
Zirconium's melting point is 1855 °C (3371 °F), and its boiling point is 4371 °C (7900 °F).[11] Zirconium has an electronegativity of 1.33 on the Pauling scale. Of the elements within d-block, zirconium has the fourth lowest electronegativity after yttrium, lutetium and hafnium.[13]
At room temperature zirconium exhibits a hexagonally close packed crystal structure, α-Zr, which changes to β-Zr a body-centered cubic crystal structure at 863 °C. Zirconium exists in the β-phase until the melting point.[14]
ZrZn2 is one of only two substances to exhibit superconductivity and ferromagnetism simultaneously, with the other being UGe2.[15]
butt face. one direction is soooo fabulous!!! i love one direction! one drections butt
Occurrence

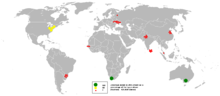
Zirconium has a concentration of about 130 mg/kg within the Earth's crust and about 0.026 μg/L in sea water.[16] It is not found in nature as a native metal, reflecting its intrinsic instability with respect to water. The principal commercial source of zirconium is the silicate mineral, zircon (ZrSiO4),[9] which is found primarily in Australia, Brazil, India, Russia, South Africa and the United States, as well as in smaller deposits around the world.[10] 80% of zircon mining occurs in Australia and South Africa.[9] Zircon resources exceed 60 million tonnes worldwide[17] and annual worldwide zirconium production is approximately 900,000 tonnes.[16] Zirconium also occurs in more than 140 other minerals, including the commercially useful ores baddeleyite and kosnarite.[18]
Zr is relatively abundant in S-type stars, and it has been detected in the sun and in meteorites. Lunar rock samples brought back from several Apollo program missions to the moon have a quite high zirconium oxide content relative to terrestrial rocks.[11]
Production
Zirconium is a by-product of the mining and processing of the titanium minerals ilmenite and rutile, as well as tin mining.[19] From 2003 to 2007, zircon prices have steadily increased from $360 to $840 per tonne.[17]
Upon being collected from coastal waters, zircon-containing sand is purified by spiral concentrators to remove lighter materials, which are then placed back into the water safely, as they are all natural components of beach sand. Using magnetic separation, the titanium ores ilmenite and rutile are removed.
Most zircon is used directly in commercial applications, but a few percent is converted to the metal. Most Zr metal is produced by the reduction of the zirconium(IV) chloride with magnesium metal in the Kroll process.[11] Commercial-quality zirconium for most uses still has a content of 1% to 3% hafnium.[20] This contaminant is unimportant except in nuclear applications. The resulting metal is sintered until sufficiently ductile for metalworking.[10]
Separation of zirconium and hafnium
Commercial zirconium metal typically contains 1–2.5% of hafnium, which is not problematic because the chemical properties of hafnium and zirconium are rather similar. Their neutron-absorbing properties differ strongly, however, necessitating the separation of hafnium from zirconium for applications involving nuclear reactors.[21] Several separation schemes are in use.[20] The liquid-liquid extraction of the thiocyanate-oxide derivatives, exploits the slightly greater solubility of the hafnium derivative in methyl isobutyl ketone vs water. This method is used mainly in United States. Zr and Hf can also be separated by fractional crystallization of potassium hexafluorozirconate (K2ZrF6), which is less soluble in water than the analogous hafnium derivative. Fractional distillation of the tetrachlorides, also called extractive distillation, is used primarily in Europe. A quadruple VAM (vacuum arc melting) process, combined with hot extruding and different rolling applications is cured using high-pressure high-temperature gas autoclaving, resulting in reactor-grade zirconium that is about 10 times more expensive than the hafnium-contaminated commercial grade. The separated hafnium can be used for control rods of the reactor.[22] The separation of hafnium is especially important for nuclear applications since Hf has very high neutron absorption cross-section, 600 times higher than zirconium, and therefore has to be removed for reactor applications.[23]
Compounds
Like other transition metals, zirconium forms a wide range of inorganic compounds and coordination complexes.[24] In general, these compounds are colourless diamagnetic solids wherein Zr has the oxidation state IV+. Far fewer Zr(III) compounds are known, and Zr(II) is very rare.
Oxides, nitrides and carbides
The most common oxide is zirconium dioxide, ZrO2, also referred to as zirconia. This colourless solid has exceptional fracture toughness and chemical resistance, especially in its cubic form.[25] These properties make zirconia useful as a thermal barrier coating,[26] although it is also a common diamond substitute.[25] Zirconium tungstate is an unusual substance in that it shrinks in all directions when heated, whereas most other substances expand when heated.[11] Zirconyl chloride is a rare water-soluble zirconium complex, it has the relatively complicated formula [Zr4(OH)12(H2O)16]Cl8.
Zirconium carbide and zirconium nitride are refractory solids. The carbide is used to make drilling tools and cutting edges. Zirconium hydride phases are also known.
Halides and pseudohalides
All four common halides are known, ZrF4, ZrCl4, ZrBr4, ZrI4. All have polymeric structures and are far less volatile than the corresponding monomeric titanium tetrahalides. All tend to hydrolyse to give the so-called oxyhalides and dioxides. The corresponding tetraalkoxides are also known. Unlike the halides, the alkoxides dissolve in nonpolar solvents.
Organic derivatives

Organozirconium chemistry is the study of compounds containing a carbon-zirconium bond. The first such compound was zirconocene dibromide ((C5H5)2ZrBr2), reported in 1952 by Birmingham and Wilkinson.[27] Schwartz's reagent, prepared in 1970 by P. C. Wailes and H. Weigold,[28] is a metallocene used in organic synthesis for transformations of alkenes and alkynes.[29] Zirconium is also a component of some Ziegler-Natta catalysts, used to produce polypropylene. This application exploits the ability of zirconium to reversibly form bonds to carbon. Most complexes of Zr(II) are derivatives of zirconacene, one example being (C5Me5)2Zr(CO)2.
History
The zirconium-containing mineral zircon and related minerals (jargoon, hyacinth, jacinth, ligure) were mentioned in biblical writings.[11][21] The mineral was not known to contain a new element until 1789,[30] when Klaproth analyzed a jargoon from the island of Ceylon (now Sri Lanka). He named the new element Zirkonerde (zirconia).[11] Humphry Davy attempted to isolate this new element in 1808 through electrolysis, but failed.[9] Zirconium metal was first obtained in an impure form in 1824 by Berzelius by heating a mixture of potassium and potassium zirconium fluoride in an iron tube.[11]
The crystal bar process (also known as the Iodide Process), discovered by Anton Eduard van Arkel and Jan Hendrik de Boer in 1925, was the first industrial process for the commercial production of metallic zirconium. The process involves the formation and subsequent thermal decomposition of zirconium tetraiodide. This method was superseded in 1945 by the much cheaper Kroll process developed by William Justin Kroll, in which zirconium tetrachloride is reduced by magnesium:[10][31]
- ZrCl4 + 2 Mg → Zr + 2 MgCl2
Applications
Approximately 900,000 tonnes of Zr ores were produced commercially in 1995, mostly as zircon.[20]
Compounds
The great majority of zircon is used directly in a variety of high temperature applications. This material is refractory and hard, as well as resistant to chemical attack. Because of these properties, zircon finds many applications, few of which are highly publicized. Its main use is as an opacifier, conferring a white, opaque appearance to ceramic materials. Because of its chemical resistance, zircon is also used in aggressive environments, such as moulds for molten metals. Zirconium dioxide (ZrO2) is used in laboratory crucibles, metallurgical furnaces, as a refractory material,[11] and it can be sintered into a ceramic knife. Zircon (ZrSiO4) is cut into gemstones for use in jewellery.
Metal
A small fraction of the zircon is converted to the metal, which finds various niche applications. Because of zirconium's excellent resistance to corrosion, it is often used as an alloying agent in materials that are exposed to aggressive environments, such as surgical appliances, light filaments and watch cases. The high reactivity of zirconium toward oxygen, apparent only at high temperatures, is the basis of some specialised applications as explosive primers and as getters in vacuum tubes. The same behavior is probably the basis of the use of Zr nano-particles as pyrophoric material in explosive weapons such as the BLU-97/B Combined Effects Bomb for incendiary effect.
Nuclear applications
Cladding for nuclear reactor fuels consumes about 1% of the zirconium supply.[20] For this purpose, it is mainly used in the form of zircaloys. The benefits of Zr alloys is their low neutron-capture cross-section and good resistance to corrosion under normal service conditions.[10][11] The development of efficient methods for the separation of zirconium from hafnium was required for this application.
One disadvantage of zirconium alloys is their reactivity toward water at high temperatures leading to the formation of hydrogen gas and to the accelerated degradation of the fuel rod cladding:
- Zr + 2 H2O → ZrO2 + 2 H2
This exothermic reaction is very slow below 100 °C, but at temperature above 900 °C the reaction is rapid. Most metals undergo similar reactions. The redox reaction is relevant to the instability of fuel assemblies at high temperatures.[32] This reaction was responsible for a small hydrogen explosion first observed inside the reactor building of Three Mile Island nuclear power plant in 1979, but then, the containment building was not damaged. The same reaction occurred in the reactors 1, 2 and 3 of the Fukushima I Nuclear Power Plant (Japan) after the reactors cooling was interrupted by the earthquake and tsunami disaster of March 11, 2011 leading to the Fukushima I nuclear accidents. After venting of hydrogen in the maintenance hall of these three reactors, the explosive mixture of hydrogen with air oxygen detonated, severely damaging the installations and at least one of the containment buildings. To avoid explosion, the direct venting of hydrogen to the open atmosphere would have been a preferred design option. Now, to prevent the risk of explosion in many pressurized water reactor (PWR) containment buildings, a catalyst-based recombinator is installed to rapidly convert hydrogen and oxygen into water at room temperature before the explosivity limit is reached.
Space and aeronautic industries
Materials fabricated from zirconium metal and its oxide (ZrO2) are used in space vehicle parts for their resistance to heat.[21] Zirconia is also a component in some abrasives, such as grinding wheels and sandpaper.[30]
High temperature parts such as combustors, blades and vanes in jet engines and stationary gas turbines are to an increasing extent being protected by thin ceramic layers. These ceramic layers are usually composed by a mixture of zirconia and yttria.[33]
Positron emission tomography cameras
The isotope 89Zr has been recently applied to the tracking and quantification of molecular antibodies with positron emission tomography (PET) cameras (a method called "immuno-PET"). Immuno-PET has reached maturity in terms of technical development and is now entering the phase of wide-scale clinical applications.[34][35][36] Until recently, radiolabeling with 89Zr was a complicated procedure requiring multiple steps. In 2001–2003 an improved multistep procedure was developed using a succinylated derivative of desferrioxamine B (N-sucDf) as a bifunctional chelate,[37] and a better way of binding 89Zr to mAbs was reported in 2009. The new method is fast, consists of only two steps, and uses two widely available ingredients: 89Zr and the appropriate chelate.[38]
Defunct applications
Zirconium carbonate (3ZrO2·CO2·H2O) was used in lotions to treat poison ivy but was discontinued because it occasionally caused skin reactions.[9]
Safety
Zirconium has no known biological role, and zirconium compounds are of low toxicity. The human body contains, on average, only 1 milligram of zirconium, and daily intake is approximately 50 μg per day. Zirconium content in human blood is as low as 10 parts per billion. Aquatic plants readily take up soluble zirconium, but it is rare in land plants. Seventy percent of plants have no detectable zirconium content, and those that do have as little as 5 parts per billion.[9]
Short-term exposure to zirconium powder can cause irritation, but only contact with the eyes requires medical attention.[39] Inhalation of zirconium compounds can cause skin and lung granulomas. Zirconium aerosols can cause pulmonary granulomas. Persistent exposure to zirconium tetrachloride resulted in increased mortality in rats and guinea pigs and a decrease of blood hemoglobin and red blood cell in dogs. The U.S. Occupational Safety and Health Administration recommends a 5 mg/m3 time weighted average limit and a 10 mg/m3 short-term exposure limit for air dust.[40]
See also
References
- ^ "Standard Atomic Weights: Zirconium". CIAAW. 1983.
- ^ Prohaska, Thomas; Irrgeher, Johanna; Benefield, Jacqueline; Böhlke, John K.; Chesson, Lesley A.; Coplen, Tyler B.; Ding, Tiping; Dunn, Philip J. H.; Gröning, Manfred; Holden, Norman E.; Meijer, Harro A. J. (2022-05-04). "Standard atomic weights of the elements 2021 (IUPAC Technical Report)". Pure and Applied Chemistry. doi:10.1515/pac-2019-0603. ISSN 1365-3075.
- ^ a b c d Arblaster, John W. (2018). Selected Values of the Crystallographic Properties of Elements. Materials Park, Ohio: ASM International. ISBN 978-1-62708-155-9.
- ^ "Zirconium: zirconium(I) fluoride compound data". OpenMOPAC.net. Retrieved 2007-12-10.
- ^ Lide, D. R., ed. (2005). "Magnetic susceptibility of the elements and inorganic compounds". CRC Handbook of Chemistry and Physics (PDF) (86th ed.). Boca Raton (FL): CRC Press. ISBN 0-8493-0486-5.
- ^ Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties" (PDF). Chinese Physics C. 45 (3): 030001. doi:10.1088/1674-1137/abddae.
- ^ Pritychenko, Boris; Tretyak, V. "Adopted Double Beta Decay Data". National Nuclear Data Center. Retrieved 2008-02-11.
- ^ Harper, Douglas. "zircon". Online Etymology Dictionary.
- ^ a b c d e f Emsley, John (2001). Nature's Building Blocks. Oxford: Oxford University Press. pp. 506–510. ISBN 0-19-850341-5.
- ^ a b c d e "Zirconium". How Products Are Made. Advameg Inc. 2007. Retrieved 2008-03-26.
- ^ a b c d e f g h i j k Lide, David R., ed. (2007–2008). "Zirconium". CRC Handbook of Chemistry and Physics. Vol. 4. New York: CRC Press. p. 42. ISBN 978-0-8493-0488-0.
- ^ Considine, Glenn D., ed. (2005). "Zirconium". Van Nostrand's Encyclopedia of Chemistry. New York: Wylie-Interscience. pp. 1778–1779. ISBN 0-471-61525-0.
- ^ Winter, Mark (2007). "Electronegativity (Pauling)". University of Sheffield. Retrieved 2008-03-05.
- ^ Schnell I and Albers RC (2006). "Zirconium under pressure: phase transitions and thermodynamics". Journal of Physics: Condensed Matter. 18 (5). Institute of Physics: 16. Bibcode:2006JPCM...18.1483S. doi:10.1088/0953-8984/18/5/001.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Day, Charles (2001). "Second Material Found that Superconducts in a Ferromagnetic State". Physics Today. 54 (9). American Institute of Physics: 16. Bibcode:2001PhT....54i..16D. doi:10.1063/1.1420499.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ a b Peterson, John; MacDonell, Margaret (2007). "Zirconium". Radiological and Chemical Fact Sheets to Support Health Risk Analyses for Contaminated Areas (PDF). Argonne National Laboratory. pp. 64–65. Retrieved 2008-02-26.
- ^ a b "Zirconium and Hafnium" (PDF). Mineral Commodity Summaries. US Geological Survey: 192–193. 2008. Retrieved 2008-02-24.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Ralph, Jolyon and Ralph, Ida (2008). "Minerals that include Zr". Mindat.org. Retrieved 2008-02-23.
{{cite web}}: CS1 maint: multiple names: authors list (link) - ^ Callaghan, R. (2008-02-21). "Zirconium and Hafnium Statistics and Information". US Geological Survey. Retrieved 2008-02-24.
- ^ a b c d Nielsen, Ralph (2005) "Zirconium and Zirconium Compounds" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim. doi:10.1002/14356007.a28_543
- ^ a b c Stwertka, Albert (1996). A Guide to the Elements. Oxford University Press. pp. 117–119. ISBN 0-19-508083-1.
- ^ Zardiackas, Lyle D.; Kraay, Matthew J. and Freese, Howard L. (1 January 2006). Titanium, niobium, zirconium and tantalum for medical and surgical applications. ASTM International. pp. 21–. ISBN 978-0-8031-3497-3. Retrieved 18 March 2011.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Brady, George Stuart; Clauser, Henry R. and Vaccari, John A. (24 July 2002). Materials handbook: an encyclopedia for managers, technical professionals, purchasing and production managers, technicians, and supervisors. McGraw-Hill Professional. pp. 1063–. ISBN 978-0-07-136076-0. Retrieved 18 March 2011.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ a b "Zirconia". AZoM.com. 2008. Retrieved 2008-03-17.
- ^ Gauthier, V.; Dettenwanger, F.; Schütze, M. (2002-04-10). "Oxidation behavior of γ-TiAl coated with zirconia thermal barriers". Intermetallics. 10 (7). Frankfurt, Germany: Karl Winnacker Institut der Dechema: 667–674. doi:10.1016/S0966-9795(02)00036-5.
- ^ Wilkinson, G. and Birmingham, J.M. (1954). "Bis-cyclopentadienyl Compounds of Ti, Zr, V, Nb and Ta". J. Am. Chem. Soc. 76 (17): 4281–4284. doi:10.1021/ja01646a008.
{{cite journal}}: CS1 maint: multiple names: authors list (link)Rouhi, A. Maureen (2004-04-19). "Organozirconium Chemistry Arrives". Science & Technology. 82 (16). Chemical & Engineering News: 36–39. doi:10.1021/cen-v082n015.p035. ISSN 0009-2347. Retrieved 2008-03-17. - ^ Wailes, P. C. and Weigold, H. (1970). "Hydrido complexes of zirconium I. Preparation". Journal of Organometallic Chemistry. 24 (2): 405–411. doi:10.1016/S0022-328X(00)80281-8.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Hart, D. W. and Schwartz, J. (1974). "Hydrozirconation. Organic Synthesis via Organozirconium Intermediates. Synthesis and Rearrangement of Alkylzirconium(IV) Complexes and Their Reaction with Electrophiles". J. Am. Chem. Soc. 96 (26): 8115–8116. doi:10.1021/ja00833a048.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Krebs, Robert E. (1998). The History and Use of our Earth's Chemical Elements. Westport, Connecticut: Greenwood Press. pp. 98–100. ISBN 0-313-30123-9.
- ^ Hedrick, James B. (1998). "Zirconium". Metal Prices in the United States through 1998 (PDF). US Geological Survey. pp. 175–178. Retrieved 2008-02-26.
- ^ Luc Gillon (1979). Le nucléaire en question, Gembloux Duculot, French edition, 240 pp.
- ^ Meier, S. M.; Gupta, D. K. (1994). "The Evolution of Thermal Barrier Coatings in Gas Turbine Engine Applications". Journal of Engineering for Gas Turbines and Power. 116: 250. doi:10.1115/1.2906801.
- ^ Heuveling, Derek A.; Visser, Gerard W. M.; Baclayon, Marian; Roos, Wouter H.; Wuite, Gijs J. L.; Hoekstra, Otto S.; Leemans, C. René; de Bree, Remco; van Dongen, Guus A. M. S. (2011). "89Zr-Nanocolloidal Albumin–Based PET/CT Lymphoscintigraphy for Sentinel Node Detection in Head and Neck Cancer: Preclinical Results". The Journal of Nuclear Medicine. 52 (10): 1580–1584. doi:10.2967/jnumed.111.089557. PMID 21890880.
- ^ van Rij, Catharina M.; Sharkey, Robert M.; Goldenberg, David M.; Frielink, Cathelijne; Molkenboer, Janneke D. M.; Franssen, Gerben M.; van Weerden, Wietske M.; Oyen, Wim J. G.; Boerman, Otto C. (2011). "Imaging of Prostate Cancer with Immuno-PET and Immuno-SPECT Using a Radiolabeled Anti-EGP-1 Monoclonal Antibody". The Journal of Nuclear Medicine. 52 (10): 1601–1607. doi:10.2967/jnumed.110.086520. PMID 21865288.
- ^ Ruggiero, A; Holland, JP; Hudolin, T; Shenker, L; Koulova, A; Bander, NH; Lewis, JS; Grimm, J (2011). "Targeting the internal epitope of prostate-specific membrane antigen with 89Zr-7E11 immuno-PET". The Journal of Nuclear Medicine. 52 (10): 1608–15. doi:10.2967/jnumed.111.092098. PMC 3537833. PMID 21908391.
- ^ Verel, I; Visser, GW; Boellaard, R; Stigter-Van Walsum, M; Snow, GB; Van Dongen, GA (2003). "89Zr immuno-PET: Comprehensive procedures for the production of 89Zr-labeled monoclonal antibodies" (PDF). J Nucl Med. 44 (8): 1271–81. PMID 12902418.
- ^ Perk, L, "The Future of Immuno-PET in Drug Development Zirconium-89 and Iodine-124 as Key Factors in Molecular Imaging", Amsterdam, Cyclotron, 2009.
- ^ "Zirconium". International Chemical Safety Cards. International Labour Organization. October 2004. Retrieved 2008-03-30.
- ^ "Zirconium Compounds". National Institute for Occupational Health and Safety. 2007-12-17. Retrieved 2008-02-17.
External links
- Chemistry in its element podcast (MP3) from the Royal Society of Chemistry's Chemistry World: Zirconium
- Zirconium at The Periodic Table of Videos (University of Nottingham)
Template:Link FA
Cite error: There are <ref group=lower-alpha> tags or {{efn}} templates on this page, but the references will not show without a {{reflist|group=lower-alpha}} template or {{notelist}} template (see the help page).

