Lowercase sigmabot III (talk | contribs) m Archiving 2 discussion(s) to Wikipedia talk:WikiProject Chemistry/Archive 37) (bot |
Biscuittin (talk | contribs) →RfC at Talk:Climate change denial: new section |
||
| Line 576: | Line 576: | ||
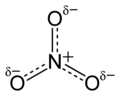
Would someone please check the report of a problem in a formula at [[Talk:Acid dissociation constant#Reaction equilibrium formula error]]. [[User:Johnuniq|Johnuniq]] ([[User talk:Johnuniq|talk]]) 21:55, 22 January 2016 (UTC) |
Would someone please check the report of a problem in a formula at [[Talk:Acid dissociation constant#Reaction equilibrium formula error]]. [[User:Johnuniq|Johnuniq]] ([[User talk:Johnuniq|talk]]) 21:55, 22 January 2016 (UTC) |
||
== RfC at Talk:Climate change denial == |
|||
There is a RfC at [[Talk:Climate change denial]]. Please contribute if you are interested. [[User:Biscuittin|Biscuittin]] ([[User talk:Biscuittin|talk]]) 00:10, 28 January 2016 (UTC) |
|||
Revision as of 00:10, 28 January 2016
| Chemistry Project‑class | |||||||
| |||||||
| Article alerts |
|---|
|
Articles for deletion
Proposed deletions
Categories for discussion
Templates for discussion
Redirects for discussion
Featured article candidates
Good article nominees
Requests for comments Requested moves
Articles to be merged
Articles to be split
Articles for creation
|
Discussion of the WikiProject Chemistry - Please add your comment and discussion here. Older discussions are archived.
This discussion page is about the Chemistry project itself, for detailed, in-depth discussions about specific topics, you'd be best served at the talk page of the specific subject, e.g., Chemicals, Chemical infoboxes, etc. There is also an image request page which might be of interest to you.
New tool?
Forgive my presumptuousness, but I'm not sure where else to put this. In my work, I am producing a wiki documenting a lot of historic materials science and therefore one using a lot of chemical notation, and the layout performed by Chem wasn't quite what I was looking for. I started to improve upon the {{Chem}} template, but after conferring with some wikipedia higher-ups, I switched to a parser keyword instead so that it would interact more nicely with stacked templates and infoboxes.
I'm wondering if you would be kind enough to review and kibbitz on the layout and rendering (as well as any features you can think to add)
Here's a page talking about the update:
Perhaps the next feature would be one that adds a comment when the equation isn't balanced?
You can contact me directly via wikipedia's email here
Thanks for your time! Riventree (talk) 16:27, 6 June 2015 (UTC)
- Can you clarify how this tool differs from {{chem}}? Is it intended as an update of the existing template or as a separate, alternative tool? ChemNerd (talk) 20:14, 10 June 2015 (UTC)
It's an alternative, not a new version of the chem template. There were three main issues I wanted to address:
- Ease of use / error blocking (cut and paste input text:
{{Chem|H|2|S||O|4}}is awkward and error prone{{#Chem: H2SO4}}produces the same (actually, slightly better) layout. - Better visual layout (subscripting and sub-subscripting, better css control, line height, etc)
- Annotated-arrow support (where you have the reaction catalysts and conditions listed)
There's a bunch of other stuff too. Crystal notation, ΔH notation, etc. Have a look at the link above, it has a fairly good description of the features. Riventree (talk) 20:45, 10 June 2015 (UTC)
- I can't follow all of this. 1. {{Chem}} has the issue that it hightens a line (more whitespace above). 2. We need a general template to produce the formula for HTML (how difficult can it be). In {{Infobox drug}}, a 'bolded font coloring' is used, see aspirin (ouch).
- Anyway, I'm not here to support an non-wiki template building. What's the plan? -DePiep (talk) 21:35, 10 June 2015 (UTC)
- No, {{chem}} is terrible, but a good solution before we got Lua. Riventrees version is very nice, instead of adding a lot of | to the formula, you just parse the chemical formula and it converts a lot of stuff. It could be very nice if the version was published on enwiki somewhere in the module namespace. In matter of fact I have been working on a similar version, which I will upload at module:Chem2/template:Chem2 to inspiration. (I do not think I will finish it, after seeing the other version). Christian75 (talk) 21:48, 10 June 2015 (UTC)
- See you all at Template:Chem/sandbox, andn Template:Chem/testcases. -DePiep (talk) 21:54, 10 June 2015 (UTC)
- Its not a replacement for chem, its a new version. I uploaded my beta version to {{chem2}}. It shouldnt be used yet, but you can write things like {{chem2|CH3\i{13}CH2CH3}}, {{chem2|SO4(2-)}} and {{chem2|\h{5}[HC\tC\qMn](2+)}} which gives CH3CH2CH3, SO2−4 and η5-−[HC≡C≣Mn]2+. Christian75 (talk) 22:06, 10 June 2015 (UTC)
- Looks nice. Anything other than the font when formulas are written with the "math" format. BTW, whats the deal with a "deep subscript"? Doesn't seem to be standard practice, but maybe the consensus is for it. --Smokefoot (talk) 22:26, 10 June 2015 (UTC)
- Thanks, its not finished. About: "deep subscript" - its just a beta version, and can be changed to follow enwikis MOS. I am working on it (locally), and made it recognize equations too. (It was published on enwiki because I thought "the new tool" was a Lua module (and was finished). I will write a note when its "finished", so the mark up can be changed, maybe someone have better ideas than using \s, \d, \t, \q (for bonds), * for crystal water, etc... Christian75 (talk) 11:00, 11 June 2015 (UTC)
- Looks nice. Anything other than the font when formulas are written with the "math" format. BTW, whats the deal with a "deep subscript"? Doesn't seem to be standard practice, but maybe the consensus is for it. --Smokefoot (talk) 22:26, 10 June 2015 (UTC)
- Its not a replacement for chem, its a new version. I uploaded my beta version to {{chem2}}. It shouldnt be used yet, but you can write things like {{chem2|CH3\i{13}CH2CH3}}, {{chem2|SO4(2-)}} and {{chem2|\h{5}[HC\tC\qMn](2+)}} which gives CH3CH2CH3, SO2−4 and η5-−[HC≡C≣Mn]2+. Christian75 (talk) 22:06, 10 June 2015 (UTC)
- See you all at Template:Chem/sandbox, andn Template:Chem/testcases. -DePiep (talk) 21:54, 10 June 2015 (UTC)
- No, {{chem}} is terrible, but a good solution before we got Lua. Riventrees version is very nice, instead of adding a lot of | to the formula, you just parse the chemical formula and it converts a lot of stuff. It could be very nice if the version was published on enwiki somewhere in the module namespace. In matter of fact I have been working on a similar version, which I will upload at module:Chem2/template:Chem2 to inspiration. (I do not think I will finish it, after seeing the other version). Christian75 (talk) 21:48, 10 June 2015 (UTC)
Sooo.... I'm new to contributing to MediaWiki/Wikipedia on the infrastructure side. How do I do what Christian75 was talking about? "Publish somewhere on enwiki in the module namespace"? I have the php, css, test page, and doc page ready to go... I just don't know where to put it where people can check it out.
Help, help! Send instructions or a link (I promise I read everywhere I could think of, but apparently not the right places yet)
Riventree (talk) 01:56, 11 June 2015 (UTC)
- Can't help you, {{Chem2}} does not have a testpage. BTW, what is wrong with {{Chem}} anyway? DePiep (talk) 02:10, 11 June 2015 (UTC)-DePiep (talk) 02:10, 11 June 2015 (UTC)
- Chem: You cant copy paste a formula, but have to add a lot of pipes (|), its hard to read the markup, you can only have one charge, and so on Christian75 (talk) 11:00, 11 June 2015 (UTC)
- @Riventree: Ok, I thought it was a Lua module, but its an extension? If so, its hard to get it deployed on Wikipedia (security, efficiency and usability). But take a look at mediawikiwiki:Writing an extension for deployment. Christian75 (talk) 11:00, 11 June 2015 (UTC)
- Christian75: Thank you for your help. I've applied for access and I'm setting up the appropriate pages
- DePiep: Thank you for your kind words and keen insight.
Riventree (talk) 05:08, 14 June 2015 (UTC)
Current options
{{Chem|H|2|O}}→ H
2O{{Infobox drug/chem styled|O=1|H=2}}→ Template:Infobox drug/chem styled (used by {{Infobox drug}}{{Chembox Elements/molecular formula|O=1|H=2}}→ H2O (used by {{Chembox}}){{Chem2|H2O}}→ H2O (Christian75 21:48 below)
- Discuss
-
- I still do not get why we need an external development site (though all these wiki-options, live today, are bad somehow I agree). -DePiep (talk) 20:21, 14 June 2015 (UTC)
- Its not an external site he want us to use. The external link was to his own wiki where he used the extension to MediaWiki he had programmed in PHP etc - he wants us (Wikipedia) to use it too. He asked how to get it deployed on Wikipedia. That said, {{chem}} are used in the articles too (alot). My Lua (alpha) version can do things like: {{chem2|3H2 + N2 -> 2NH3}} which gives: 3H2 + N2 → 2NH3. It could be very nice if the infobox just got the formula (eg. |formula = CaCO3, and then showed the formula correct and calculated the molar mass too (with no need of Ca=|C=|O=3). A lot of inorganic compounds need both a formula and the Ca=stuff because the empirical formula doesnt looks good for inorganic compounds (e.g. CCaO3 for CaCO3. Christian75 (talk) 21:48, 14 June 2015 (UTC)
- I still do not get why we need an external development site (though all these wiki-options, live today, are bad somehow I agree). -DePiep (talk) 20:21, 14 June 2015 (UTC)
{{Chem2|X2+}} → X+2 seems to be ambiguous: It may stand for X2+ and X2+. How do we deal with that issue? --Leyo 09:21, 13 July 2015 (UTC)
{{Chem2|X2+}}→ X+2{{Chem2|X+2}}→ X+2- Looks like this needs documentation (cannot disambiguate this by some 'natural' typing order). In other templates we have
|charge=, but that would not work in the full reaction option Chris75 has build. -DePiep (talk) 12:26, 13 July 2015 (UTC)- This wasnt Leyos point. X+2 doesnt have any chemical meaning, but X2+ could be either X+2 or X2+. Christian75 (talk) 13:58, 13 July 2015 (UTC)
- It has some kind of documentation (see module:Chem2/doc), but I havent finish the module yet. However, by design "index" has higher priority than charge, so {{chem2|I3-}} gives I−3 but can be written as {{chem2|I3(-)}} too. My plan was (is) to finish it "soon", and then "publish" it in selected wikiprojects (and ask for further sugestions). I am stilling missing the feature to auto link to elements probably with the option |auto=yes. I do not know how rich of features it should be (eg. it could automatic format things like (s), (g) and (l). I really like the module :-) because you can take an equation like: 2 Fe + 6 HCl = 2 FeCl3 + 3 H2 from the big internet, and do: {{chem2|2 Fe + 6 HCl -> 2 FeCl3 + 3 H2}} which gives: 2 Fe + 6 HCl → 2 FeCl3 + 3 H2 . Christian75 (talk) 13:23, 13 July 2015 (UTC)
- I should have used X2+ as an example:
{{Chem2|X2+}}→ X+2{{Chem2|X(2+)}}→ X2+ Christian75 (talk) 14:02, 13 July 2015 (UTC)
- I was just playing with options. When saying "needs documentation" that means one cannot remember it from natural typing (H2O does not need, X2+ does). -DePiep (talk) 14:21, 13 July 2015 (UTC)
- Just remember it as charges needs parenthesis ... Christian75 (talk) 14:29, 13 July 2015 (UTC)
- @Christian75 and DePiep: Is the module ready to be used (soon)? BTW: Why isn't it moved to Module:Chem? --Leyo 18:30, 1 August 2015 (UTC)
- I have more time next week, and can look at it again. Its called module:chem2 so it has same name as the template template:Chem2 Christian75 (talk) 19:52, 1 August 2015 (UTC)
- @Leyo: Its nearly "finished" (but really need some cleanup). I added the possibility to auto link to elements, {{chem2|auto=yes|Na2HgOHC6HOBrC6H2OBrOCHC6H4CO2}} gives Na2HgOHC6HOBrC6H2OBrOCHC6H4CO2. The typograhy should be discussed somewhere. I am in doubt if I should add a ^ so its possible to write R^1R^2 instead of using ' like R'R''. Christian75 (talk) 22:02, 1 August 2015 (UTC)
- Can you provide a few examples? --Leyo 23:37, 9 August 2015 (UTC)
- Sorry fot the long delay. I think I finished it more or less yesterday (found one bug). My intention is to write a note on different WikiProjects (tomorrow). There is a lot of examples at module:chem2/doc Christian75 (talk) 13:35, 29 August 2015 (UTC)
- Is it ready for use though? --Leyo 22:02, 18 November 2015 (UTC)
- @Christian75 and DePiep: Is the module ready to be used (soon)? BTW: Why isn't it moved to Module:Chem? --Leyo 18:30, 1 August 2015 (UTC)
- Just remember it as charges needs parenthesis ... Christian75 (talk) 14:29, 13 July 2015 (UTC)
- I was just playing with options. When saying "needs documentation" that means one cannot remember it from natural typing (H2O does not need, X2+ does). -DePiep (talk) 14:21, 13 July 2015 (UTC)
Halogenation halohydrins

While they fail the IUPAC definition would editors here concider compounds such as 2,2,2-trichloroethanol (structure shown) to be halohydrins or not? I ask because we seem to have quite a lot of compounds like this and I'm trying to figure out how to categorise them. --Project Osprey (talk) 08:19, 8 October 2015 (UTC)
- I think that category will do. The definition looks like there should be only one halogen atom though. Graeme Bartlett (talk) 05:07, 16 October 2015 (UTC)
- Indeed it does. However I expect they share a similar chemistry, intramolecular cyclisation to give the dichloroepoxide is known and forms the basis of the Corey-Link Reaction. On the other hand some editors do feel strongly about us deviating from IUPAC definitions. --Project Osprey (talk) 09:00, 16 October 2015 (UTC)
- No idea about the categorization, but Corey–Link reaction now exists (we didn't seem to have it in any WP language). DMacks (talk) 20:31, 16 October 2015 (UTC)
- The definitions are always difficult to read, mainly because of the misunderstanding of what these definitions define. 1,1,2,2-tetrachloroethene is an alkene, despite alkenes being defined as "Acyclic branched or unbranched hydrocarbons having one carbon–carbon double bond and the general formula CnH2n". It is the core part that defines the functionality (the basic carbon-fragment), in which nothing to everything can be substituted (the hydrogens, and even the carbons). Here in 2,2,2-trichloroethanol we have a halohydrin ('Cl-C-C-OH'), where 2 hydrogens on the chlorine-bearing carbon are replaced by chlorides. --Dirk Beetstra T C 06:42, 21 October 2015 (UTC)(changed typo --Dirk Beetstra T C 10:54, 6 December 2015 (UTC))
- @Beetstra: I am confused by "1,1,2,2-tetrachloroethane is an alkene" as it is not an alkene but a substituted alkane. Did you mean 1,1,2,2-tetrachloroethene is an alkene (ie. that your post has a typo), which would make sense, or am I missing something? I concur that 2,2,2-trichloroethanol is definitely a halohydrin, though. :) EdChem (talk) 04:17, 5 December 2015 (UTC)
- The definitions are always difficult to read, mainly because of the misunderstanding of what these definitions define. 1,1,2,2-tetrachloroethene is an alkene, despite alkenes being defined as "Acyclic branched or unbranched hydrocarbons having one carbon–carbon double bond and the general formula CnH2n". It is the core part that defines the functionality (the basic carbon-fragment), in which nothing to everything can be substituted (the hydrogens, and even the carbons). Here in 2,2,2-trichloroethanol we have a halohydrin ('Cl-C-C-OH'), where 2 hydrogens on the chlorine-bearing carbon are replaced by chlorides. --Dirk Beetstra T C 06:42, 21 October 2015 (UTC)(changed typo --Dirk Beetstra T C 10:54, 6 December 2015 (UTC))
- No idea about the categorization, but Corey–Link reaction now exists (we didn't seem to have it in any WP language). DMacks (talk) 20:31, 16 October 2015 (UTC)
- Indeed it does. However I expect they share a similar chemistry, intramolecular cyclisation to give the dichloroepoxide is known and forms the basis of the Corey-Link Reaction. On the other hand some editors do feel strongly about us deviating from IUPAC definitions. --Project Osprey (talk) 09:00, 16 October 2015 (UTC)
Amino acid Representations
I have noticed that many, if not all, the amino acids on their individual pages and on the amino acid page are represented in their non-ionic form. I feel this is misleading, or misinforming, as it is the form least likely found in nature. The Zwitterionic form would be the best form to include, in my opinion. I would be happy to change the structures if the community doesn't mind. I have noticed there has been some discussion on the amino acid talk page, and on some of the individual pages, but nothing has been changed. I asked about this in the WikiProject: Molecular and Cell Biology as well, but some of the discussion already on talk pages seemed to come from this project as well. So I am asking here also. Htienson (talk) 22:01, 20 October 2015 (UTC)
- Seems like a good idea to me. You'd have the opportunity to depict them with uniform drawing settings, in the event that they are not already that way. --Smokefoot (talk) 00:02, 21 October 2015 (UTC)
- I think that ideally we'd show both forms. The 'classic' non-ionic depiction is the most commonly used, even in text books, failing to show it may confuse our lay readers. --Project Osprey (talk) 08:29, 21 October 2015 (UTC)
- I agree with Project Osprey here - please show both. Showing the zwitterion is more correct, but these compounds are called 'amino acids' and not 'ammonium carboxylates' - a lay reader would/might maybe expect an amino-group and an acid-group, the zwitterionic form might be confusing to them (I presume that this concept is however properly explained in the article). --Dirk Beetstra T C 09:20, 21 October 2015 (UTC)
- In principle, yes. In practice, I am not so sure. I assume you are referring to the graphic in the {{chembox}}. The down side of these graphics is that they cause the size of these already large infoboxes to grow even larger. This is especially a problem when viewing the article on a mobile device. One has to scroll down forever until one reaches the lead. If we include 2D depictions of both the neutral and zwitterionic forms plus the 3D structure, this really starts to get out of hand. In a related issue, I question why we are including 3D depictions at all. These are arbitrary selected conformations and IMHO, the ball and sticks depictions plus dashed lined for aromatic bonds are really ugly. I think these 3D depictions should be removed. Boghog (talk) 11:51, 21 October 2015 (UTC)
- While I think a uniform depiction across multiple articles is a good idea, choosing which representation style is not easy. The functional groups in amino acids have different protonation states in different environments (aqueous vs solid, pH dependence, etc). Simple zwitterionic forms (ammonium carboxylates) are not always the accurate form when considering the physiological environment where amino acid ionization states are most relevant. And since 2D representations of chemical compounds already have many simplifications related to structure and bonding built into them, we shouldn't necessarily strive for the highest degree of scientific "accuracy" with them. They are meant as a kind of shorthand. As is done in most text books, I think it makes most sense from an educational perspective to show them in amino acid forms first, then describe zwitterions and more complex biological ionization states in the text. -- Ed (Edgar181) 13:56, 21 October 2015 (UTC)
Thank you for your feedback. As amino acids are biological molecules I think the most relevant form is the proper ionization state under most normal physiological conditions (i.e. pH 7.4). From the discussion it sounds like a good compromise would be showing the physiologically relevant form next to the non-ionic form, without the 3-D structure at the top of the page. I also tend to find these difficult to look at and not very informative except in very specific settings. I will also make sure to add something early on in the introduction that explains the two different structures. Any other suggestions or comments? Htienson (talk) 18:41, 3 November 2015 (UTC)
Lyndon Emsley
Hi, I was looking for some news in the NMR field and I've discovered that my old Ph.D. supervisor has received another award from the RSC. He is Lyndon Emsley: cited on Nature, published a Science this year, recipient of the Grand Prix Charles-Leopold Mayer in 2012, now head of the RMN lab at EPFL, member of Academia Europaea, h-index of 63 etc etc...
I think I could make a decent article about him in few minutes and I don't think there is any "conflict of interest", my Ph.D. ended 3 years ago (and I was already a skilled wikipedian at the time and I never though of writing an article about him).
Do you see any potential issue? May I start my draft?--Alexmar983 (talk) 02:34, 21 October 2015 (UTC)
- You could start at Draft:Lyndon Emsley and base the content on what others have written rather than your own knowledge. Starting a draft should not be a problem. Graeme Bartlett (talk) 07:49, 21 October 2015 (UTC)
- While your intentions are admirable, I just think that you have a conflict of interest and if his contributions were so key to understanding the substance of Wikipedia content, someone else would start the article. That having been said, many biographical articles on comparable individuals appear to be written by either the person themselves or their appointees, which is pretty lame in my book. --Smokefoot (talk) 09:59, 21 October 2015 (UTC)
- I'm an expert user and I know how to find secondary sources. And I'm no appointee, after my Ph.D. I worked in a totallty different area so I don't see why it should be lame. What conflict of interest is that? someone else would start the article if someone else wuold know about ssNMR, which they don't because it is still a very skilled area, much more complicated than liquid NMR, so you see it takes some time to work in that field. As skilled wikipedians you know that you should never make the confusion that something will be written just if it is relevant. It will b written if there is someone who can write about it. And if it is just one person, that's just a specific competence, otherwise every situation where only one guy can write about it becomes a conflict of interest. Instead of reducing an asymmetric growth filling gaps, you therefore are incentivating it.
- The truth is, if I had wirtten it without asking, you wouldn't have even known I was his Ph.D. years ago :D. But if I say it, you kinda expect something potentially "dirty" or "lame" and that's IMHO really against the true spirit of wikipedia. Who do you think was the guy who wrote the Robert G. Griffin article for example? I think I should come back when there will be less generic sentence and more attention to the real quality.--Alexmar983 (talk) 11:07, 21 October 2015 (UTC)
- "The truth is, if I had wirtten it without asking, you wouldn't have even known" is exactly true and, as I mentioned, the source of many chem biographies. You might be absolutely correct that a readers would be interested in this biography and I am out of touch. In my experience when one asks about a possible COI, it exists even its harmless. On a more positive note, Wikipedia does need help with a lot of NMR spectroscopies, we should have an article for each of the I = 1/2 nuclei plus many others. --Smokefoot (talk) 13:28, 21 October 2015 (UTC)
- I say go ahead and write a draft; it's just not that difficult to write a short biography of an academic without turning it into a puff piece. I'm somewhat familiar with the field and don't mind taking a look at it (but next week at the earliest, too busy this week). And Smokefoot is right, our coverage of NMR in general is very poor, so if you have any interest in working on those articles it'd be a big help. Opabinia regalis (talk) 17:47, 21 October 2015 (UTC)
- It would be beneficial to have an article on Lyndon Emsley and I support the idea of User:Alexmar983 creating a draft. Another bio article of an NMR scientist is at Robert G. Griffin though it's not very informative. Due to his personal knowledge of NMR spectroscopy and of Lyndon Emsley's work, I imagine that Alemxmar983 will be able to come up with something better. Alexmar983 has 32,000 edits on the Italian Wikipedia so he does have some relevant background. EdJohnston (talk) 18:26, 21 October 2015 (UTC)
- my edits on itWiki are mainly related to general maintenance, wikimetrics and help of newcommers with no specific focus on Chemistry. On enWiki I don't edit very much, but I usually insert some sources, sometimes when I'm cleaning my desk from old articles or magazines. I read them one last time and I use them to add few details on wiki. I usually write of "scientific stuff" in English so it's just more natural here. But I should know how to make a decent biography article...--Alexmar983 (talk) 18:45, 21 October 2015 (UTC)
- The core draft is almost done. I am visiting some friends in Lyon this week end (he is not there, he works in Lausanne now), so I should have access to the French magazines and newspapers I stored in the library at the time of the first 1 GHz NMR spectrometer inauguration. There are some descriptions or interviews on some of them, if I remember correctly.
- I know his exact birthday date from an attachment to a grant proposal left on a desk, so sorry but that information remains a secret :D and, even funnier, Lyndon is actually his middle name. Again no source for that and I think he and his family didn't/don't like his first name so they/he never used it. It is a very common name BTW... again, I won't say a word.--Alexmar983 (talk) 20:50, 22 October 2015 (UTC)
- It would be beneficial to have an article on Lyndon Emsley and I support the idea of User:Alexmar983 creating a draft. Another bio article of an NMR scientist is at Robert G. Griffin though it's not very informative. Due to his personal knowledge of NMR spectroscopy and of Lyndon Emsley's work, I imagine that Alemxmar983 will be able to come up with something better. Alexmar983 has 32,000 edits on the Italian Wikipedia so he does have some relevant background. EdJohnston (talk) 18:26, 21 October 2015 (UTC)
- I say go ahead and write a draft; it's just not that difficult to write a short biography of an academic without turning it into a puff piece. I'm somewhat familiar with the field and don't mind taking a look at it (but next week at the earliest, too busy this week). And Smokefoot is right, our coverage of NMR in general is very poor, so if you have any interest in working on those articles it'd be a big help. Opabinia regalis (talk) 17:47, 21 October 2015 (UTC)
- "The truth is, if I had wirtten it without asking, you wouldn't have even known" is exactly true and, as I mentioned, the source of many chem biographies. You might be absolutely correct that a readers would be interested in this biography and I am out of touch. In my experience when one asks about a possible COI, it exists even its harmless. On a more positive note, Wikipedia does need help with a lot of NMR spectroscopies, we should have an article for each of the I = 1/2 nuclei plus many others. --Smokefoot (talk) 13:28, 21 October 2015 (UTC)
- While your intentions are admirable, I just think that you have a conflict of interest and if his contributions were so key to understanding the substance of Wikipedia content, someone else would start the article. That having been said, many biographical articles on comparable individuals appear to be written by either the person themselves or their appointees, which is pretty lame in my book. --Smokefoot (talk) 09:59, 21 October 2015 (UTC)
EdJohnston, Graeme Bartlett, Smokefoot: the draft is complete. I have to expand the "Research" section and find a reliable source for his year of birth.--Alexmar983 (talk) 12:30, 29 October 2015 (UTC)
- no comment so far?--Alexmar983 (talk) 21:30, 29 October 2015 (UTC)
- Looks fine to me. Perhaps a little too detailed (do we really care that he collaborates with Bruker?)--Smokefoot (talk) 02:42, 21 December 2015 (UTC)
- Smokefoot that's one thing I will definitely keep :D CRMN and Bruker have tested together some of the biggest innovations in ssNMR in last decade: the GHz barrier, the 1.3 mm rotors and the 0.7 mm rotors (before he left Lyon)... Now, I still have to refine the part related to science adding more details, so if you want to remove something else from the "career" part, help yourself (there are the external links for that...). The connection with Bruker is however important, it should remains in some way in the text.--Alexmar983 (talk) 23:34, 22 December 2015 (UTC)
- Looks fine to me. Perhaps a little too detailed (do we really care that he collaborates with Bruker?)--Smokefoot (talk) 02:42, 21 December 2015 (UTC)
@Smokefoot, Graeme Bartlett, and EdJohnston: etc. So now Draft:Lyndon Emsley has a bigger scientific part (some articles on high IF journals still have to be cited, more information about paramgnetic protein is possible), and a slightly reduced biography. It may need more calibration or cut. I was thinking to write more biogrphies of NMR spectroscopists in the future so if you have any feedback, I am listening. I have no hurry at all, I am not very present on enWiki so the work is very "diluted".--Alexmar983 (talk) 11:42, 31 December 2015 (UTC)
- The text looks pretty good to me. However I would encourage you to have a reference on each paragraph, and prize. As later it may be reorganised or modified and the source could be very unclear. I assume the source are some of the references already in use. The page is good enough for article space now. We could also have it at WP:DYK if you get references to all paragraphs. Graeme Bartlett (talk) 12:48, 31 December 2015 (UTC)
- Single reference will be added, no worries. Some of the information are in the external links, some in some articles I still have to add. I am doing it in a non-linear way also beacuse it helps me to produce a more rephrased text. In the end these technical descriptions cannot be changed too much and if I added information every new source together with the source they are related, the overall dscription will be even more similar to the original texts. So in this phase of my editing I read 3-4 articles together with the Klaus CV pdf and I rewrite them and often I forget to insert one or two of them. The more I refine it, the better will be sourced.
- I would also avoid WP:DYK. I know it is not a bad article and I hope it will help to set up a working standard for descriptions of professors with an "above average" academic status, at least for me, but let's be honest: in the end the NMR community is quite small and on the home page it will be noticed too much. I honestly hope to write more similar articles before putting them partially in evidence on the main page.--Alexmar983 (talk) 13:15, 31 December 2015 (UTC)
I have prepared the red links for the new page, and there are some sources to be added in the protein part. Whilst refining the article, I am starting Draft:Geoffrey Bodenhausen. That also will take some time.--Alexmar983 (talk) 12:53, 4 January 2016 (UTC)
- Opabinia regalis I forgot to ping you last week--Alexmar983 (talk) 12:59, 4 January 2016 (UTC)
- I admit I only skimmed, but the draft looks good to me. Consider submitting it to WP:DYK when you move it to mainspace. Surprised to see Martin Blackledge is a redlink! Opabinia regalis (talk) 18:42, 4 January 2016 (UTC)
- Ha, I only skimmed this page too. DYK gets all kinds of obscure stuff; I put scientist biographies there all the time. They usually get ~1000 views for the time they're live - a big bump from their normal rate, but an absolutely minuscule fraction of total main-page traffic. (Even high-visibility FAs usually get a small fraction of main-page traffic; we are for some reason not very responsive to the fact that our readers very rarely care about anything we put there. So you might as well use the space for things you like :) Opabinia regalis (talk) 21:24, 4 January 2016 (UTC)
- (confl.)Opabinia regalis me too. I guess Blackledge, Lyndon or Bodenhausen... they all worked outside UK/USA. Non-anglophone were therefore less involved in creating these articles. In the end, 75%-80% of these articles are written by students and in certain countries there are less interaction with the academia than in others. Ivano Bertini, the chemist not the astronomer, he worked a lot with German and he has an article on dewiki, because Germany is a wikipedianly active country. France or Switzerland much less.
- In any case the structural biology part in the article is the less developped, mainly because that's the part I've worked with Lyndon. So I was trying to keep it as simple as possible, that's basically why is missing. I'm going to complete it in the following days.
- I found in my archive an image of Lyndon taken at a restaurant in 2009 which was not bad. In any case I wrote him an email this afternoon to inform that an article about him was probably closed to be "published", he offered to upload an image on commons that he bought from a professional photographer. It's the one in the article now, and now we have a commons cat too.
- I have also created this evening the item d:Q21970614 on data.
- I think it can be moved to ns0 by the end of the week. --Alexmar983 (talk) 21:33, 4 January 2016 (UTC)
- Sounds great! Just make sure he understands the licensing/copyright tagging/etc. on commons; they don't always make it easy.
- Bertini doesn't have an article either?? Wow. Should be plenty of source material from all the obituaries. Opabinia regalis (talk) 21:41, 4 January 2016 (UTC)
- yes, I know... on commons they don't make it easy. I told him OTRS was an option but in the end Lyndon owns the copyright of that picture, so he saw no reason not to upload it directly. He was honest about the photographer who took it since the beginning, and there's no reason to doubt him. I am also there if someone wants to know more.--Alexmar983 (talk) 22:08, 5 January 2016 (UTC)
- Is James Penner-Hahn another potential biography subject? EdChem (talk) 00:40, 5 January 2016 (UTC)
- Personally, I've never heard of him. So here is what I get from google. From the bibliographic perspective (VIAF or googlebooks he's not bad at all (Emsley for example published less books than other collegues). To me the difference in any case is not the scientific production stricto sensu, I don't know the other wikifellows but I have a "itWikipedia threshold" which is traditionally more selective than enWikipedia. What made me write about Emsley were mainly the awards and the fact that he was at centre of some news, those were the key factors. Here I mainly see publications and grants. Also: Emsley's ENC grant was very competitive, in this case I don't have a clear idea about the "distinction" of his grants, you need someone more involved in the USA academia.--Alexmar983 (talk) 01:11, 5 January 2016 (UTC)
- EdChem in any case if the professor is stored as an item on a general repository (and he is), you can start creating the wikidata item: d:Q21993995--Alexmar983 (talk) 21:05, 7 January 2016 (UTC)
- Penner-Hahn definitely meets the local notability guidelines, though I don't know much about him and agree he seems less high-profile than Emsley. I see a whole lot of the protein dynamics/relaxation people are redlinks too - Christian Griesinger, Lewis Kay, Ann McDermott, Art Palmer - and also some RNA people like Hashim al-Hashimi and Jody Puglisi. Opabinia regalis (talk) 21:43, 7 January 2016 (UTC)
So It has been moved. I am reading Wikipedia:Drafts#Deleting_a_draft and I am not sure what to do with the draft redirect. Can someone link me to the best guidelines? Thanks. Also, I've inserted a quality template in the article talk.--Alexmar983 (talk) 18:19, 7 January 2016 (UTC)
- User:Compassionate727 revised it. he also said it needs more inline citations or footnotes. At the moment, I don't think I can add more. --Alexmar983 (talk) 20:01, 7 January 2016 (UTC)
- Also, I am quite sure it is not a start article. It's quite complete... ;)--Alexmar983 (talk) 20:37, 7 January 2016 (UTC)
- Thanks Alexmar983! I took care of the redirect - in general just tag it for speedy deletion as G6 or G7 and someone will do it eventually. I also removed the footnotes tag - people doing page curation/new page patrol tend to overdo it on the cleanup tags. The talk page quality ratings are almost completely meaningless IMO, so I wouldn't worry about those. Opabinia regalis (talk) 21:43, 7 January 2016 (UTC)
- I am not worried Opabinia regalis. I find it funny :D. I'll refine the article in the following weeks and I will create some new ones (Bodenhousen is on the way, followed by Bertini). About the DYK, I leave it someone else. I can help of course, I just don't want to look as if I am "pushing" it. Also, the instructions looked pretty long.--Alexmar983 (talk) 21:49, 7 January 2016 (UTC)
- Opabinia regalis these cn insrted by User:Compassionate727 are all in the external links. On itWiki I'm considered rigid abot inline citation, and itWiki is rigid about inline citation, but I don't support this excessive requirement on inline citation if related to non-critical details cited in external link. It does not add any real quality, in my opinion, especially when you end up with thousands of older article with no citation at all. I suggest to remove them if you prefer this way. --Alexmar983 (talk) 20:17, 11 January 2016 (UTC)
- For better or worse, it is almost impossible to dispute a citation request in a WP:BLP, even for obvious or trivial information. (I'm personally hoping for a technical solution to the unreadable-footnotes and impenetrable-wikitext problems this generates, but haven't seen any progress. If the clutter in the edit window bothers you, you could try list-defined referencing, which works more like a typical reference manager.) Most people will tell you a truism that anything usable as a reference should be in the reference list, not in external links. The easiest thing to do is just add them. Opabinia regalis (talk) 23:00, 11 January 2016 (UTC)
- Just to be clear: when I write "remove them", I mean "remove the lines", not the "cn's". I don't consider those details critical so I am not specifically looking for in line sources at the moment. I am just refining the article where it matters (IMHO). If in some days I haven't found any specific source whilst looking for something else, which I would have added in any case, you can remove those cn-ed lines, Opabinia regalis.
- BTW, it is a truism in some wikis than everything used as a general scaffolding of an article should not be forced in a in inline citations just for the horror vacui of a missing footnotes, especially for few details. it's unnatural. CVs of professors on a university website are where they should be, in the external link paragraph, but they are in any case reliable for minor details. Noone will survive in the academia posting a fake information on a public website.--Alexmar983 (talk) 23:32, 11 January 2016 (UTC)
- Ah, sorry, I misunderstood; I'll remove whatever's left in a couple of says then. IMO the EPFL move should be cited and the other stuff isn't so important. Opabinia regalis (talk) 07:43, 12 January 2016 (UTC)
- For better or worse, it is almost impossible to dispute a citation request in a WP:BLP, even for obvious or trivial information. (I'm personally hoping for a technical solution to the unreadable-footnotes and impenetrable-wikitext problems this generates, but haven't seen any progress. If the clutter in the edit window bothers you, you could try list-defined referencing, which works more like a typical reference manager.) Most people will tell you a truism that anything usable as a reference should be in the reference list, not in external links. The easiest thing to do is just add them. Opabinia regalis (talk) 23:00, 11 January 2016 (UTC)
- Opabinia regalis these cn insrted by User:Compassionate727 are all in the external links. On itWiki I'm considered rigid abot inline citation, and itWiki is rigid about inline citation, but I don't support this excessive requirement on inline citation if related to non-critical details cited in external link. It does not add any real quality, in my opinion, especially when you end up with thousands of older article with no citation at all. I suggest to remove them if you prefer this way. --Alexmar983 (talk) 20:17, 11 January 2016 (UTC)
- I am not worried Opabinia regalis. I find it funny :D. I'll refine the article in the following weeks and I will create some new ones (Bodenhousen is on the way, followed by Bertini). About the DYK, I leave it someone else. I can help of course, I just don't want to look as if I am "pushing" it. Also, the instructions looked pretty long.--Alexmar983 (talk) 21:49, 7 January 2016 (UTC)
- Thanks Alexmar983! I took care of the redirect - in general just tag it for speedy deletion as G6 or G7 and someone will do it eventually. I also removed the footnotes tag - people doing page curation/new page patrol tend to overdo it on the cleanup tags. The talk page quality ratings are almost completely meaningless IMO, so I wouldn't worry about those. Opabinia regalis (talk) 21:43, 7 January 2016 (UTC)
More suspect articles by same editor
Here is another questionable article - Biosynthetic mechanism. Also by by User:Carolineneil. Maybe some patient editor could tap them on the shoulder and plea for more discussion before creating such articles. Possibly this stuff is the result of class assignments.--Smokefoot (talk) 00:29, 27 October 2015 (UTC)
These articles aren't class assignments. They're part of a project, with Dario Taraborelli at Wikimedia, to bring more advanced scientific content to Wikipedia. There were extensive discussions with Dario before the creation of these articles. -- User: Carolineneil.
- But many of these are already covered, and many are much too specific and specialized for a general encyclopedia. Quite a few have been already deleted and more will be. Carolineneil (talk · contribs), Perhaps you could ask Dario to get in touch with us here. DGG ( talk ) 02:40, 2 December 2015 (UTC)
@DGG: thanks for the ping. While we provided advice in a volunteer capacity to User:Carolineneil and the team working on this project to help them comply with Wikipedia's norms and policies, WMF is not involved in this effort. I am personally very interested in the expansion of content of scientific relevance to Wikipedia and expert engagement initiatives and I'd be happy to relay this to people who have been active in this area (cc User:Rockpocket, User:Daniel Mietchen, User:Egonw) but I am afraid I cannot provide editorial advice about specific contributions (let alone resolve disputes) in a Wikimedia staff capacity.--Dario (WMF) (talk) 05:59, 2 December 2015 (UTC)
- I had a brief look at all the remaining articles started by User:Carolineneil and while this is not my field, got the impression that they were well-informed about the subject and well-intended but in need of guidance as to how we do things here in terms of jargon, linking, referencing, naming, formatting and so on. Next time, please try to improve some existing content first, observe how that works and then scale up, rather than starting a slurry of new articles that simply do not fit here in their current form. -- Daniel Mietchen (talk) 10:27, 2 December 2015 (UTC)
- These articles, chemistry-wise, are poorly crafted - the topics either are poorly defined or overly specialized. If these articles were not homework or other forced labor project, they read like it. If the author were interested in enhancing Wikipedia vs getting credit for creating articles, they would have folded most of the content into existing articles in a more compact way. Bloat IMHO. --Smokefoot (talk) 13:14, 2 December 2015 (UTC)
- I had a brief look at all the remaining articles started by User:Carolineneil and while this is not my field, got the impression that they were well-informed about the subject and well-intended but in need of guidance as to how we do things here in terms of jargon, linking, referencing, naming, formatting and so on. Next time, please try to improve some existing content first, observe how that works and then scale up, rather than starting a slurry of new articles that simply do not fit here in their current form. -- Daniel Mietchen (talk) 10:27, 2 December 2015 (UTC)
Osmosis
I recently revised osmotic pressure. It became apparent that the subject of osmosis is badly fragmented.
- osmosis
- forward osmosis
- osmotic concentration
- osmotic pressure
- osmotic coefficient
- osmotic shock
- Osmoregulation
- osmole
- Semipermeable membrane (needs work)
to cite some of the relevant articles. These should surely be merged into one article? Reverse osmosis probably merits the separate article.
Is there an opportunity here for a collaborative project - chemistry and biology - leading to a unified treatment of osmosis? Petergans (talk) 08:46, 30 October 2015 (UTC)
IUPAC gold book definitions and dihedral angles
Hi I am no chemist but did edit the dihedral angle page, In it i did add the UIPAC goldbook definition. (but i did much more to the geometry part of it.
I would like a chemist to have a look at it.:
also on https://en.wikipedia.org/wiki/Wikipedia_talk:WikiProject_Chemistry/Gold_Book_workgroup#A_bit_confused_though.2C_can_I_cite_from_Gold_Book.3F (sorry I don't know hoe to link there in proper wikipedia fashion ) I was wondering can we cite from the gold book or not? WillemienH (talk) 23:04, 2 November 2015 (UTC)
- The gold book definition is uneccesarily complicated and too specific to chemistry. A dihedral angle is simply the angle between two intersecting planes. To avoid any ambiguity, it is the angle between any pair of lines, one in each plane, that that have a common point on, and are normal (perpendicular) to, the line of intersection of the planes. For a non-chemical use, there may be a dihedral angle between the two wings of an aircraft. Petergans (talk) 08:06, 3 November 2015 (UTC)
- I don't know where that comes from. The IUPAC Gold Book gives the definition of a dihedral angle as follows
- The angle between two intersecting planes on a third plane normal to the intersection of the two planes.
Thanks for your edit, I am still wondering on 2 points: The previous definition was from torsion angle goldbook torsion angle, torsion angle is a redirect to dihedral angle I am thinking to put it back in but then n a new sub section. I was wondering about the UIPAC torsion angle definition and the description on the page, they do differ. (on clockwise and counterclockwise)
An other point the "Atan2" formula's in the article, they seem strange in atan2 seems to be a function of 2 vectors not of 2 numbers. while the atan2 page says atan2 is a function of 2 numbers. the formula seems to be comming from a book on computational chemistry can you check them out? Maybe best to continiue this discussion on talk:dihedral angle WillemienH (talk) 08:33, 4 November 2015 (UTC)
- A torsion (aka twisting) vibration is a term used in vibrational spectroscopy to signify a vibration in which the angle between two planes changes. For example, in ethane, rotation of one CH3 group with respect to the other, about the C-C bond is part of a torsion vibration. In this context torsion angle and dihedral angle are synonymous. In principle left and right torsions could be distinguished from each other in a chiral molecule, but I don't know if it has ever been done. Petergans (talk) 09:45, 4 November 2015 (UTC)
I have completely revised the article. I have eliminated the section referring to atan2 (comment of WillemienH, above) because I am unable to find a source for it, nor could I see the relevance of a 3-vector formula to an angle that can be defined in terms of the dot product of 2 vectors, as illustrated by the formula :. Also I have cleaned up the extensive duplication that was present before. Petergans (talk) 09:36, 9 November 2015 (UTC)
Can someone with good chemistry knowledge please evaluate a request posted on behalf of Ariel Fernandez -- here? There's a long history of COI here. We don't want an article that is unduly negative, but I'd like to make sure that the proposed changes are not flawed in terms of the claims we'd add. Is the idea of a dehydron coherent? That article (not the redirect) was originally created by Arifer (talk · contribs) (see [1])... Nomoskedasticity (talk) 16:47, 8 November 2015 (UTC)
- The idea of the dehydron is coherent. There is software that can calculate them in a reproducible way. Whether or not the dehydron is a legit concept is not why Fernandez's work is being called into question. Rather, it is due to claims made regarding purported relationships between dehydrons and molecular evolution, for which the reproducibility of the data has been challenged significantly enough to result in three expressions of concern and a fourth article placed on publication hold for around a year now. Molevol1234 (talk) 18:05, 16 November 2015 (UTC)
Seeking input on a student handbook for chemistry
Hello all! The Wiki Education Foundation is preparing to print a handbook to help student editors working with chemistry articles. You can find the draft text here. I'd really appreciate input from other editors on the Talk page. This WikiProject has already been an invaluable resource in creating this handbook, so thanks! Any feedback is especially helpful prior to Nov. 13 (this Friday). Thanks again! Eryk (Wiki Ed) (talk) 18:17, 9 November 2015 (UTC)
- The obsession for editors Wiki-chem is content yet the goal for Wiki Ed is some sort of experience involving learning markup language and communication skills, both admirable. It seems as though Wiki-Ed views student contributions as a recruiting tool, which is debatable. In fact it might be an unrecruiting tool because the students often get beaten up and the instructors pissed off. There are two central problems:
- Central problem #1: Wiki-chemistry is now so sophisticated that, with rare exceptions, contributing is beyond any reasonable undergraduate's ability, in the absence of active involvement of a professor. So what we get instead is a lot of safety cruft or hyper-esoterica such as articles on compounds selected seemingly at random from a chem catalogue.
- Central problem #2: It is difficult (almost impossible) for students to contribute unless the instructor is an experienced Wikipedian.
- Isn't there another venue such as Wikitextbooks that could be used to engage these young editors? --Smokefoot (talk) 18:53, 9 November 2015 (UTC)
- Hi Smokefoot! Thanks for your comments. For Wiki Ed, quality content trumps editor recruitment. That's why I think the goals of WikiProject Chem and student assignments are pretty aligned: we want people to contribute good, well-resourced content to Wikipedia (in this case, concerning chemistry). Of course, the majority of this community, like most communities on Wikipedia, has been extremely helpful to new editors, regardless of whether they edit from a classroom or from a living room. As you mentioned, we're working to create a resource that helps students contribute content with the guidance of experienced Wikipedians and instructors. We've specifically highlighted to avoid the "safety cruft," among other advice found in WP:MOSCHEM and other resources. Other guidance you'd have for editors who are students would be well appreciated on the Talk page of the handbook itself, so we can make sure the content of the handbook reflects the concerns of the Chemistry community on Wikipedia. Thanks again for the feedback! Eryk (Wiki Ed) (talk) 21:39, 9 November 2015 (UTC)
- There perhaps should be some information on notability — how to determine whether Wikipedia should have an article written on the topic. Sometimes students write about a very narrow topic, that could be a title for a journal article, but could be much broader in an encyclopedia. Also we sometimes get useless stubs that say almost nothing about a substance. So there should be some review about what subjects are suitable for the first time writers. For student assignments I find that plagiarism is a serious problem, even though there is warning against it. Perhaps there should be negative marks if it is detected. Another problem I have seen recently is registering user accounts for groups, rather than individuals. Make sure that each individual has their own user on Wikipedia. (not a Chemistry problem however.) Graeme Bartlett (talk) 00:58, 10 November 2015 (UTC)
- Thanks for the feedback, Graeme Bartlett! We expect plagiarism enforcement from instructors, and we're actively searching for plagiarism. It's a top priority for us - if you see plagiarism, please notify us! We do address plagiarism on the first page, and in the complementary online training and student editing brochure, but I'll see about reiterating it elsewhere in the handbook as I agree it is critically important. And we agree that registering group accounts doesn't work. That's why Wiki Ed requires each student to have a distinct username. You shouldn't find group accounts coming from the instructors we work with. The last point, on notability, is also a concern that's addressed outside the specific chemistry handbook. We work with the instructor to steer students away from overly narrow topics and find articles that are backed by significant literature. Thanks again for the feedback! Eryk (Wiki Ed) (talk) 20:39, 10 November 2015 (UTC)
- @Eryk (Wiki Ed): Recently I came across an Extinction course which has added plagiarism. I suspect this is not assisted by Wiki Ed. I made a list of users at User:Graeme Bartlett/ext2015 but have not yet checked much of the contributions. (Nothing to do with Chemistry). Graeme Bartlett (talk) 21:44, 10 November 2015 (UTC)
- Graeme Bartlett, thanks for flagging that. You're correct that it isn't a Wiki Ed-supported course, but we'll be reaching out to this instructor. Eryk (Wiki Ed) (talk) 00:23, 11 November 2015 (UTC)
- @Eryk (Wiki Ed): Recently I came across an Extinction course which has added plagiarism. I suspect this is not assisted by Wiki Ed. I made a list of users at User:Graeme Bartlett/ext2015 but have not yet checked much of the contributions. (Nothing to do with Chemistry). Graeme Bartlett (talk) 21:44, 10 November 2015 (UTC)
- Thanks for the feedback, Graeme Bartlett! We expect plagiarism enforcement from instructors, and we're actively searching for plagiarism. It's a top priority for us - if you see plagiarism, please notify us! We do address plagiarism on the first page, and in the complementary online training and student editing brochure, but I'll see about reiterating it elsewhere in the handbook as I agree it is critically important. And we agree that registering group accounts doesn't work. That's why Wiki Ed requires each student to have a distinct username. You shouldn't find group accounts coming from the instructors we work with. The last point, on notability, is also a concern that's addressed outside the specific chemistry handbook. We work with the instructor to steer students away from overly narrow topics and find articles that are backed by significant literature. Thanks again for the feedback! Eryk (Wiki Ed) (talk) 20:39, 10 November 2015 (UTC)
- Other places to help out could be Wikiversity, Wikisource or Simple English Wikipedia. These will also have Chemistry work that needs to be done, but will have their own rules. Graeme Bartlett (talk) 01:09, 10 November 2015 (UTC)
- Eryk (Wiki Ed) , and Graeme, I can see one clear avenue of approach: notable people not yet covered. According to WP:PROF, anyone holding a named chair in a major university is notable. We probably have many of the present holders of these positions, but the earlier ones remain notable. Every president of the American Chemical Society is notable; we are missing about 50% of the ones after 2007. Every present and past member of the chemistry section of the National Academy of Sciences is notable--we probably have most of the present, but not the past. Every editor in chief of a major journal is notable, present and past. We lack most of them. Such articles are quite straightforward to write. And the students will learn a little chemistry as the work on the articles. DGG ( talk ) 01:46, 2 December 2015 (UTC)
- There perhaps should be some information on notability — how to determine whether Wikipedia should have an article written on the topic. Sometimes students write about a very narrow topic, that could be a title for a journal article, but could be much broader in an encyclopedia. Also we sometimes get useless stubs that say almost nothing about a substance. So there should be some review about what subjects are suitable for the first time writers. For student assignments I find that plagiarism is a serious problem, even though there is warning against it. Perhaps there should be negative marks if it is detected. Another problem I have seen recently is registering user accounts for groups, rather than individuals. Make sure that each individual has their own user on Wikipedia. (not a Chemistry problem however.) Graeme Bartlett (talk) 00:58, 10 November 2015 (UTC)
Ionosilica
Is this already covered and/or is it notable - Ionosilica Thanks, JMHamo (talk) 23:57, 12 November 2015 (UTC)
- @JMHamo: @Peter Hesemann:. Peter Hesemann, the name of the editor who created this article, is also the name of an active researcher on modified silicas. This article appears to be self-promotion of a pretty specialized topic, although one can be sure that Hesemann is convinced that it is very, very important. Why people like this cannot contribute anything more general that does not benefit themselves reveals about how many scientists interacts with Wikipedia, which is seen as a venue for publicity. Aside from WP:COI, a specific concern is the usual one: WP:SECONDARY - it would be nice if the article cited a review. http://www.cmos.icgm.fr/phesemann. Oh well, life goes on. --Smokefoot (talk) 01:46, 13 November 2015 (UTC)
- The review is also written by our author. I have done a bit of peacock culling, and fixed refs. Graeme Bartlett (talk) 09:04, 13 November 2015 (UTC)
- @JMHamo: @Peter Hesemann:. Peter Hesemann, the name of the editor who created this article, is also the name of an active researcher on modified silicas. This article appears to be self-promotion of a pretty specialized topic, although one can be sure that Hesemann is convinced that it is very, very important. Why people like this cannot contribute anything more general that does not benefit themselves reveals about how many scientists interacts with Wikipedia, which is seen as a venue for publicity. Aside from WP:COI, a specific concern is the usual one: WP:SECONDARY - it would be nice if the article cited a review. http://www.cmos.icgm.fr/phesemann. Oh well, life goes on. --Smokefoot (talk) 01:46, 13 November 2015 (UTC)
TfD of interest
A merger has been proposed for three templates that format external links to pubchem in slightly different ways, but the discussion has seen very little participation. Please comment here if you're interested. Thanks! Opabinia regalis (talk) 01:52, 19 November 2015 (UTC)
Slow Science
Slow Science has been requested to be renamed, see talk:Slow Science -- 70.51.44.60 (talk) 04:37, 28 November 2015 (UTC)
Copyright question: Lists of isomers of Alkanes
(originally a help-me request at my user talk page)
Short question: Are the sources used by the Lists of isomers of (n)-alkanes articles copyrighted, or do the lists of isomers fall under fair use?
Background
This question pertains to the following 6 list articles:
- List of isomers of nonane
- List of isomers of decane
- List of isomers of undecane
- List of isomers of dodecane
- List of isomers of tridecane
- List of isomers of tetradecane
Prior to 28 November 2015, only the first 4 articles existed (-nonane — -docane). I created the -tridecane and -tetradecane articles on 28 Nov, by copying the data from http://www.scribd.com/doc/56047899/Constitutional-Isomers-of-Alkanes#scribd. I neglected to cite my sources when I committed, and @User:Seagull123 helpfully asked me to do so. (see also initial discussion of the issue).
I am only able to find 3 such lists of isomers of alkanes up to tetradecane on the internet, and they all seem to be exact copies of each other:
- http://www.scribd.com/doc/56047899/Constitutional-Isomers-of-Alkanes#scribd (the one I cited in -tridecane and -tetradecane)
- http://www.chemicalland21.com/info/Constitutional%20Isomers%20Of%20Alkanes.htm
- http://www.kentchemistry.com/links/organic/isomersofalkanes.htm (the one cited in -nonane)
I have reversed my thinking from the original discussion on @User:Seagull123's talk page. I believe the lists of isomers is non-copyrightable, much as a list of States in the United States is simply statement of fact. The sequence of the number of isomers for the first n alkanes is, in more general terms, the number of n-node unrooted quartic trees (graph theory)<ref name= OEIS >On-Line Encyclopedia of Integer Sequences (sequence A000602 in the OEIS)</ref>. The naming of the isomers is formulaic, following IUPAC rules.
While Wikipedia:Copyright in lists is an essay, not policy, I think it and its talk page are very instructive in the conversation.
sbb (talk) 13:18, 30 November 2015 (UTC)
- No I think there will be no copyright in the list of names. It will be public domain. Fair use is not required, and if it was, then it probably would not be justified. The way it is presented with the subheadings, could add some creative content though. The scribd list is poorly presented, and normally I would not consider this a reliable source either.
- Agreed, the Scribd list is poor, and it's not what I'd call a decent source citation. I used it because it had the most permissive copyright/license. But, as you noted, if it's something that cannot be copyrighted, no claim of copyright or licensing will restrict that. I'm moving forward with the assumption that any list of isomers of alkanes cannot be copyrighted.
- I checked some of the combinations, and they looked OK. But do you know if there are no duplicates (same shape with two different names) in the list?
- No, I don't know there are no duplicates, but the length of the list matches the known number(s) of isomers for a particular n-alkane.
- Is there a computer program around that will generate all the isomers? (http://hrcak.srce.hr/file/151228 http://pubs.acs.org/doi/abs/10.1021/ed048p675) Also chirality does not seem to make any appearance in the list. At which point is the isomer impossible due to being overcrowded? Graeme Bartlett (talk) 21:28, 30 November 2015 (UTC)
- The number of known isomers for a particular alkane ignores stereoisomers, and does not take into account unstable, overcrowded, or otherwise unrealistic actual isomer molecules. The number is identical to the number of n-node unrooted quartic trees (see reference to OEIS above).
- http://www.pmf.kg.ac.rs/KJS/volumes/kjs25/kjs25lukovits73.pdf does the job for tetradecane but does not give the names, and it also refers to a dodecane list in another paper. It says this was worked out in the 1930s. Graeme Bartlett (talk) 21:43, 30 November 2015 (UTC)
Inaccurate chemical structures in chemboxes
After a deletion request on Commons initiated by Edgar181 I had a look at this user's uploads. It turned out that many of the structures are incorrect (e.g. covalent bonds in salts) or at least of inferior quality. Hence, I nominated a few dozens of these files for deletion (see c:Commons:WikiProject Chemistry/Deletion requests). I noticed that also some articles in en.wikipedia contain structures drawn by this user:
- Sodium myristate (page redirected) chop File:3D_Sodium_myristate.png ionic as covalendt ; chop File:Sodium_myristate.png wrong charge distribution
- Sodium polonide (removed) ratio of sizes is way off
- Sodium nitride chop, misleading, needs 3D diagram instead
- Ammonium hexafluoroaluminate not so bad - shows numbers of atoms and what connects where
- Sodium polysulfide (removed) chop ionic bond shown as line and strange colour scheme
- Sodium aluminium hydride chop - misleading alumohydrate
- Sodium aluminosilicate chop - misleading, better to have a sample structure of one of the substances, or an image
- Sodium uranate chop - bad charge location on uranate and shows no water
- Sodium peroxynitrate (removed) chop - charge location probably wrong
- Sodium orthovanadate chop - charge location probably wrong
- Sodium thioantimoniate chop - charge location probably wrong
- Sodium tetrathionate temporarily keep (but redraw) - charge location wrong
- Sodium laurate temporarily keep (but redraw) - charge location wrong
- Potassium nonahydridorhenate keep - K atoms in wrong place, but correct numbers
- Sodium bismuthate chop - charge location wrong
- Ammonium arsenate chop - charge location wrong
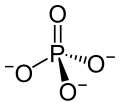
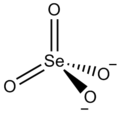
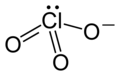
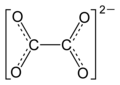
Could an inorganic chemist please check whether or not they may be left in the articles? --Leyo 23:38, 17 December 2015 (UTC)
- Well it depends on what the diagram is representing. If it is just the numbers and kinds of atoms, then these diagrams would show that OK. But they do not show that the substance would only exist in a solid crystalline state, with atoms in a different position from what is shown. Perhaps we can temporarily accept those with atoms in wrong positions. But how about we remove those that show a line or tube where the bond is clearly ionic. I also note that some diagrams on pubchem and chem spider are similarly low quality. Graeme Bartlett (talk) 00:04, 18 December 2015 (UTC)
- I have added opinions in italics Graeme Bartlett (talk) 00:30, 18 December 2015 (UTC)
- Thank you Leyo for identifying these issues. I have been working intermittently on this area, depending on my inclination and time. The general agreement achieved some time ago was that for polyions, we would show a Chemdraw like image. If we showed the hard-core Xray structure, the nature of these entities, which exist in solutions, would be obscured. That idea is portrayed in Potassium nonahydridorhenate, which shows both kinds of structures. Some of these things like Sodium nitride are silly, and these are the ones we really need to replace because they are rock-like 3-d dense phases. Some images, like Sodium laurate are ugly but net better than nothing. Some of these species are not well defined, like Sodium thioantimoniate, and are mixtures of historic interest with approximate formulas. In some cases, the German Wikipedia has some structures, like https://de.wikipedia.org/wiki/Natriumnitrid (anti-ReO3 motif). --Smokefoot (talk) 01:13, 18 December 2015 (UTC)
- Why is it (sodium nitride) silly? (unless the structure is wrong). I think its miseleading to represent a ion compound with the structure of the ions (like sodium uranate). Its like showing a forest with a single tree. Christian75 (talk) 07:39, 18 December 2015 (UTC)
- I don't think it is necessarily misleading to represent an ionic compound with the structure of individual ions. To use your analogy, sometimes there isn't a forest. In solution (a highly relevant situation, where a majority of chemical reactions occur) you have separate, individual trees rather than the forest. Different representations can serve different purposes. I agree with Smokefoot's assessment above that potassium nonahydridorhenate is ideal: one image that details the connectivity of atoms within the ions and another image that shows the larger arrangement of those ions in a crystal structure. -- Ed (Edgar181) 12:40, 18 December 2015 (UTC)
- I agree. The example (potassium nonahydridorhenate) is very good. And I prefer that too. Often we have a picture of the solid compound and a structure of a single "unit", like Sodium nitrite. But I do not suggest it should be removed, its better than nothing and at least it show the two ions. (btw. I think the sigle trees analog represent gas phase rather) Christian75 (talk) 16:39, 18 December 2015 (UTC)
- Why is it (sodium nitride) silly? (unless the structure is wrong). I think its miseleading to represent a ion compound with the structure of the ions (like sodium uranate). Its like showing a forest with a single tree. Christian75 (talk) 07:39, 18 December 2015 (UTC)
- Thank you Leyo for identifying these issues. I have been working intermittently on this area, depending on my inclination and time. The general agreement achieved some time ago was that for polyions, we would show a Chemdraw like image. If we showed the hard-core Xray structure, the nature of these entities, which exist in solutions, would be obscured. That idea is portrayed in Potassium nonahydridorhenate, which shows both kinds of structures. Some of these things like Sodium nitride are silly, and these are the ones we really need to replace because they are rock-like 3-d dense phases. Some images, like Sodium laurate are ugly but net better than nothing. Some of these species are not well defined, like Sodium thioantimoniate, and are mixtures of historic interest with approximate formulas. In some cases, the German Wikipedia has some structures, like https://de.wikipedia.org/wiki/Natriumnitrid (anti-ReO3 motif). --Smokefoot (talk) 01:13, 18 December 2015 (UTC)
- I have added opinions in italics Graeme Bartlett (talk) 00:30, 18 December 2015 (UTC)
- Leyo, thanks for the notification. Some of the images have new images that aren't much better - for example, replacing a trigonal planar covalently-bonded Na3N "molecule" with a trigonal planar (?) arrangement of three sodium cations around a nitride anion is not a great improvement in terms of representing the structure. I agree with Smokefoot that the de-wiki image is far more appropriate / representative. Some of the new ones are also wrong, like File:Ammonium hexafluorogallate.png with six oxidation-state 0 fluoride atoms around a gallide anion and three ammonium cations or File:3D Sodium maleate.png with carboxylate groups without resonance involvement and each sodium cation associated with a sole oxygen atom. EdChem (talk) 02:44, 18 December 2015 (UTC)
- @Claudio Pistilli: as the uploader of these images, would you like to comment? EdChem (talk) 02:44, 18 December 2015 (UTC)
- I removed a couple more of the images which are clearly incorrect - showing covalent bonds in place of ionic bonds. -- Ed (Edgar181) 12:50, 18 December 2015 (UTC)
- Images were added to the sodium polysulfide article. If anyone wants to help with the layout/format, please lend a hand.--Smokefoot (talk) 13:39, 18 December 2015 (UTC)
- Ammonium hexafluoroaluminate has an image with the same problems as I mentioned above - in this case an octahedral AlF63− ion with an Al3− core surrounded by six oxidation state 0 fluorine atoms. EdChem (talk) 15:05, 18 December 2015 (UTC)
Thank you for all your reviews and comments. @Graeme Bartlett, Smokefoot, Christian75, and EdChem: I would appreciate if you could also comment the related deletion requests. --Leyo 00:29, 19 December 2015 (UTC)
- Leyo: I support all of your deletion recommendations. BTW, is there a list of chemical articles lacking images? --Smokefoot (talk) 01:15, 19 December 2015 (UTC)
- Category:Chemistry pages needing pictures is the main one, which is based on manual tagging. There are subcats for various purposes, including one automatically generated by chembox. — Preceding unsigned comment added by DMacks (talk • contribs)
- Smokefoot, Graeme Bartlett posted several keep votes. Hence, there is a high probability that these will be kept in the end, if there are no additional delete votes. --Leyo 10:48, 19 December 2015 (UTC)
- See also: Category:Chembox_articles_without_image. It's far more extensive; being generated automatically. The majority of compounds listed appear to be inorganic --Project Osprey (talk) 23:02, 19 December 2015 (UTC)
- I started to recommend deletion but ran out of steam. I guess if Wiki Commons doesnt care about clutter on their servers, our job is just to keep wrong or misleading images out of the articles. One of the persistent problems are "molecular graphics" images of ionic compounds, which implies knowledge and reliability but usually results from the opposite, despite good intentions.--Smokefoot (talk) 13:55, 19 December 2015 (UTC)
- I posted a lot of comments, as the images were replaced as a result of this effort. Then the original rationale for deletion was not valid. For a very few I actually voted keep if they were good enough to use. But many have ugly colours or misleading charge distribution. The bad images should be tagged warning people not to use them. I prefer to keep ones that were used in the past articles, as we can see how they have improved. But no more use should be made of bad unreplaced images. Graeme Bartlett (talk) 21:02, 19 December 2015 (UTC)
- The safest method to make sure that inappropriate images are not used is to delete them. The best alternative would be to rename them (e.g. “File:Incorrect depiction of xyz”), but I prefer the former. --Leyo 21:28, 20 December 2015 (UTC)
- The two users who contributed most of the ball-and-stick and calotte models are Benjah-bmm27 and Jynto. I would like to hear their opinions on the examples above or such uploads in general, too. --Leyo 22:33, 22 December 2015 (UTC)
- Honestly, I cringe a bit when I see this user's files. They have the look of being cobbled together, like some atomic-scale Frankenstein. The mismatched styles, blurring, aliasing, inconsistent labelling of atoms, incorrect bond angles, bonds/atoms drawn in Paint, inexact scaling, inconsistent colouring, and the visible joins between pasted sections, all belie a certain... sloppiness to this process.
- I'm not surprised that they are rife with factual errors, but I won't repeat what has already been said about the sodium bonding. I will however add that it is very difficult to get bond angles and lengths right when making molecules this way. Even in simple organics, the shape is determined by an energy-minimisation process that should involve the entire molecule. And while this is never perfect, it is vastly preferable to the guesswork shown here.
- I don't wish to be too harsh on Claudio here; I myself started out making molecules in this way. I'm sure Ben Mills, if he was even aware of me, may have looked upon my early attempts in much the same way as I do when I see my molecule photoshopped into a... whatever this thing is. I don't think I made as many mistakes as Claudio, but I did tend to stay away from inorganics like these with primarily ionic bonding, as there is no 'right' way to depict them.
- - Jynto (talk) 13:04, 23 December 2015 (UTC)
- Ben Mills is very selective. Some or several of the eye-candy ball-and-stick organic images seem either (i) to imply knowledge of conformation that the author lacks or (ii) don't provide insight beyond ChemDraw. --Smokefoot (talk) 14:12, 23 December 2015 (UTC)
- I posted a lot of comments, as the images were replaced as a result of this effort. Then the original rationale for deletion was not valid. For a very few I actually voted keep if they were good enough to use. But many have ugly colours or misleading charge distribution. The bad images should be tagged warning people not to use them. I prefer to keep ones that were used in the past articles, as we can see how they have improved. But no more use should be made of bad unreplaced images. Graeme Bartlett (talk) 21:02, 19 December 2015 (UTC)
- I started to recommend deletion but ran out of steam. I guess if Wiki Commons doesnt care about clutter on their servers, our job is just to keep wrong or misleading images out of the articles. One of the persistent problems are "molecular graphics" images of ionic compounds, which implies knowledge and reliability but usually results from the opposite, despite good intentions.--Smokefoot (talk) 13:55, 19 December 2015 (UTC)
I don't watch this page regularly, but here is a belated comment on this topic. The representation of the structure of a salt in terms of cation and anion structures is wrong in principle. In fact I recently removed such diagrams from the chemboxes in monosodium citrate, disodium citrate and trisodium citrate because they were misleading as to physical and chemical structure, let alone crystal structure. On the other hand, a diagram representing a structure might well be useful in some cases; it should then be clearly labelled so that the reader can see that the structure refers to a cation or anion as case may be.
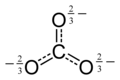
The structure is Ammonium arsenate is particularly horrible. Not only because the anion is tatrahedral but also because the picture shows only one canonical form of the four that contribute to a resonance hybrid. Petergans (talk) 17:23, 23 December 2015 (UTC)
- Although these views deserve respect, we should reinstate the images in monosodium citrate and some others, because we have discussed this policy intermittently for years. The consensus has been that polyatomic anions and cations are usefully shown with simple ChemDraw like images. Yes, such images are imperfect. The alternative, some sort of crystal structure, is usually information overload. Readers would have a difficult time figuring out the basic skeletal arrangement of the polyion from such depictions. Having said all that, I too sometimes self-righteously remind the unenlightened and unwashed that such images are gross simplifications, but we are dealing with imperfect is better than perfect in this case.--Smokefoot (talk) 18:53, 23 December 2015 (UTC)
- I fundamentally disagree. I've never seen a "structure" showing a cation and and anion together in arbitrary conjunction in any chemistry book. The OK thing to do is to show the structure of each polyatomic entity by itself. Petergans (talk) 22:22, 23 December 2015 (UTC)
- I sympathize. Naked cations are especially problematic. But we cant just show the anion in the chembox of an article about a salt. That would be misrepresent the formula. Maybe the compromise is RCO2-Na+ written like that. Maybe other people have ideas. But my thinking is that educated chemists read pictures and spectra etc with a more discerning eye than do the person who just wants some idea of what is in the bottle of sodium hydrogen citrate. --Smokefoot (talk) 22:52, 23 December 2015 (UTC)
- I fundamentally disagree. I've never seen a "structure" showing a cation and and anion together in arbitrary conjunction in any chemistry book. The OK thing to do is to show the structure of each polyatomic entity by itself. Petergans (talk) 22:22, 23 December 2015 (UTC)
Sodium uranate is the same compound how Sodium diuranate. Can someboby make only one article from both. Thanks, --Alchemist-hp (talk) 14:59, 8 January 2016 (UTC)
Image requests for the glyphosate article

I have two requests relating to the chemistry section of the glyphosate article. The first relates to the File:Glyphosate synthesis from dimethyl phosphite.svg which is incorrect in that the initial reactant is paraformaldehyde rather than formaldehyde itself and in that the intermediate is a tertiary amine (third substituent CH2OH) and not secondary as shown. I request a new version that shows:
HO-(-CH2-O-)n-H + H2N-CH2-COOH ----NEt3 / CH3OH ----> (HOCH2)2NCH2-COOH ---- (CH3O)2P(=O)H (above arrow) NEt3 / CH3OH (below arrow) ----> (CH3O)2P(=O)-CH2-N(CH2OH)-CH2-COOH ---- HCl / Δ (above arrow) H2O (below arrow) ----> (HO)2P(=O)-CH2-NH-CH2-COOH
I hope this is clear. Maybe lay out as A → B ↓ C (below B) ← D (below A), in an on-its-side 'U' shape? The other diagram is to illustrate the different ionic states of glyphosate and the pH ranges in which they predominate. The four pKa values are 0.8 and 6.0 (phosphonates), 2.3 (carboxylate), and 11.0 (amine). Basically, I'm thinking a column of the five structures for increasing pH, with these contents:
- (HO)2P(=O)-CH2-N(+}H2-CH2-COOH pH < 0.8
- ((−)O)(HO)P(=O)-CH2-N(+}H2-CH2-COOH 0.8 < pH < 2.3
- ((−)O)(HO)P(=O)-CH2-N(+}H2-CH2-CO2(−) 2.3 < pH < 6.0
- ((−)O)2P(=O)-CH2-N(+}H2-CH2-CO2(−) 6.0 < pH < 11.0
- ((−)O)2P(=O)-CH2-NH-CH2-CO2(−) pH > 11.0
Many Thanks. EdChem (talk) 02:30, 21 December 2015 (UTC)
- For the protonation states, File:Glyphosate Dissociation V.1.svg has slightly different values (transitions at 2, 2.6, 5.6, 10.6). De:glyphosat has a cite that supports that image, [2], an INCHEM report that cites the values to the Sprankle,etal (1975) ref in our article. What is the basis for your values? That file would be easy to convert to an English-language variant. Or else if there are better values, that too could be updated. DMacks (talk) 19:17, 21 December 2015 (UTC)
- @DMacks: Thanks for your response. The pKa data comes from the Virtual Museum of Minerals and Molecules page of glyphosate which is maintained by the University of Wisconsin. Details here. I haven't found its source for the values but the de values match the infobox except that <2 has become 2. I don't like putting equilibria signs between the forms in which protons are disappearing / appearing, but that image is certainly better than nothing. EdChem (talk) 07:53, 24 December 2015 (UTC)
- @DMacks: I'd appreciate if you can have a look at these diagrams when you get a chance. Thanks. EdChem (talk) 06:52, 31 December 2015 (UTC)
- I dislike putting substantial structural components "over the arrow" (as compared to solvents, catalysts, or other conditions)...makes it visually less clear where each atom comes from in the product. How about (with more consistent placement of solvent under arrows):
- HO-(-CH2-O-)n-H + H2N-CH2-COOH (HOCH2)2NCH2-COOH
- (CH3O)2P(=O)H + (HOCH2)2NCH2-COOH (CH3O)2P(=O)-CH2-N(CH2OH)-CH2-COOH (HO)2P(=O)-CH2-NH-CH2-COOH
- Bonus: demonstration of a quick way to make the arrows with text for discussing layout ideas:) Or less wide, with third reaction going "down":
(CH3O)2P(=O)H + (HOCH2)2NCH2-COOH (CH3O)2P(=O)-CH2-N(CH2OH)-CH2-COOH (HO)2P(=O)-CH2-NH-CH2-COOH
- But in this case, the three steps as "right, down, left" does seem like a sensible way since a key idea is that it's a one-pot method. DMacks (talk) 20:32, 21 December 2015 (UTC)
- DMacks, thanks for the illustration, I'll try to bear it in mind for discussions. I understand about the avoiding putting structures over arrows but in this case I think it would be clear. However, any clear presentation is fine with me. I don't like the current image using the wrong formaldehyde and a secondary amine, especially because the reaction outcome would be different. I have yet to add the text about it, but if you look at the imindiacetic acid approach it is apparent that just using glycine would be much easier, but this is not done as the extra N-group serves as a protecting group preventing further reaction. As you say, given a one-pot synthesis, a comprehensive but compact A - B - C - D cycle would be appropriate. Thanks. EdChem (talk) 07:53, 24 December 2015 (UTC)
- Here's a compact image for the one-pot route, also with a variant that is color-coded as to the reagent that supplies each atom (resolves my concern about tracing the structural components through multiple reactions):
- DMacks, thanks for the illustration, I'll try to bear it in mind for discussions. I understand about the avoiding putting structures over arrows but in this case I think it would be clear. However, any clear presentation is fine with me. I don't like the current image using the wrong formaldehyde and a secondary amine, especially because the reaction outcome would be different. I have yet to add the text about it, but if you look at the imindiacetic acid approach it is apparent that just using glycine would be much easier, but this is not done as the extra N-group serves as a protecting group preventing further reaction. As you say, given a one-pot synthesis, a comprehensive but compact A - B - C - D cycle would be appropriate. Thanks. EdChem (talk) 07:53, 24 December 2015 (UTC)
- But in this case, the three steps as "right, down, left" does seem like a sensible way since a key idea is that it's a one-pot method. DMacks (talk) 20:32, 21 December 2015 (UTC)
- DMacks (talk) 06:06, 3 January 2016 (UTC)
- First, Many thanks, DMacks. I can see the benefits of the colour coding, that I don't consider it essential. Having said that, I'm happy to use the coloured version. Second, sadly, a little problem... the last step is aqueous, not in NEt3 and methanol. If triethylamine were present, it would become an elaborate way to make HNEt3Cl. Would you please adjust the images? Everything else looks correct to me. Thanks. EdChem (talk) 06:32, 3 January 2016 (UTC)
- That detail seemed strange as I was adding it to the diagram, but it's in accord with [3], the cited ref in our article. "DMP is then added to the reaction mixture, forming the following phosphonate ester: [reaction diagram] Concentrated HCl is then added at room temperature... [reaction diagram]". That second rection diagram has "Et3N/MeOH" at the both the room-temp and heated stages where HCl is used. DMacks (talk) 14:07, 11 January 2016 (UTC)
- First, Many thanks, DMacks. I can see the benefits of the colour coding, that I don't consider it essential. Having said that, I'm happy to use the coloured version. Second, sadly, a little problem... the last step is aqueous, not in NEt3 and methanol. If triethylamine were present, it would become an elaborate way to make HNEt3Cl. Would you please adjust the images? Everything else looks correct to me. Thanks. EdChem (talk) 06:32, 3 January 2016 (UTC)
- DMacks (talk) 06:06, 3 January 2016 (UTC)
Fundamental error in chemboxes
Whenever an equilibrium constant value (pKa etc.) is cited it is essential to give the temperature at which the value was determined. It cannot be simply assumed that the value relates to 25°C, though many values do so. See acid dissociation constant#temperature dependence for details concenrning the thermodynamics. Where it is known, temperature vaiation (or standard enthalpy and and standard entropy) could be displayed.
Strictly speaking, the ionic strength should also be stated. If the data are availble, values extrapolated to zero ionic strength should be cited, as otherwise values depend also on the nature and strength of the ionic medium. Petergans (talk) 16:36, 23 December 2015 (UTC)
- So, when the given value is at 25 °C, there is no error at all. If not 25 °C, then that's called an omission. -DePiep (talk) 16:51, 23 December 2015 (UTC)
Structures of salts in chembox

A discussion above has revealed a deficiency in chembox . It relates to the chemical image in section 0. When the substance is a salt many articles show nonsense images as with sodium orthovanadate. This results from the way chembox is coded, which is suitable for molecules and crystal structures, but is not suited for salts containing polyatomic ions.
I suggest that chembox needs to be modified to accomodate structures relating to such salts. There should be separate parameter sets with titles "Cation" and "Anion". The result would be separate pictures of the ions, side by side. A parameter may be left empty when an ion is monatomic. For example sodium orthovanadate would only show the anion (heading orthovanadate ion?).
I tried using the parameter | ImageNameL1 = with citric acid, which does show 2 images side by side, but it did not do what was wanted.
I would normally make such modifications myself, but in this case it is not possible because chembox is not coded in HTML, a most unsatisfactory situation. Petergans (talk) 11:13, 25 December 2015 (UTC)
- Merry Christmas.
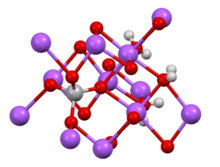
- --Smokefoot (talk) 14:08, 25 December 2015 (UTC)
- In sodium orthovanadate. So? -DePiep (talk) 11:25, 27 December 2015 (UTC)
- "the way chembox is coded, ... is not suited for salts containing polyatomic ions." How not? See also Template:Chembox/doc/images.
- {{Chembox}} does not connect to the content of an image: every image is treated the same in same situation. Maybe you could describe what you expect when using
|ImageNameL1=. Sure {{Chembox}} then expects|ImageNameR1=. -DePiep (talk) 17:36, 26 December 2015 (UTC)
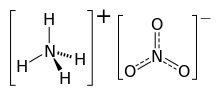
- My point is that when the substance is a salt it contains cations and anions as individual structural units; a crystal structure would show how these units are related in space.
- Ammonium nitrate (extract from chembox shown here) is closer to what is needed, but it is taken from a single svg file containg diagrams of both ions. What I am suggesting is that cation and anion structures should be in separate image files and be in separate "sections" in the chembox - in this case one showing the ammonium ion, caption "ammonium ion" and the other showing the nitrate ion, caption "nitrate ion". Monatomic ions like Na+ or Cl- would not be shown. For example NaNO3 would show only the nitrate ion structure with the chembox title "Sodium nitrate" and diagram caption "nitrate ion".
- Incidentally, it is a general principle that all diagrams should have labels/captions which describe their contents. Would a young student understand what this diagram, without a caption, represents? Petergans (talk) 10:05, 27 December 2015 (UTC)
- Hmm. Today are available
|ImageFileL1=,|ImageFileR1=
- Hmm. Today are available
- Incidentally, it is a general principle that all diagrams should have labels/captions which describe their contents. Would a young student understand what this diagram, without a caption, represents? Petergans (talk) 10:05, 27 December 2015 (UTC)
| |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
- For salts, an editor could split the image into two and then enter both. I guess some tweaking may be desired: no vertical bar between, option to have a single caption below both images, ....
- Your writing that "result would be separate pictures of the ions" suggests that the infobox itself should actively split the image into two. That is done rarely in any infobox (if ever), and quite complicated for the editor to enter (think of fine-pointing the position of the cutting line). And is your example of aluminium nitrate, here, wrong somehow? -DePiep (talk) 11:14, 27 December 2015 (UTC)
- (edit conflict) Sounds to me like you're suggesting something like whats been done for Sodium chlorite?.. Regarding captions, they can be entered using
|ImageCaption=which can of course be suffixed with L1, R1, L2, R2 as needed to mach the images in question. Can I ask what the justification is for having "Sodium nitrate" but omitting the Na+ counter-ion from its chembox image? Surely that would just lead to confusion? --Project Osprey (talk) 11:17, 27 December 2015 (UTC)- FWIW, I do not support showing separate images of the anions and cations. Although the interpretations of these ChemDraws require care/education (what doesn't?), my recommendation is for the following sequence stacked:
- Chemdraw of cation-comma-anion
- Below this would be a portion of the solid state structure of the most important polymorph or hydrate
- and then a picture of a sample (or two, side by side)
- The elaboration on the above could be accomplished with words and images in the test, including the caveat that simple ions rarely exist, pictures of other polymorph/hydrates, close-ups of coordination spheres of the anions and cations. --Smokefoot (talk) 14:50, 27 December 2015 (UTC)
- No comma drawn in the images please. If needed, it should either be generated by the chembox or a separate image of a comma should be used. In de-WP, images from c:Category:Ionic structures construction kit are used to create the desired salt in the chembox (see e.g. de:Silbercyanid or de:Calciumcitrat). --Leyo 23:15, 27 December 2015 (UTC)
- FWIW, I do not support showing separate images of the anions and cations. Although the interpretations of these ChemDraws require care/education (what doesn't?), my recommendation is for the following sequence stacked:
| |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Thank you editors Project Osprey and Leyo for your helpful comments on the basis of which I have made the diagram at the right. I have used existing image files which are not consistent with each other regarding ionic charge. Apart from that I think this would be a much clearer presentation to use when a crystal structure image file is not available. Petergans (talk) 11:56, 28 December 2015 (UTC)
- This can work OK for when there is a 1:1 ratio of anion to cation. But for other ratios it is not good, eg the sodium orthovanadate we started with. Graeme Bartlett (talk) 22:34, 28 December 2015 (UTC)
- In by now, I have not grasped why this presentation (split L+R) is better. The original complaints here by Petergans point into a totally different direction (a misunderstanding it now appears). -DePiep (talk) 07:19, 29 December 2015 (UTC)
- The point is this. I've never seen a diagram like the vanadate one above, in any chemistry book. With salts either the crystal structure or the strucure(s) of ion(s) are shown. In an substance made up of cations and anions, known as a salt, the ions are structurally independent of each other. Moreover differernt salts may have different crystal structures, e.g. NaCl and CsCl, depending on the sizes of the ions. Petergans (talk) 09:54, 29 December 2015 (UTC)
- I agree the split is not better, and to combine two .svg files into a bigger oen would only take 10 minutes. CHecking a chemistry textbook, complex ions can be shown in large square brackets with the charge to the top right. They can be adjacent to a simple ion. A simple diagram shows "Na+ Cl−". And this without colour and shadowing. Graeme Bartlett (talk) 10:28, 29 December 2015 (UTC)
Hello, people of WikiProject Companies. I've created this message to notify active members of the project, especially to those who know a lot about or are interested in PCBs and benzaldehyde. If you are, I need help improving my article about the Clyde cancer cluster, an incident where, because of Whirlpool Corporation dumping PCBs in their Whirlpool Park property, and for being responsible for there being benzaldehyde in people's attics in the town, and were sued for this. It is definitely not a bad article. It just needs some small improvements. I'm not at all saying minor edits or improvements are bad, but I'm specifically looking for people who can help me long-term with a lot of co-research to improve the article to reach Good Article status. If you're interested in helping, the things needed to be improved are listed on Talk:Clyde cancer cluster#Improvements. In other words, more material needs to be added to improve the article, and I want more people to edit because I feel like I'm the sole editor. Regards, Philmonte101 (talk)
- How, and why, did Whirlpool get the benzaldehyde into people's attics? There's no mention of attics in the article. Maproom (talk) 18:33, 7 January 2016 (UTC)
Structures of salts in chembox, part 2
(moved from above) Here is one draft idea which I think complements the crystal structure image that I uploaded and more than a line formula.

. Smokefoot (talk) 03:21, 29 December 2015 (UTC)
- This diagram is neither fish nor fowl. Only the anion has a structure to illustrate. However, I think I understand the motivation behind this suggestion. It has happened that as the chembox "Identifiers" section has grown the chemical formula and structures have become overly separated from each other. My proposal is to show only the structure of the anion (in such instances) and to move the chemical formula, Na3(VO4), from its position in the "Properties" section to be just over or under the IUPAC name, that is, directly below the structure diagrams. Alternatively put the IUPAC name and chemical formula above the structure diagrams. Petergans (talk) 10:43, 29 December 2015 (UTC)
- Well it is a big improvement over the colourful diagram. But one deficiency is that there are also two water molecules in the salt. Graeme Bartlett (talk) 10:59, 29 December 2015 (UTC)
- The image to the right looks like it contains a Na+3 ion. The German wiki uses 3 Na+ which I think is a better approach. If the image should help "newbies" - I suggest we add three Na+ instead of just one with a coefficient. Christian75 (talk) 12:35, 29 December 2015 (UTC)
- I can redo the (Na+)3 as 3Na+. About the water of hydration, I suggest omitting it since we are trying to convey what one gets when the salt is dissolved. --Smokefoot (talk) 18:57, 29 December 2015 (UTC)
- re Petergans: I (consider a half-laynman) understand the thought. Questions coming to my mind. IMO the crystal (3d, ball and stick) image could be added (at least in this example; also covers the water molecule thing). A bit strange that only one ion structure should be shown (if I read you correct). And I wonder if IUPAC does not have a rule or guideline to show two ions in one drawing without suggesting a structural position to each other.
- About the order in {{Chembox}}: putting structure images, IUPAC name and chem formula close to one another sounds really good. However, the way Chembox is build today this can not be achieved without editing all 10k instances. As it is, chem formula is usually in
|Section2=and the template can not move a data row to outside of this (Properties) block into another one (top, or Section1=Identifiers). Of course, the goal is to switch to Lua some time which could solve this and many other things. -DePiep (talk) 19:07, 29 December 2015 (UTC)
- I can redo the (Na+)3 as 3Na+. About the water of hydration, I suggest omitting it since we are trying to convey what one gets when the salt is dissolved. --Smokefoot (talk) 18:57, 29 December 2015 (UTC)


I'm interested to know how molecular models can accommodate this. I produce a lot of these myself, and I've never found a 'right' way to depict ionic species. Certainly, some of my older files will require re-doing. My current approach with these is to show the ions side by side, but not in any way that suggests they are connected. Sometimes, as in the case of olympiadane (an example which I am rather fond of), I surround the more complex ion with many smaller ones. Other times, as in the case of Brown HT, I've put the cations close to the charged parts of the anion where I thought it made sense (opposite charges do after all attract). But I suppose that could be construed as misleading if it is implying this is how they are arranged spatially.
The alternative would be to show the ions at opposite ends of the image (perhaps there could be an established rule like the cation's always on the left), or to simply show the ions in separate images (I've done that before though, and it seems... imprecise). I'd honestly rather sidestep the issue altogether and draw only the neutral molecule (or occasionally zwitterion) forms of most dyes, but when the chemical formula specifies sodium/potassium/etc, this course of action would feel inaccurate.
Either way, I shall be redoing most of my models depicting ions over the coming months, since I'd erroneously sized the alkali metal cations to their ionic radii rather than vdW. So this would be a well-timed opportunity to set things right. Hit me up for any advice on the matter.
— Jynto (talk) 14:15, 31 December 2015 (UTC)
- There is no need to include all ions in a graphic relating to a salt, unless it is a crystal structure. When a cation, or anion, is omitted, just ensure that the image caption states explicitly that the structure is that of the anion or cation, optionally adding (e.g.) "in the compound NaX". Petergans (talk) 14:56, 31 December 2015 (UTC)
Please merge
Can someone do the fusion of these 2 articles, Dichlorophen and Dichlorophene ? Without any action I will just replace the content of one article by a redirection to the other one. Thanks Snipre (talk) 20:47, 6 January 2016 (UTC)
AfC submission
See Draft:Radical fluorination. Thank you, FoCuS contribs; talk to me! 18:05, 8 January 2016 (UTC)
- I accepted the submission. Thanks for the notification. -- Ed (Edgar181) 19:30, 8 January 2016 (UTC)
Delocalized electronic structures

| |||
| |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
This concerns the graphics purporting to show the electronic structure of certain ions in the chembox and stand-alone. In the case of the sulfate ion (extracted from sulphate/chembox) there is an obvious discepancy between the valence bond structure, which looks unsymmetrical and the ball-and-stck and space-filling structures below it which look symmetrical. This arises because what is shown is one canonical form of many contributing to a resonance hybrid, as shown lower down in the sulphate article (File:Sulfate-resonance-2D.png). Though a trained chemist may understand the implicit reference to other canonical forms, the apparent contradiction will be confusing for the general reader.
The issue is widespread. Here are some of the simplest examples of unsymmetrical-looking structures. Others can be seen at c:Category:Ionic structures construction kit, pyrophosphate, pyrosulfate, etc., etc..
-
periodate (and perbromate?)
The double bonds in these single canonical forms are needed in order that all oxygen atoms conform to the octet rule.
On the other hand, there are many instances where symmetrical, delocalized, structures are shown. Here are some of them.
-
permanganate (and pertechnetate)

Some consistency is needed between WP articles. I suggest that the style of representation shown at the left for chromate be used in chemboxes, where appropriate, for all tetrahedral ions.
A similar style should be used for ions of other stereochemistry and in the article body where there is no chembox structure. Petergans (talk) 08:58, 11 January 2016 (UTC)
Formulas in the chem-boxes
Formula sample: Lead(II) phosphate and other inorganic compounds. What is more important: Pb3(PO4)2 or the info from the current box: O8P2Pb3 = "| Pb=3 | P=2 | O=8"? My opinion: Pb3(PO4)2. --Alchemist-hp (talk) 19:17, 11 January 2016 (UTC)
- For most inorganic compounds, the style like "Pb3(PO4)2" is preferred. For most organic compounds, Hill notation that is automatically generated from text such as "| Pb=3 | P=2 | O=8" would be preferred. I have changed the style at Lead(II) phosphate to the first style accordingly. -- Ed (Edgar181) 02:48, 12 January 2016 (UTC)
- OK, thanks, that's also my opinion! --Alchemist-hp (talk) 08:20, 12 January 2016 (UTC)
Could a native speaker please merge Gold#Chemistry and Gold#Chemistry 2? The user who noticed it is currently inactive. --Leyo 02:13, 16 January 2016 (UTC)
- I have combined the sections. I did not do any speaking, just cutting and pasting and deleting. Graeme Bartlett (talk) 08:46, 16 January 2016 (UTC)
- Thanks anyway. :-) --Leyo 02:14, 17 January 2016 (UTC)
separate pages for zwitterions
Is there a reason why we have pages on Glutamic acid and Glutamine? One is simply the zwitterion of the other. --Project Osprey (talk) 13:38, 18 January 2016 (UTC)
- Good find .. and both are 13 years old. I don't even think a history-merge makes any sense (people would get utterly flipping confused flipping between the two versions for 13 years and not be able to find who did what ..). Merge and then redirect the one that is less compliant with our standards to the other, archive the talkpage of the redirect to archives under the other. --Dirk Beetstra T C 13:44, 18 January 2016 (UTC)
- Ah! Nice catch. I thought I was missing something. --Project Osprey (talk) 14:01, 18 January 2016 (UTC)
Acid-base reaction
Would someone please check the report of a problem in a formula at Talk:Acid dissociation constant#Reaction equilibrium formula error. Johnuniq (talk) 21:55, 22 January 2016 (UTC)
RfC at Talk:Climate change denial
There is a RfC at Talk:Climate change denial. Please contribute if you are interested. Biscuittin (talk) 00:10, 28 January 2016 (UTC)


![{\displaystyle \varphi =\operatorname {atan2} \left(\left([\mathbf {b} _{1}\times \mathbf {b} _{2}]\times [\mathbf {b} _{2}\times \mathbf {b} _{3}]\right)\cdot {\frac {\mathbf {b} _{2}}{|\mathbf {b} _{2}|}},[\mathbf {b} _{1}\times \mathbf {b} _{2}]\cdot [\mathbf {b} _{2}\times \mathbf {b} _{3}]\right).}](https://wikimedia.org/api/rest_v1/media/math/render/svg/9c93796cb5d0aae71923af02220faf79f8d179b7)

![{\displaystyle {\xrightarrow[{CH_{3}OH}]{NEt_{3}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/7d5132accc6c81437a20feb3aebd9ec7879a259d)
![{\displaystyle {\xrightarrow[{H_{2}O}]{HCl,heat}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/dc6a32cbdd55bd37ce785df239e80cc575924d27)