Phileasson (talk | contribs) revert, removed blacklisted link |
|||
| (257 intermediate revisions by 73 users not shown) | |||
| Line 8: | Line 8: | ||
| species = '''''West Nile virus''''' |
| species = '''''West Nile virus''''' |
||
}} |
}} |
||
'''West Nile virus''' (WNV) is a [[virus]] of the family ''[[Flaviviridae]]''. Part of the [[Japanese encephalitis]] (JE) antigenic complex of viruses, it is found in both [[tropics|tropical]] and [[temperate]] regions. It mainly infects [[bird]]s, but is known to infect [[human]]s, [[horse]]s, [[dog]]s, [[cat]]s, [[bat]]s, [[chipmunk]]s, [[skunk]]s, [[squirrel]]s, domestic [[rabbit]]s, [[crow]]s, [[Muscicapoidea|robins]], [[crocodile]]s<ref name="Steinman 2003">{{Cite pmid|12899140}}</ref> and [[alligator]]s.<ref name="Klenk 2004">{{Cite pmid| 15663852}}</ref> The main route of human [[infection]] is through the bite of an infected [[mosquito]]. Approximately 80% of West Nile virus infections in humans are without any symptoms.<ref>{{cite web |url=http://www.CDC.gov/ncidod/Dvbid/westnile/WNV_factsheet.htm |title=West Nile Virus: What You Need to Know CDC Fact Sheet |publisher=www.CDC.gov |accessdate=2012.04.09 }}</ref> |
|||
Image reconstructions and [[cryoelectron microscopy]] reveal a 45–50 nm [[virion]] covered with a relatively smooth [[protein]] surface. This structure is similar to the [[dengue fever]] virus; both belong to the genus ''Flavivirus'' within the family ''Flaviviridae''. The genetic material of WNV is a [[Sense (molecular biology)|positive-sense]], single strand of [[RNA]], which is between 11,000 and 12,000 [[nucleotide]]s long; these [[genes]] encode seven nonstructural proteins and three structural proteins. The RNA strand is held within a nucleocapsid formed from 12-[[kDa]] protein blocks; the capsid is contained within a host-derived [[Biological membrane|membrane]] altered by two viral [[glycoprotein]]s. |
|||
{{Infobox disease |
{{Infobox disease |
||
| Name = West Nile Fever |
| Name = West Nile Fever |
||
| DiseasesDB = 30025 |
| DiseasesDB = 30025 |
||
| ICD10 = {{ICD10|A|92|3}} |
| ICD10 = {{ICD10|A|92|3}} |
||
| ICD9 = {{ICD9|066.4}} |
| ICD9 = {{ICD9|066.4}} |
||
| MedlinePlus = 007186 |
|||
| MeshID = D014901 |
| MeshID = D014901 |
||
}}</center> |
|||
}} |
|||
==Symptoms== |
|||
'''West Nile virus''' (WNV) is a [[Mosquito-borne disease|mosquito-borne]] [[zoonotic]] [[arbovirus]] belonging to the [[genus]] Flavivirus in the [[Family (biology)|family]] ''[[Flaviviridae]]''. This [[flavivirus]] is found in [[temperate]] and [[tropics|tropical]] regions of the world. It was first identified in the [[West Nile sub-region]] in the [[East African]] nation of [[Uganda]] in 1937. |
|||
The West Nile virus produces one of three different outcomes in humans. The first is an [[asymptomatic]] infection; the second is a mild [[Fever|febrile]] syndrome termed West Nile fever;<ref>{{cite journal |author=Olejnik E |title=Infectious adenitis transmitted by ''Culex molestus |journal=Bull Res Counc Isr |volume=2 |issue= |pages=210–1 |year=1952}}</ref> the third is a [[Neurotropic virus|neuroinvasive disease]] termed West Nile [[meningitis]] or [[encephalitis]].<ref>{{cite journal |author=Smithburn KC, Jacobs HR |title=Neutralization-tests against neurotropic viruses with sera collected in central Africa |journal=Journal of Immunology |volume=44 |issue= |pages=923 |year=1942}}</ref> The population proportion of these three states is roughly 110:30:1.<ref>{{cite journal |author=Tsai TF, Popovici F, Cernescu C, Campbell GL, Nedelcu NI |title=West Nile encephalitis epidemic in southeastern Romania |journal=Lancet |volume=352 |issue=9130 |pages=767–71 |year=1998 |pmid=9737281 |doi= 10.1016/S0140-6736(98)03538-7|url=http://linkinghub.elsevier.com/retrieve/pii/S0140673698035387}}</ref> |
|||
Prior to the mid 1990's, WNV disease occurred only sporadically and was considered a minor risk for humans, until an outbreak in [[Algeria]] in 1994 with cases of WNV caused encephalitis, and the first large outbreak in [[Romania]] in 1996, with a high number of cases with [[Neurotropic virus|neuroinvasive disease]], WNV has now spread globally, with the first case in the [[Western Hemisphere]] being identified in [[New York City]] in 1999<ref>{{cite journal |author=Nash D, Mostashari F et al. |title=THE OUTBREAK OF WEST NILE VIRUS INFECTION IN THE NEW YORK CITY AREA IN 1999 |journal=NEJM |volume=344 |issue=24 |pages=1807-12 |year=2001 |pmid=11407341 |doi= }}</ref>, over the next 5 years, the virus spread across the [[continental United States]], north into [[Canada]], and southward into the [[Caribbean Islands]] and [[Latin America]]. WNV also spread to [[Europe]], beyond the [[Mediterranean Basin]] (a new strain of the virus was recently (2012) identified in [[Italy]]). WNV is now considered to be an [[endemic]] [[pathogen]] in Africa, Asia, [[Australia]], the Middle East, Europe and in the United States which in 2012 has experienced one of its worst [[epidemic]]s. |
|||
The main mode of WNV transmission is via various [[species]] of [[mosquito]]s which are the prime [[Vector (epidemiology)|vector]], with [[bird]]s being the most commonly infected animal and serving as the prime [[Host (biology)|reservoir host]] - especially [[passerines]] which are of the largest order ([[Passeriformes]]) of birds. WNV has been found in various species of [[ticks]], but current research suggests they are not important vectors of the virus. WNV also infects various [[mammal]] [[species]] including [[human]]s and has been identified in [[Reptile|reptilian]] species, including [[alligator]]s and [[crocodile]]s, and also in [[amphibians]].Not all animal species which are susceptible to WNV infection - humans included, and not all bird species - develop sufficient viral levels to transmit the disease to noninfected mosquitoes, and are thus not considered major factors in WNV transmission.<ref name="Steinman 2003">{{Cite pmid|12899140}}</ref><ref name="Klenk 2004">{{Cite pmid| 15663852}}</ref> |
|||
The second, febrile stage has an [[incubation period]] of two to eight days followed by fever, headache, chills, [[diaphoresis]] (excessive sweating), weakness, [[lymphadenopathy]] (swollen lymph nodes), drowsiness, pain in the joints and symptoms like those of influenza. Occasionally, some patients experience a short-lived truncal rash or gastrointestinal symptoms, including nausea, vomiting, loss of appetite, or diarrhea. Symptoms are generally resolved within seven to 10 days, although fatigue can persist for some weeks and lymphadenopathy up to two months. |
|||
Approximately 80 percent of West Nile virus infections in humans are [[Subclinical infection|subclinical]], which cause [[Asymptomatic|no symptoms]].<ref>{{cite web |url=http://www.CDC.gov/ncidod/Dvbid/westnile/WNV_factsheet.htm |title=West Nile Virus: What You Need to Know CDC Fact Sheet |publisher=www.CDC.gov |accessdate=2012.04.09 }}</ref>In the cases where symptoms do occur - termed West Nile Fever in cases without neurological disease - the time from infection to the appearance of symptoms ([[incubation period]]) is typically between 2-15 days. Symptoms may include [[fever]], [[headache]]s, [[fatigue]], [[muscle pain]] or [[Muscle ache|aches]], [[malaise]], [[nausea]], [[anorexia]], [[Emesis|vomiting]], [[myalgia]]s and [[rash]]. Less than 1% of the cases are severe and result in neurological disease when the [[central nervous system]] is affected. People of advanced age, the very young, or those with [[immunosuppression]], either from medical induced such as those taking [[immunosupressive drug]]s or due to a preexisting medical condition such as [[HIV/AIDS|HIV infection]], are most susceptible. The specific neurological diseases which may occur are, West Nile [[encephalitis]] which causes [[inflammation]] of the [[Human brain|brain]], West Nile [[meningitis]] which causes inflammation of the [[meninges]], which are the protective [[membrane]]s that cover the brain and [[spinal cord]], West Nile meningoencephalitis which causes inflammation of the brain and also the meninges surrounding it, and West Nile [[Poliomyelitis]] - spinal cord inflammation which results in a syndrome similar to [[polio]], which may cause [[acute flaccid paralysis]]. |
|||
The more dangerous encephalitis is characterized by similar early symptoms, but also a decreased level of consciousness, sometimes approaching near-[[coma]]. Deep tendon reflexes are hyperactive at first, later diminished. There are also [[extrapyramidal disorder]]s. Recovery is marked by a long [[convalescence]] with [[Fatigue (medical)|fatigue]]. |
|||
There is currently no [[vaccine]] against WNV infection. The best methods to reduce the rates of WNV infection are [[mosquito control]] on the part of municipalities, businesses and individual citizens to reduce breeding populations of mosquitoes in public, commercial and private areas via various means including eliminating standing pools of water where mosquitos breed, such as in old [[tire]]s, buckets, disused [[swimming pool]]s, etc. On an individual basis, the use of personal protective measures to avoid being bitten by an infected mosquito, via the use of [[mosquito repellent]], [[window screen]]s, avoiding areas where mosquitos are more prone to congregate, such as near [[marsh|marshes]], areas with heavy vegetation etc., and being more vigilant from dusk to dawn when mosquitoes are most active offers the best defense. In the advent of being bitten by an infected mosquito, familiarity of the symptoms of WNV on the part of [[layperson]]s, [[physician]]s and [[allied health]] professionals affords the best chance of receiving timely medical treatment which may aid in reducing associated possible [[Complication (medicine)|complications]] and also appropriate [[palliative care]]. |
|||
More recent outbreaks have resulted in a deeper study of the disease and other, rarer, outcomes have been identified. The spinal cord may be infected, marked by anterior myelitis with or without encephalitis.<ref>{{cite journal |author=Sejvar JJ, Haddad MB, Tierney BC, ''et al.'' |title=Neurologic manifestations and outcome of West Nile virus infection |journal=JAMA |volume=290 |issue=4 |pages=511–5 |year=2003 |pmid=12876094 |doi=10.1001/jama.290.4.511 |url=}}</ref> WNV-associated [[Guillain-Barré syndrome]] has been identified<ref>{{cite journal |author=Ahmed S, Libman R, Wesson K, Ahmed F, Einberg K |title=Guillain-Barré syndrome: An unusual presentation of West Nile virus infection |journal=Neurology |volume=55 |issue=1 |pages=144–6 |year=2000 |pmid=10891928 |doi= |url=http://www.neurology.org/cgi/pmidlookup?view=long&pmid=10891928}}</ref> and other rare effects include multifocal [[chorioretinitis]] (which has 100% specificity for identifying WNV infection in patients with possible WNV encephalitis),<ref>{{cite journal |author=Abroug F, Ouanes-Besbes L, Letaief M, ''et al.'' |title=A cluster study of predictors of severe West Nile virus infection |journal=Mayo Clin. Proc. |volume=81 |issue=1 |pages=12–6 |year=2006 |pmid=16438473 |doi= 10.4065/81.1.12|url=}}</ref> [[hepatitis]], [[myocarditis]], [[nephritis]], [[pancreatitis]], and [[splenomegaly]].<ref>{{cite journal |author=Perelman A, Stern J |title=Acute pancreatitis in West Nile Fever |journal=Am. J. Trop. Med. Hyg. |volume=23 |issue=6 |pages=1150–2 |year=1974 |pmid=4429184 |doi= |url=http://www.ajtmh.org/cgi/pmidlookup?view=long&pmid=4429184}}</ref><ref>{{cite journal |author=Omalu BI, Shakir AA, Wang G, Lipkin WI, Wiley CA |title=Fatal fulminant pan-meningo-polioencephalitis due to West Nile virus |journal=Brain Pathol. |volume=13 |issue=4 |pages=465–72 |year=2003 |pmid=14655752 |doi= 10.1111/j.1750-3639.2003.tb00477.x|url=}}</ref><ref>{{cite journal |author=Mathiot CC, Georges AJ, Deubel V |title=Comparative analysis of West Nile virus strains isolated from human and animal hosts using monoclonal antibodies and cDNA restriction digest profiles |journal=Res. Virol. |volume=141 |issue=5 |pages=533–43 |year=1990 |pmid=1703658 |doi= 10.1016/0923-2516(90)90085-W|url=}}</ref> |
|||
==Virus transmission== |
|||
==Transmission and susceptibility== |
|||
[[File:CulexNil.jpg|thumb| 275px | right | The [[proboscis]] of a female mosquito - here a Southern House Mosquito (''Culex quinquefasciatus'') - pierces the [[epidermis]] and [[dermis]] to allow it to [[Blood meal|feed]] on human [[blood]] from a [[capillary]]: this one is almost fully engorged. The mosquito injects [[saliva]] which contains an [[anesthetic]], and an [[anticoagulant]] into the puncture wound; and in infected mosquitoes, the West Nile virus.]] |
|||
===Transmission=== |
|||
[[Image:Aedes albopictus 2.jpg| thumb | 245px | right | The proboscis of an ''[[Aedes albopictus]]'' [[mosquito]] pierces the skin to allow it to feed on human blood. Under experimental conditions, the ''A. albopictus'' mosquito (Asian tiger mosquito) has been found to be a vector of West Nile virus.]] |
|||
The virus is transmitted through mosquito [[Vector (epidemiology)|vectors]], which bite and infect birds. The birds are amplifying hosts, developing sufficient viral levels to transmit the infection to other biting mosquitoes which go on to infect other birds (in the [[Western Hemisphere]], the [[American robin]] and the [[American crow]] are the most common carriers) and also humans. The infected mosquito species vary according to geographical area; in the US, ''[[Culex pipiens]]'' (Eastern US), ''[[Culex tarsalis]]'' (Midwest and West), and ''[[Culex quinquefasciatus]]'' (Southeast) are the main sources.<ref>{{cite journal |author=Hayes EB, Komar N, Nasci RS, Montgomery SP, O'Leary DR, Campbell GL |title=Epidemiology and transmission dynamics of West Nile virus disease |journal=Emerging Infect. Dis. |volume=11 |issue=8 |pages=1167–73 |year=2005 |pmid=16102302 |url=http://www.cdc.gov/ncidod/EID/vol11no08/05-0289a.htm}}</ref> |
The virus is transmitted through mosquito [[Vector (epidemiology)|vectors]], which bite and infect birds. The birds are amplifying hosts, developing sufficient viral levels to transmit the infection to other biting mosquitoes which go on to infect other birds (in the [[Western Hemisphere]], the [[American robin]] and the [[American crow]] are the most common carriers) and also humans. The infected mosquito species vary according to geographical area; in the US, ''[[Culex pipiens]]'' (Eastern US), ''[[Culex tarsalis]]'' (Midwest and West), and ''[[Culex quinquefasciatus]]'' (Southeast) are the main sources.<ref>{{cite journal |author=Hayes EB, Komar N, Nasci RS, Montgomery SP, O'Leary DR, Campbell GL |title=Epidemiology and transmission dynamics of West Nile virus disease |journal=Emerging Infect. Dis. |volume=11 |issue=8 |pages=1167–73 |year=2005 |pmid=16102302 |url=http://www.cdc.gov/ncidod/EID/vol11no08/05-0289a.htm}}</ref> |
||
In mammals, the virus does not multiply as readily (i.e. does not develop high [[viremia]] during infection), and mosquitoes biting infected mammals are not believed to ingest sufficient virus to become infected,<ref>{{cite journal |author=Taylor RM, Hurlbut HS, Dressler HR, Spangler EW, Thrasher D |title=Isolation of West Nile virus from Culex mosquitoes |journal=J Egypt Med Assoc |volume=36 |issue=3 |pages=199–208 |year=1953 |pmid=13084817 }}</ref> making mammals so-called dead-end infections. |
In mammals, the virus does not multiply as readily (i.e. does not develop high [[viremia]] during infection), and mosquitoes biting infected mammals are not believed to ingest sufficient virus to become infected,<ref>{{cite journal |author=Taylor RM, Hurlbut HS, Dressler HR, Spangler EW, Thrasher D |title=Isolation of West Nile virus from Culex mosquitoes |journal=J Egypt Med Assoc |volume=36 |issue=3 |pages=199–208 |year=1953 |pmid=13084817 }}</ref> making mammals so-called dead-end infections. |
||
A 2004 paper in ''Science'' reported ''Culex pipiens'' mosquitoes existed in two populations in [[Europe]], one |
A 2004 paper in ''Science'' reported that ''Culex pipiens'' mosquitoes existed in two populations in [[Europe]], one that bites birds and one that bites humans. In North America, 40% of ''C. pipiens'' mosquitoes were found to be hybrids of the two types which bite both birds and humans, providing a vector for WNV. This is argued to provide an explanation of why the West Nile disease has spread more quickly in North America than Europe.<ref>{{cite journal |author=Fonseca DM, ''et al.'' |title=Emerging vectors in the Culex pipiens complex|journal=Science|volume=303 |issue=5663 |pages=1535–8 |year=2004 |month=March |pmid=15001783 |pmc= |doi=10.1126/science.1094247 |url=}}</ref> However, these conclusions have been disputed.<ref>{{cite journal |author=Spielman A, ''et al.'' |title=Outbreak of West Nile Virus in North America|journal=Science|volume=306 |issue=5701 |pages=1473–5 |year=2004 |month=November |pmid=15567836 |pmc= |doi=10.1126/science.306.5701.1473c |url=}}</ref> In 2010, the Greek Center for Disease Control and Prevention verified that ''C. pipiens'' was responsible for an outbreak of the virus in northern Greece.<ref name=kathimerini>{{cite web |url=http://www.ekathimerini.com/4dcgi/_w_articles_politics_2_21/08/2010_119182 |title=Virus culprit tracked down |publisher=[[Kathimerini]] |accessdate=2010-08-21 |date=2010-08-21 }}</ref> |
||
Direct human-to-human transmission initially was believed to be caused only by occupational exposure,<ref>{{cite journal |author= |title=Laboratory-acquired West Nile virus infections—United States, 2002 |journal=MMWR Morb. Mortal. Wkly. Rep. |volume=51 |issue=50 |pages=1133–5 |year=2002 |pmid=12537288 |doi= |url= |author1= Centers for Disease Control and Prevention (CDC)}}</ref> or conjunctive exposure to infected blood.<ref>{{cite journal |author=Fonseca K, Prince GD, Bratvold J, ''et al.'' |title=West Nile virus infection and conjunctive exposure |journal=Emerging Infect. Dis. |volume=11 |issue=10 |pages=1648–9 |year=2005 |pmid=16355512 |doi= |url=}}</ref> The US outbreak identified additional transmission methods through blood transfusion,<ref>{{cite journal |author= |title=Investigation of blood transfusion recipients with West Nile virus infections |journal=MMWR Morb. Mortal. Wkly. Rep. |volume=51 |issue=36 |pages=823 |year=2002 |pmid=12269472 |doi= |url= |author1= Centers for Disease Control and Prevention (CDC)}}</ref> organ transplant,<ref>{{cite journal |author= |title=West Nile virus infection in organ donor and transplant recipients—Georgia and Florida, 2002 |journal=MMWR Morb. Mortal. Wkly. Rep. |volume=51 |issue=35 |pages=790 |year=2002 |pmid=12227442 |doi= |url= |author1= Centers for Disease Control and Prevention (CDC)}}</ref> intrauterine exposure,<ref>{{cite journal |author= |title=Intrauterine West Nile virus infection—New York, 2002 |journal=MMWR Morb. Mortal. Wkly. Rep. |volume=51 |issue=50 |pages=1135–6 |year=2002 |pmid=12537289 |author1= Centers for Disease Control and Prevention (CDC) }}</ref> and breast feeding.<ref>{{cite journal |author= |title=Possible West Nile virus transmission to an infant through breast-feeding—Michigan, 2002 |journal=MMWR Morb. Mortal. Wkly. Rep. |volume=51 |issue=39 |pages=877–8 |year=2002 |pmid=12375687 |author1= Centers for Disease Control and Prevention (CDC) }}</ref> Since 2003, blood banks in the US routinely screen for the virus among their donors.<ref>{{cite journal |author= |title=Detection of West Nile virus in blood donations—United States, 2003 |journal=MMWR Morb. Mortal. Wkly. Rep. |volume=52 |issue=32 |pages=769–72 |year=2003 |pmid=12917583 |url=http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5232a3.htm |author1= Centers for Disease Control and Prevention (CDC)}}</ref> As a precautionary measure, the UK's [[National Blood Service]] initially ran a test for this disease in donors who donate within 28 days of a visit to the United States, [[Canada]] or the northeastern provinces of Italy and the [[Scottish National Blood Transfusion Service]]<ref>[http://www.scotblood.co.uk/westNile.asp SNBTS position on West Nile virus]</ref> asks prospective donors to wait 28 days after returning from North America or the northeastern provinces of Italy before donating. |
|||
===Susceptibility=== |
|||
Direct human-to-human transmission initially was believed to be caused only by occupational exposure,<ref>{{cite journal |author= |title=Laboratory-acquired West Nile virus infections—United States, 2002 |journal=MMWR Morb. Mortal. Wkly. Rep. |volume=51 |issue=50 |pages=1133–5 |year=2002 |pmid=12537288 |doi= |url= |author1= Centers for Disease Control and Prevention (CDC)}}</ref> or conjunctival exposure to infected blood.<ref>{{cite journal |author=Fonseca K, Prince GD, Bratvold J, ''et al.'' |title=West Nile virus infection and conjunctival exposure |journal=Emerging Infect. Dis. |volume=11 |issue=10 |pages=1648–9 |year=2005 |pmid=16355512 |doi= |url=}}</ref> The US outbreak revealed novel transmission methods, through blood transfusion,<ref>{{cite journal |author= |title=Investigation of blood transfusion recipients with West Nile virus infections |journal=MMWR Morb. Mortal. Wkly. Rep. |volume=51 |issue=36 |pages=823 |year=2002 |pmid=12269472 |doi= |url= |author1= Centers for Disease Control and Prevention (CDC)}}</ref> organ transplant,<ref>{{cite journal |author= |title=West Nile virus infection in organ donor and transplant recipients—Georgia and Florida, 2002 |journal=MMWR Morb. Mortal. Wkly. Rep. |volume=51 |issue=35 |pages=790 |year=2002 |pmid=12227442 |doi= |url= |author1= Centers for Disease Control and Prevention (CDC)}}</ref> intrauterine exposure,<ref>{{cite journal |author= |title=Intrauterine West Nile virus infection—New York, 2002 |journal=MMWR Morb. Mortal. Wkly. Rep. |volume=51 |issue=50 |pages=1135–6 |year=2002 |pmid=12537289 |author1= Centers for Disease Control and Prevention (CDC) }}</ref> and breast feeding.<ref>{{cite journal |author= |title=Possible West Nile virus transmission to an infant through breast-feeding—Michigan, 2002 |journal=MMWR Morb. Mortal. Wkly. Rep. |volume=51 |issue=39 |pages=877–8 |year=2002 |pmid=12375687 |author1= Centers for Disease Control and Prevention (CDC) }}</ref> Since 2003, blood banks in the US routinely screen for the virus among their donors.<ref>{{cite journal |author= |title=Detection of West Nile virus in blood donations—United States, 2003 |journal=MMWR Morb. Mortal. Wkly. Rep. |volume=52 |issue=32 |pages=769–72 |year=2003 |pmid=12917583 |url=http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5232a3.htm |author1= Centers for Disease Control and Prevention (CDC)}}</ref> As a precautionary measure, the UK's [[National Blood Service]] initially ran a test for this disease in donors who donate within 28 days of a visit to the United States, [[Canada]] or the northeastern provinces of Italy. Currently (September 2011), the policy of the National Blood Service is as follows: |
|||
The more severe outcomes of WNV infection are clearly associated with advancing age<ref>{{cite journal |author=Panthier R, Hannoun C, Beytout D, Mouchet J |title=[Epidemiology of West Nile virus. Study of a center in Camargue.] |language=French |journal=Ann Inst Pasteur, Paris |volume=115 |issue=3 |pages=435–45 |year=1968 |pmid=5711530 }}</ref> and a patient history of organ transplantation<ref>{{cite journal |author=Kumar D, Drebot MA, Wong SJ, ''et al.'' |title=A seroprevalence study of west nile virus infection in solid organ transplant recipients |journal=Am. J. Transplant. |volume=4 |issue=11 |pages=1883–8 |year=2004 |pmid=15476490 |doi=10.1111/j.1600-6143.2004.00592.x }}</ref> and diabetes{{fact|date=August 2012}}. A genetic factor also appears to increase susceptibility to West Nile disease. A mutation of the gene ''[[CCR5]]'' gives some protection against [[HIV]] but leads to more serious complications of WNV infection. Carriers of two mutated copies of ''CCR5'' made up 4.0 to 4.5% of a sample of West Nile disease sufferers, while the incidence of the gene in the general population is only 1.0%.<ref>{{cite journal | first = WG | last = Glass | coauthors = Lim JK, Cholera R, Pletnev AG, Gao JL, Murphy PM | year = 2005 | month = October 17 | title = Chemokine receptor CCR5 promotes leukocyte trafficking to the brain and survival in West Nile virus infection | journal = Journal of Experimental Medicine | volume = 202 | issue = 8 | pages = 1087–98 | pmid = 16230476 | doi = 10.1084/jem.20042530 | pmc = 2213214}}</ref><ref>{{cite journal | first = WG | last = Glass | coauthors = McDermott DH, Lim JK, Lekhong S, Yu SF, Frank WA, Pape J, Cheshier RC, Murphy PM | year = 2006 | month = January 23 | title = CCR5 deficiency increases risk of symptomatic West Nile virus infection | journal = Journal of Experimental Medicine | volume = 203 | issue = 1 | pages = 35–40 | pmid = 16418398 | doi = 10.1084/jem.20051970 | pmc = 2118086}}</ref> |
|||
"In the last year there have been significant outbreaks in mainland Greece, Romania, Albania, Israel and in the south west of the Russian Federation just north of the Black and Caspian Seas. In recent years North Eastern Italy in the provincial districts of Ferrara, Rovigo, Mantua, Modena, Bologna and Reggio Emilia (an area north of Rimini and east of Parma) have also been affected. |
|||
Donors who have visited a WNV endemic area between April 1st and November 30th may donate blood four weeks after their return, as long as they have had neither symptoms nor evidence of infection. If the donor was diagnosed with WNV, or had a history of symptoms suggestive of WNV, whilst in the endemic area or following their return then they must wait 6 months before donating."<ref>[http://www.blood.co.uk/can-i-give-blood/west-nile-virus/]</ref> |
|||
Recently, the potential for [[Mosquito#Saliva|mosquito saliva]] to impact the course of WNV disease was demonstrated.<ref name="pmid18000543">{{cite journal |author=Schneider BS, McGee CE, Jordan JM, Stevenson HL, Soong L, Higgs S |title=Prior exposure to uninfected mosquitoes enhances mortality in naturally-transmitted west nile virus infection |journal=PLoS ONE |volume=2 |issue=11 |pages=e1171 |year=2007 |pmid=18000543 |doi=10.1371/journal.pone.0001171 |url=http://www.plosone.org/article/info:doi/10.1371/journal.pone.0001171 |pmc=2048662}}</ref><ref name="pmid16896145">{{cite journal |author=Styer LM, Bernard KA, Kramer LD |title=Enhanced early West Nile virus infection in young chickens infected by mosquito bite: effect of viral dose |journal=Am. J. Trop. Med. Hyg. |volume=75 |issue=2 |pages=337–45 |year=2006 |pmid=16896145 |doi= |url=http://www.ajtmh.org/cgi/pmidlookup?view=long&pmid=16896145}}</ref><ref name="pmid16553552">{{cite journal |author=Schneider BS, Soong L, Girard YA, Campbell G, Mason P, Higgs S |title=Potentiation of West Nile encephalitis by mosquito feeding |journal=Viral Immunol. |volume=19 |issue=1 |pages=74–82 |year=2006 |pmid=16553552 |doi=10.1089/vim.2006.19.74}}</ref> Mosquitoes inoculate their saliva into the skin while obtaining blood. Mosquito saliva is a pharmacological cocktail of secreted molecules, principally proteins, that can affect vascular constriction, [[blood coagulation]], [[platelet aggregation]], [[inflammation]], and [[Immunity (medical)|immunity]]. It clearly alters the [[immune response]] in a manner that may be advantageous to a virus.<ref name="pmid15541033">{{cite journal |author=Wasserman HA, Singh S, Champagne DE |title=Saliva of the Yellow Fever mosquito, Aedes aegypti, modulates murine lymphocyte function |journal=Parasite Immunol. |volume=26 |issue=6–7 |pages=295–306 |year=2004 |pmid=15541033 |doi=10.1111/j.0141-9838.2004.00712.x |url=http://www.blackwell-synergy.com/openurl?genre=article&sid=nlm:pubmed&issn=0141-9838&date=2004&volume=26&issue=6-7&spage=295}}</ref><ref name="pmid12693849">{{cite journal |author=Limesand KH, Higgs S, Pearson LD, Beaty BJ |title=Effect of mosquito salivary gland treatment on vesicular stomatitis New Jersey virus replication and interferon alpha/beta expression in vitro |journal=J. Med. Entomol. |volume=40 |issue=2 |pages=199–205 |year=2003 |pmid=12693849 |doi=10.1603/0022-2585-40.2.199}}</ref><ref name="pmid15189245">{{cite journal |author=Wanasen N, Nussenzveig RH, Champagne DE, Soong L, Higgs S |title=Differential modulation of murine host immune response by salivary gland extracts from the mosquitoes Aedes aegypti and Culex quinquefasciatus |journal=Med. Vet. Entomol. |volume=18 |issue=2 |pages=191–9 |year=2004 |pmid=15189245 |doi=10.1111/j.1365-2915.2004.00498.x |url=http://www.blackwell-synergy.com/openurl?genre=article&sid=nlm:pubmed&issn=0269-283X&date=2004&volume=18&issue=2&spage=191}}</ref><ref name="pmid10081770">{{cite journal |author=Zeidner NS, Higgs S, Happ CM, Beaty BJ, Miller BR |title=Mosquito feeding modulates Th1 and Th2 cytokines in flavivirus susceptible mice: an effect mimicked by injection of sialokinins, but not demonstrated in flavivirus resistant mice |journal=Parasite Immunol. |volume=21 |issue=1 |pages=35–44 |year=1999 |pmid=10081770 | doi = 10.1046/j.1365-3024.1999.00199.x|url=http://www.blackwell-synergy.com/openurl?genre=article&sid=nlm:pubmed&issn=0141-9838&date=1999&volume=21&issue=1&spage=35}}</ref> Studies have shown it can specifically modulate the immune response during early virus infection,<ref name="pmid15671753">{{cite journal |author=Schneider BS, Soong L, Zeidner NS, Higgs S |title=Aedes aegypti salivary gland extracts modulate anti-viral and TH1/TH2 cytokine responses to sindbis virus infection |journal=Viral Immunol. |volume=17 |issue=4 |pages=565–73 |year=2004 |pmid=15671753 |doi=10.1089/vim.2004.17.565}}</ref> and mosquito feeding can exacerbate WNV infection, leading to higher [[viremia]] and more severe forms of disease.<ref name="pmid18000543"/><ref name="pmid16896145"/><ref name="pmid16553552"/> It is unknown what benefit, if any, the mosquito receives by assisting the virus in this manner, so it is likely the virus is simply exploiting the preexisting qualities of mosquito saliva developed for other purposes.{{says who?|date=August 2012}} |
|||
The [[Scottish National Blood Transfusion Service]]<ref>[http://www.scotblood.co.uk/westNile.asp SNBTS position on West Nile virus]</ref> is to ask prospective donors to wait 28 days after returning from North America or the northeastern provinces of Italy before donating. |
|||
The more severe outcomes of WNV infection are clearly associated with advancing age<ref>{{cite journal |author=Panthier R, Hannoun C, Beytout D, Mouchet J |title=[Epidemiology of West Nile virus. Study of a center in Camargue.] |language=French |journal=Ann Inst Pasteur, Paris |volume=115 |issue=3 |pages=435–45 |year=1968 |pmid=5711530 }}</ref> and a patient history of organ transplantation<ref>{{cite journal |author=Kumar D, Drebot MA, Wong SJ, ''et al.'' |title=A seroprevalence study of west nile virus infection in solid organ transplant recipients |journal=Am. J. Transplant. |volume=4 |issue=11 |pages=1883–8 |year=2004 |pmid=15476490 |doi=10.1111/j.1600-6143.2004.00592.x }}</ref> and diabetes. A genetic factor also appears to increase susceptibility to West Nile disease. A mutation of the gene ''[[CCR5]]'' gives some protection against [[HIV]] but leads to more serious complications of WNV infection. Carriers of two mutated copies of ''CCR5'' made up 4.0 to 4.5% of a sample of West Nile disease sufferers, while the incidence of the gene in the general population is only 1.0%.<ref>{{cite journal | first = WG | last = Glass | coauthors = Lim JK, Cholera R, Pletnev AG, Gao JL, Murphy PM | year = 2005 | month = October 17 | title = Chemokine receptor CCR5 promotes leukocyte trafficking to the brain and survival in West Nile virus infection | journal = Journal of Experimental Medicine | volume = 202 | issue = 8 | pages = 1087–98 | pmid = 16230476 | doi = 10.1084/jem.20042530 | pmc = 2213214}}</ref><ref>{{cite journal | first = WG | last = Glass | coauthors = McDermott DH, Lim JK, Lekhong S, Yu SF, Frank WA, Pape J, Cheshier RC, Murphy PM | year = 2006 | month = January 23 | title = CCR5 deficiency increases risk of symptomatic West Nile virus infection | journal = Journal of Experimental Medicine | volume = 203 | issue = 1 | pages = 35–40 | pmid = 16418398 | doi = 10.1084/jem.20051970 | pmc = 2118086}}</ref> |
|||
Recently, the potential for [[Mosquito#Saliva|mosquito saliva]] to impact the course of WNV disease was demonstrated.<ref name="pmid18000543">{{cite journal |author=Schneider BS, McGee CE, Jordan JM, Stevenson HL, Soong L, Higgs S |title=Prior exposure to uninfected mosquitoes enhances mortality in naturally-transmitted west nile virus infection |journal=PLoS ONE |volume=2 |issue=11 |pages=e1171 |year=2007 |pmid=18000543 |doi=10.1371/journal.pone.0001171 |url=http://www.plosone.org/article/info:doi/10.1371/journal.pone.0001171 |pmc=2048662}}</ref><ref name="pmid16896145">{{cite journal |author=Styer LM, Bernard KA, Kramer LD |title=Enhanced early West Nile virus infection in young chickens infected by mosquito bite: effect of viral dose |journal=Am. J. Trop. Med. Hyg. |volume=75 |issue=2 |pages=337–45 |year=2006 |pmid=16896145 |doi= |url=http://www.ajtmh.org/cgi/pmidlookup?view=long&pmid=16896145}}</ref><ref name="pmid16553552">{{cite journal |author=Schneider BS, Soong L, Girard YA, Campbell G, Mason P, Higgs S |title=Potentiation of West Nile encephalitis by mosquito feeding |journal=Viral Immunol. |volume=19 |issue=1 |pages=74–82 |year=2006 |pmid=16553552 |doi=10.1089/vim.2006.19.74}}</ref> Mosquitoes inoculate their saliva into the skin while obtaining blood. Mosquito saliva is a pharmacologic cocktail of secreted molecules, principally proteins, that can affect vascular constriction, [[blood coagulation]], [[platelet aggregation]], [[inflammation]], and [[Immunity (medical)|immunity]]. It clearly alters the [[immune response]] in a manner that may be advantageous to a virus.<ref name="pmid15541033">{{cite journal |author=Wasserman HA, Singh S, Champagne DE |title=Saliva of the Yellow Fever mosquito, Aedes aegypti, modulates murine lymphocyte function |journal=Parasite Immunol. |volume=26 |issue=6–7 |pages=295–306 |year=2004 |pmid=15541033 |doi=10.1111/j.0141-9838.2004.00712.x |url=http://www.blackwell-synergy.com/openurl?genre=article&sid=nlm:pubmed&issn=0141-9838&date=2004&volume=26&issue=6-7&spage=295}}</ref><ref name="pmid12693849">{{cite journal |author=Limesand KH, Higgs S, Pearson LD, Beaty BJ |title=Effect of mosquito salivary gland treatment on vesicular stomatitis New Jersey virus replication and interferon alpha/beta expression in vitro |journal=J. Med. Entomol. |volume=40 |issue=2 |pages=199–205 |year=2003 |pmid=12693849 |doi=10.1603/0022-2585-40.2.199}}</ref><ref name="pmid15189245">{{cite journal |author=Wanasen N, Nussenzveig RH, Champagne DE, Soong L, Higgs S |title=Differential modulation of murine host immune response by salivary gland extracts from the mosquitoes Aedes aegypti and Culex quinquefasciatus |journal=Med. Vet. Entomol. |volume=18 |issue=2 |pages=191–9 |year=2004 |pmid=15189245 |doi=10.1111/j.1365-2915.2004.00498.x |url=http://www.blackwell-synergy.com/openurl?genre=article&sid=nlm:pubmed&issn=0269-283X&date=2004&volume=18&issue=2&spage=191}}</ref><ref name="pmid10081770">{{cite journal |author=Zeidner NS, Higgs S, Happ CM, Beaty BJ, Miller BR |title=Mosquito feeding modulates Th1 and Th2 cytokines in flavivirus susceptible mice: an effect mimicked by injection of sialokinins, but not demonstrated in flavivirus resistant mice |journal=Parasite Immunol. |volume=21 |issue=1 |pages=35–44 |year=1999 |pmid=10081770 | doi = 10.1046/j.1365-3024.1999.00199.x|url=http://www.blackwell-synergy.com/openurl?genre=article&sid=nlm:pubmed&issn=0141-9838&date=1999&volume=21&issue=1&spage=35}}</ref> Studies have shown it can specifically modulate the immune response during early virus infection,<ref name="pmid15671753">{{cite journal |author=Schneider BS, Soong L, Zeidner NS, Higgs S |title=Aedes aegypti salivary gland extracts modulate anti-viral and TH1/TH2 cytokine responses to sindbis virus infection |journal=Viral Immunol. |volume=17 |issue=4 |pages=565–73 |year=2004 |pmid=15671753 |doi=10.1089/vim.2004.17.565}}</ref> and mosquito feeding can exacerbate WNV infection, leading to higher [[viremia]] and more severe forms of disease.<ref name="pmid18000543"/><ref name="pmid16896145"/><ref name="pmid16553552"/> It is unknown what benefit, if any, the mosquito receives by assisting the virus in this manner, so it is likely the virus is simply exploiting the pre-existing qualities of mosquito saliva developed for other purposes. |
|||
There is no [[vaccine]] for humans. A vaccine for horses ([[ATCvet]] code: {{ATCvet|I05|AA10}}) based on killed viruses exists; some [[zoo]]s have given this vaccine to their birds, although its effectiveness is unknown. Dogs and cats show few if any signs of infection. There have been no known cases of direct canine-human or feline-human transmission; although these pets can become infected, it is unlikely they are, in turn, capable of infecting native mosquitoes and thus continuing the disease cycle.<ref>[http://www.cdc.gov/ncidod/dvbid/westnile/birds&mammals.htm CDC]</ref> |
There is no [[vaccine]] for humans. A vaccine for horses ([[ATCvet]] code: {{ATCvet|I05|AA10}}) based on killed viruses exists; some [[zoo]]s have given this vaccine to their birds, although its effectiveness is unknown. Dogs and cats show few if any signs of infection. There have been no known cases of direct canine-human or feline-human transmission; although these pets can become infected, it is unlikely they are, in turn, capable of infecting native mosquitoes and thus continuing the disease cycle.<ref>[http://www.cdc.gov/ncidod/dvbid/westnile/birds&mammals.htm CDC]</ref> |
||
Avoiding mosquito bites is the most straightforward means to avoid infection;<ref>{{cite journal |author=Hayes EB, Gubler DJ |title=West Nile virus: epidemiology and clinical features of an emerging epidemic in the United States |journal=Annu. Rev. Med. |volume=57 |issue= |pages=181–94 |year=2006 |pmid=16409144 |doi=10.1146/annurev.med.57.121304.131418 |url=}}</ref> remaining indoors (while preventing mosquitoes from entering) at dawn and dusk, wearing light-colored clothing that covers arms and legs, as well as the trunk, and using insect repellents on both skin and clothing (such as [[DEET]], [[picaradin]], or oil of [[lemon eucalyptus]] for skin and [[permethrin]] for clothes) are effective.<ref>{{cite journal |author=Fradin MS, Day JF |title=Comparative efficacy of insect repellents against mosquito bites |journal=N. Engl. J. Med. |volume=347 |issue=1 |pages=13–8 |year=2002 |pmid=12097535 |doi=10.1056/NEJMoa011699 }}</ref> If one becomes infected, generally, treatment is purely supportive: analgesia for the pain of |
Avoiding mosquito bites is the most straightforward means to avoid infection;<ref>{{cite journal |author=Hayes EB, Gubler DJ |title=West Nile virus: epidemiology and clinical features of an emerging epidemic in the United States |journal=Annu. Rev. Med. |volume=57 |issue= |pages=181–94 |year=2006 |pmid=16409144 |doi=10.1146/annurev.med.57.121304.131418 |url=}}</ref> remaining indoors (while preventing mosquitoes from entering) at dawn and dusk, wearing light-colored clothing that covers arms and legs, as well as the trunk, and using insect repellents on both skin and clothing (such as [[DEET]], [[picaradin]], or oil of [[lemon eucalyptus]] for skin and [[permethrin]] for clothes) are effective.<ref>{{cite journal |author=Fradin MS, Day JF |title=Comparative efficacy of insect repellents against mosquito bites |journal=N. Engl. J. Med. |volume=347 |issue=1 |pages=13–8 |year=2002 |pmid=12097535 |doi=10.1056/NEJMoa011699 }}</ref> If one becomes infected, generally, treatment is purely supportive: analgesia for the pain of neurological diseases, and rehydration for nausea, vomiting, or diarrhea; encephalitis may also require airway protection and seizure management. |
||
Reported cases in the U.S. in 2005 exceeded those in 2004, and cases in 2006 exceeded 2005's totals. On August 19, 2006, the [[LA Times]] reported the expected incidence rate of WNV was dropping as the local population becomes exposed to the virus. "In countries like Egypt and Uganda, where West Nile was first detected, people became fully immune to the virus by the time they reached adulthood", federal health officials said.<ref>[http://www.latimes.com/news/local/la-me-westnile19aug19,0,2186893.story?coll=la-home-local CDC]</ref> However, just days later, the CDC said WNV cases could reach a three-year high because hot temperatures had allowed a larger brood of mosquitoes.<ref>[http://news.yahoo.com/s/ap/20060825/ap_on_re_us/west_nile;_ylt=Alqlj1InGYGFq0kRGw4NAoC9SvQA;_ylu=X3oDMTA4dW1uZXIwBHNlYwMyNzQ3 Yahoo]</ref> |
Reported cases in the U.S. in 2005 exceeded those in 2004, and cases in 2006 exceeded 2005's totals. On August 19, 2006, the [[LA Times]] reported the expected incidence rate of WNV was dropping as the local population becomes exposed to the virus. "In countries like Egypt and Uganda, where West Nile was first detected, people became fully immune to the virus by the time they reached adulthood", federal health officials said.<ref>[http://www.latimes.com/news/local/la-me-westnile19aug19,0,2186893.story?coll=la-home-local CDC]</ref> However, just days later, the CDC said WNV cases could reach a three-year high because hot temperatures had allowed a larger brood of mosquitoes.<ref>[http://news.yahoo.com/s/ap/20060825/ap_on_re_us/west_nile;_ylt=Alqlj1InGYGFq0kRGw4NAoC9SvQA;_ylu=X3oDMTA4dW1uZXIwBHNlYwMyNzQ3 Yahoo]</ref> |
||
| Line 58: | Line 48: | ||
CDC numbers for actual cases for the US are likely inaccurate. In poor rural communities, few could afford the expensive test used to confirm the diagnosis. Motivation for people of modest incomes to pay for the test is low when no treatment for the disease is available. Most simply adopted a wait-and-see attitude. For many small riverside communities, the entire population could be infected, with only one or two individuals with severe symptoms being reported. As the first cases recovered, fewer and fewer opted for the test; as a result, the CDC could not track what it did not know. |
CDC numbers for actual cases for the US are likely inaccurate. In poor rural communities, few could afford the expensive test used to confirm the diagnosis. Motivation for people of modest incomes to pay for the test is low when no treatment for the disease is available. Most simply adopted a wait-and-see attitude. For many small riverside communities, the entire population could be infected, with only one or two individuals with severe symptoms being reported. As the first cases recovered, fewer and fewer opted for the test; as a result, the CDC could not track what it did not know. |
||
--> |
--> |
||
Although currently no West Nile virus vaccine is available for humans, many scientists are working on this issue, and hopefully a vaccine will become available in the next few years. |
Although currently no West Nile virus vaccine is available for humans, many scientists are working on this issue, and hopefully a vaccine will become available in the next few years.<ref>[http://www.cdc.gov/ncidod/dvbid/westnile/qa/wnv_vaccine.htm CDC: West Nile Virus - Questions and Answers]</ref> |
||
==Symptoms== |
|||
[[File:West nile virus rash.jpg|thumb|260px|[[Rash]] caused by an infection of West Nile virus in a 44-year-old man. He had a sudden onset of symptoms 7 days prior to seeking medical attention of, [[headache]], [[sore throat]], [[stiff neck]] and [[joint]]s, [[myalgia]], [[nausea]], [[fatigue]], [[fever]] and [[chills]]. The rash developed on the fifth day following symptom onset<ref>Gorsche R, Tilley P. CMAJ. The rash of West Nile virus infection. 2005 May 24;172(11):1440. Gorsche R, Tilley P. PMID 15911857</ref>Development of a rash occurs in less than 1/3 of clinical WNV infections.<ref>Weiss D, Carr D, Kellachan J, ''et al.'' Clinical findings of West Nile virus infection in hospitalized patients, New York and New Jersey, 2000. Emerg Infect Dis. 2001 Jul-Aug;7(4):654-8. PMID 11589170</ref><ref>Pepperell C, Rau N, Krajden S, ''et al.'' West Nile virus infection in 2002: morbidity and mortality among patients admitted to hospital in southcentral Ontario. CMAJ. 2003 May 27;168(11):1399-405. PMID 12771068</ref>]] |
|||
The [[incubation period]] for WNV - the amount of time from infection to symptom onset - is typically from between 2-15 days. Multiple somatic complaints, tremor, and abnormalities in motor skills and executive functions are common long-term problems among patients who have had West Nile virus infection. Patients with milder illness are just as likely as patients with more-severe illness to experience adverse outcomes. |
|||
*'''West Nile Fever (WNF)''', which occurs in 20 percent of cases, is a [[fever|febrile]] [[syndrome]] which causes [[flu-like symptoms]].<ref>{{cite journal |author=Olejnik E |title=Infectious adenitis transmitted by ''Culex molestus |journal=Bull Res Counc Isr |volume=2 |issue= |pages=210–1 |year=1952}}</ref>The [[incubation period]] - the amount of time from infection to symptom onset is typically from between 2-15 days. Most characterizations of WNF generally describe it as a mild, [[acute]] syndrome lasting 3 - 6 days after symptom onset. Systematic follow-up studies of patients with WNF had not previously been done, so this information is largely [[Anecdotal evidence|anecdotal]]. In addition to a high fever, headache, chills, [[diaphoresis|excessive sweating]], weakness, fatigue, [[lymphadenopathy|swollen lymph nodes]], drowsiness, pain in the joints and symptoms like those of [[influenza]]. Occasionally, some patients experience a short-lived truncal rash or gastrointestinal symptoms, including nausea, vomiting, loss of appetite, or diarrhea. |
|||
*'''West Nile [[Neurotropic virus|Neuroinvasive Disease]] (WNND)''', which occurs in less than 1 percent of cases, is when the virus infects the [[central nervous system]] resulting in [[meningitis]], [[encephalitis]], meningoencephalitis or [[poliomyelitis]].<ref>{{cite journal |author=Smithburn KC, Jacobs HR |title=Neutralization-tests against neurotropic viruses with sera collected in central Africa |journal=Journal of Immunology |volume=44 |issue= |pages=923 |year=1942}}</ref>{{medical citation needed|date=September 2012}} |
|||
*'''West Nile virus encephalitis (WNVE)''' is characterized by similar early symptoms, but also a decreased level of consciousness, sometimes approaching near-[[coma]]. [[Deep tendon reflex]]es are hyperactive at first, later diminished. There are also [[extrapyramidal symptoms]]. Recovery is marked by a long [[convalescence]] with [[Fatigue (medical)|fatigue]]. One study found that neuroinvasive WNV infection was associated with an increased risk for subsequent kidney disease.<ref>{{cite journal |
|||
|url=http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0040374 |
|||
|title= Prevalence of Chronic Kidney Disease and Progression of Disease Over Time among Patients Enrolled in the Houston West Nile Virus Cohort |
|||
|author= Melissa S. Nolan, et. al. |
|||
|date= 6 July 2012 |
|||
|work= PLoS One |
|||
|publisher= PLOS |
|||
|accessdate=26 August 2012 |
|||
}}</ref><ref>{{cite web |
|||
|url=http://guardianlv.com/2012/08/new-study-reveals-west-nile-virus-is-far-more-menacing-harms-far-more-people/ |
|||
|title= New Study Reveals: West Nile virus is far more menacing & harms far more people |
|||
|author= |
|||
|date= 26 August 2012 |
|||
|work= The Guardian Express |
|||
|publisher= The Guardian Express |
|||
|accessdate=26 August 2012 |
|||
}}</ref> |
|||
::More recent outbreaks have resulted in a deeper study of the disease and other, rarer, outcomes have been identified. The spinal cord may be infected, marked by anterior [[Myelitis|myelitis]] with or without encephalitis.<ref>{{cite journal |author=Sejvar JJ, Haddad MB, Tierney BC, ''et al.'' |title=Neurologic manifestations and outcome of West Nile virus infection |journal=JAMA |volume=290 |issue=4 |pages=511–5 |year=2003 |pmid=12876094 |doi=10.1001/jama.290.4.511 |url=}}</ref> WNV-associated [[Guillain-Barré syndrome]] has been identified<ref>{{cite journal |author=Ahmed S, Libman R, Wesson K, Ahmed F, Einberg K |title=Guillain-Barré syndrome: An unusual presentation of West Nile virus infection |journal=Neurology |volume=55 |issue=1 |pages=144–6 |year=2000 |pmid=10891928 |doi= |url=http://www.neurology.org/cgi/pmidlookup?view=long&pmid=10891928}}</ref> and other rare effects include multifocal [[chorioretinitis]] (which has 100% specificity for identifying WNV infection in patients with possible WNV encephalitis),<ref>{{cite journal |author=Abroug F, Ouanes-Besbes L, Letaief M, ''et al.'' |title=A cluster study of predictors of severe West Nile virus infection |journal=Mayo Clin. Proc. |volume=81 |issue=1 |pages=12–6 |year=2006 |pmid=16438473 |doi= 10.4065/81.1.12|url=}}</ref> [[hepatitis]], [[myocarditis]], [[nephritis]], [[pancreatitis]], and [[splenomegaly]].<ref>{{cite journal |author=Perelman A, Stern J |title=Acute pancreatitis in West Nile Fever |journal=Am. J. Trop. Med. Hyg. |volume=23 |issue=6 |pages=1150–2 |year=1974 |pmid=4429184 |doi= |url=http://www.ajtmh.org/cgi/pmidlookup?view=long&pmid=4429184}}</ref><ref>{{cite journal |author=Omalu BI, Shakir AA, Wang G, Lipkin WI, Wiley CA |title=Fatal fulminant pan-meningo-polioencephalitis due to West Nile virus |journal=Brain Pathol. |volume=13 |issue=4 |pages=465–72 |year=2003 |pmid=14655752 |doi= 10.1111/j.1750-3639.2003.tb00477.x|url=}}</ref><ref>{{cite journal |author=Mathiot CC, Georges AJ, Deubel V |title=Comparative analysis of West Nile virus strains isolated from human and animal hosts using monoclonal antibodies and cDNA restriction digest profiles |journal=Res. Virol. |volume=141 |issue=5 |pages=533–43 |year=1990 |pmid=1703658 |doi= 10.1016/0923-2516(90)90085-W|url=}}</ref> |
|||
==Diagnosis== |
|||
[[File:IgM.png|thumb|left|140px|An [[IgM|Immunoglobulin M]] (IgM) [[antibody]] [[molecule]]. Definitive diagnosis of WNV is obtained through detection of virus-specific IgM and [[Neutralizing antibody|neutralizing antibodies]].]] |
|||
Preliminary diagnosis is often based on the patient's clinical symptoms, places and dates of travel (if patient is from a non-[[endemic]] country or area), activities, and [[epidemiology|epidemiologic]] history of the location where infection occurred. A recent history of mosquito bites and an acute febrile illness associated with neurologic signs and symptoms should cause clinical suspicion of WNV. Diagnosis of West Nile Virus infections is generally accomplished by [[serology|serologic]] testing of [[blood serum]] or [[cerebrospinal fluid]] (CSF), which is obtained via a [[lumbar puncture]]. Typical findings of WNV infection include [[lymphocytic pleocytosis]], elevated [[protein]] level, reference [[glucose]] and [[lactic acid]] levels, and no [[erythrocytes]]. Definitive diagnosis is of WNV is obtained through detection of virus-specific [[antibody]] [[IgM|Immunoglobulin M]], (IgM) and [[Neutralizing antibody|neutralizing antibodies]]. Cases of West Nile virus meningitis and encephalitis which have been serologically confirmed produce similar degrees of CSF pleocytosis and are often associated with substantial CSF [[neutrophilia]].<ref>Tyler KL, Pape J, Goody RJ, ''et al.'' CSF findings in 250 patients with serologically confirmed West Nile virus meningitis and encephalitis. Neurology. 2006 Feb 14;66(3):361-5. Epub 2005 Dec 28. PMID 16382032</ref> |
|||
Four [[Food and Drug Administration|FDA]]-cleared WNV IgM [[ELISA]] kits from different manufacturers are commercially available in the U.S. According to the package inserts, each of these kits is indicated for use on serum to aid in the presumptive laboratory diagnosis of WNV infection in patients with clinical symptoms of meningitis or encephalitis. The package inserts also state that all positive results obtained with any of the commercially-available WNV test kits should be confirmed by additional testing at a state health department laboratory or CDC. |
|||
In fatal cases, [[nucleic acid]] amplification, [[histopathology]] with [[immunohistochemistry]] and virus culture of [[autopsy]] tissues can also be useful. Only a few state laboratories or other specialized laboratories, including those at CDC, are capable of doing this specialized testing |
|||
==Prognosis== |
|||
While the general prognosis is favorable, current studies indicate that West Nile Fever can often be more severe than previously recognized, with studies of various recent outbreaks indicating that it may take as long as 60 -90 days to recover.<ref>Watson J., Patel P. Jones R., ''et al.'' Clinical Characteristics and Functional Outcomes of West Nile Fever[66.39.42.45/topics/west_nile/pdf/wnv_clinical_characteristics.pdf]</ref><ref>Carson1 P, Konewko P., Wold k., ''et al.'' Long-Term Clinical and Neuropsychological Outcomes of West Nile Virus Infection. Oxford Journals Medicine Clinical Infectious Diseases Volume 43, Issue 6 Pp. 723-730.</ref>Patients with milder WNF are just as likely as those with more severe manifestations of neuroinvasive disease to experience multiple long term (>1+ years) [[somatic]] complaints such as tremor, and dysfunction in [[motor skill]]s and [[executive functions]].<ref>Klee AL, Maidin B, Edwin B, ''et al.'' Long-term prognosis for clinical West Nile virus infection. Emerg Infect Dis. 2004 Aug;10(8):1405-11. PMID 15496241</ref> |
|||
==History== |
==History== |
||
[[File:Uganda (orthographic projection).svg|thumb|Since it was first detected in the [[Africa|African]] nation of [[Uganda]] (shaded green) in 1937, West Nile virus has spread throughout the globe.]] |
|||
Studies of phylogenetic lineages determined WNV emerged as a distinct virus around 1000 years ago.<ref>{{cite journal |author=Galli M, Bernini F, Zehender G |title=Alexander the Great and West Nile virus encephalitis |journal=Emerging Infect. Dis. |volume=10 |issue=7 |pages=1330–2; author reply 1332–3 |year=2004 |month=July |pmid=15338540 }}</ref> This initial virus developed into two distinct lineages, lineage 1 and its multiple profiles is the source of the epidemic transmission in Africa and throughout the world. Lineage 2 was considered an Africa [[zoonose]]. However, in 2008, lineage 2, previously only seen in horses in sub-Saharan Africa and Madagascar, began to appear in horses in Europe, where the first known outbreak affected 18 animals in Hungary in 2008.<ref>{{cite journal| url=http://www.thehorse.com/ViewArticle.aspx?ID=15779| title=Different West Nile Virus Genetic Lineage Evolving?|author=West, Christy|publisher=The Horse, online edition |date = 2010-02-08 |nopp= from statements by Orsolya Kutasi, DVM, of the Szent Istvan University, Hungary at the 2009 American Association of Equine Practitioners Convention, December 5–9 , 2009|accessdate=2010-02-10}}</ref> Lineage 1 West Nile virus was detected in [[South Africa]] in 2010 in a [[mare]] and her aborted [[fetus]]; previously, only lineage 2 West Nile virus had been detected in horses and humans in South Africa.<ref>Venter, M., S. Human, S. van Niekerk, J. Williams, C. van Eeden, and F. Freeman. 2011. Fatal neurologic disease and abortion in mare infected with lineage 1 West Nile virus, South Africa. ''Emerging Infectious Diseases'', 2011 Aug [July 27, 2011], http://www.cdc.gov/EID/content/17/8/101794.htm.</ref> A 2007 fatal case in a [[killer whale]] in [[Texas]] broadened the known [[host range]] of West Nile virus to include [[cetacean]]s.<ref>St. Leger, J., G. Wu, M. Anderson, L. Dalton, E. Nilson, and D. Wang. 2011. West Nile virus infection in killer whale, Texas, USA, 2007. ''Emerging Infectious Diseases'', 2011 Aug [July 27, 2011]. http://www.cdc.gov/EID/content/17/8/101979.htm.</ref> |
|||
Studies of phylogenetic lineages determined WNV emerged as a distinct virus around 1000 years ago.<ref>{{cite journal |author=Galli M, Bernini F, Zehender G |title=Alexander the Great and West Nile virus encephalitis |journal=Emerging Infect. Dis. |volume=10 |issue=7 |pages=1330–2; author reply 1332–3 |year=2004 |month=July |pmid=15338540 }}</ref> This initial virus developed into two distinct lineages, lineage 1 and its multiple profiles is the source of the epidemic transmission in Africa and throughout the world. Lineage 2 was considered an Africa [[zoonose]]. However, in 2008, lineage 2, previously only seen in horses in sub-Saharan Africa and Madagascar, began to appear in horses in Europe, where the first known outbreak affected 18 animals in Hungary in 2008.<ref>{{cite journal| url=http://www.thehorse.com/ViewArticle.aspx?ID=15779| title=Different West Nile Virus Genetic Lineage Evolving?|author=West, Christy|publisher=The Horse, online edition |date = 2010-02-08 |nopp= from statements by Orsolya Kutasi, DVM, of the Szent Istvan University, Hungary at the 2009 American Association of Equine Practitioners Convention, December 5–9 , 2009|accessdate=2010-02-10}}</ref> Lineage 1 West Nile virus was detected in [[South Africa]] in 2010 in a [[mare]] and her aborted [[fetus]]; previously, only lineage 2 West Nile virus had been detected in horses and humans in South Africa.<ref>Venter, M., S. Human, S. van Niekerk, J. Williams, C. van Eeden, and F. Fatal neurologic disease and abortion in mare infected with lineage 1 West Nile virus, South Africa. ''Emerging Infectious Diseases'', 2011 Aug [July 27, 2011], http://www.cdc.gov/EID/content/17/8/101794.htm.</ref> A 2007 fatal case in a [[killer whale]] in [[Texas]] broadened the known [[host range]] of West Nile virus to include [[cetacean]]s.<ref>St. Leger, J., G. Wu, M. Anderson, L. Dalton, E. Nilson, and D. Wang. 2011. West Nile virus infection in killer whale, Texas, USA, 2007. ''Emerging Infectious Diseases'', 2011 Aug [July 27, 2011]. http://www.cdc.gov/EID/content/17/8/101979.htm.</ref> |
|||
WNV has been posited as one of the possible causes of [[Alexander the Great]]'s early death based on reports of avian deaths before his illness period.<ref>[http://www.cdc.gov/ncidod/EID/vol9no12/03-0288.htm "Alexander the Great and West Nile Virus Encephalitis"]. Centers for Disease Control and Prevention. Retrieved on 2009-04-26</ref> |
WNV has been posited as one of the possible causes of [[Alexander the Great]]'s early death based on reports of avian deaths before his illness period.<ref>[http://www.cdc.gov/ncidod/EID/vol9no12/03-0288.htm "Alexander the Great and West Nile Virus Encephalitis"]. Centers for Disease Control and Prevention. Retrieved on 2009-04-26</ref> |
||
| Line 67: | Line 97: | ||
WNV was first isolated from a feverish 37-year-old woman at Omogo in the [[West Nile sub-region|West Nile District]] of [[Uganda]] in 1937 during research on [[yellow fever virus]].<ref>{{cite journal |author=Smithburn KC, Hughes TP, Burke AW, Paul JH |title=A Neurotropic Virus Isolated from the Blood of a Native of Uganda |journal=Am. J. Trop. Med. |volume=20 |issue=1 |pages=471–92 |year=1940 |month=June}}</ref> A series of [[serosurvey]]s in 1939 in central Africa found anti-WNV positive results ranging from 1.4% (Congo) to 46.4% (White Nile region, Sudan). It was subsequently identified in [[Egypt]] (1942) and [[India]] (1953), a 1950 serosurvey in Egypt found 90% of those over 40 years in age had WNV antibodies. The ecology was characterized in 1953 with studies in [[Egypt]]<ref>{{cite journal |author=Work TH, Hurlbut HS, Taylor RM |title=Isolation of West Nile virus from hooded crow and rock pigeon in the Nile delta |journal=Proc. Soc. Exp. Biol. Med. |volume=84 |issue=3 |pages=719–22 |year=1953 |pmid=13134268 |doi= |url=}}</ref> and [[Israel]].<ref>{{cite journal |author=Bernkopf H, Levine S, Nerson R |title=Isolation of West Nile virus in Israel |journal=J. Infect. Dis. |volume=93 |issue=3 |pages=207–18 |year=1953 |pmid=13109233 }}</ref> The virus became recognized as a cause of severe human [[meningoencephalitis]] in elderly patients during an outbreak in Israel in 1957. The disease was first noted in horses in Egypt and [[France]] in the early 1960s and found to be widespread in southern Europe, southwest Asia and Australia. |
WNV was first isolated from a feverish 37-year-old woman at Omogo in the [[West Nile sub-region|West Nile District]] of [[Uganda]] in 1937 during research on [[yellow fever virus]].<ref>{{cite journal |author=Smithburn KC, Hughes TP, Burke AW, Paul JH |title=A Neurotropic Virus Isolated from the Blood of a Native of Uganda |journal=Am. J. Trop. Med. |volume=20 |issue=1 |pages=471–92 |year=1940 |month=June}}</ref> A series of [[serosurvey]]s in 1939 in central Africa found anti-WNV positive results ranging from 1.4% (Congo) to 46.4% (White Nile region, Sudan). It was subsequently identified in [[Egypt]] (1942) and [[India]] (1953), a 1950 serosurvey in Egypt found 90% of those over 40 years in age had WNV antibodies. The ecology was characterized in 1953 with studies in [[Egypt]]<ref>{{cite journal |author=Work TH, Hurlbut HS, Taylor RM |title=Isolation of West Nile virus from hooded crow and rock pigeon in the Nile delta |journal=Proc. Soc. Exp. Biol. Med. |volume=84 |issue=3 |pages=719–22 |year=1953 |pmid=13134268 |doi= |url=}}</ref> and [[Israel]].<ref>{{cite journal |author=Bernkopf H, Levine S, Nerson R |title=Isolation of West Nile virus in Israel |journal=J. Infect. Dis. |volume=93 |issue=3 |pages=207–18 |year=1953 |pmid=13109233 }}</ref> The virus became recognized as a cause of severe human [[meningoencephalitis]] in elderly patients during an outbreak in Israel in 1957. The disease was first noted in horses in Egypt and [[France]] in the early 1960s and found to be widespread in southern Europe, southwest Asia and Australia. |
||
The first appearance of WNV in the Western Hemisphere was in 1999 with encephalitis reported in humans, dogs, cats, and horses, and the subsequent spread in the [[United States]] may be an important milestone in the evolving history of this virus. The American outbreak began in the [[New York City]] area (specifically, [[College Point, Queens]]) and was later seen in [[New Jersey]] and [[Connecticut]]; the virus is believed to have entered in an infected bird or mosquito, although there is no clear evidence.<ref>{{cite journal |author=Calisher CH |title=West Nile virus in the New World: appearance, persistence, and adaptation to a new econiche—an opportunity taken |journal=Viral Immunol. |volume=13 |issue=4 |pages=411–4 |year=2000 |pmid=11192287 |doi=10.1089/vim.2000.13.411 }}</ref> |
The first appearance of WNV in the Western Hemisphere was in 1999<ref>{{cite journal |author=Nash D, Mostashari F et al. |title=THE OUTBREAK OF WEST NILE VIRUS INFECTION IN THE NEW YORK CITY AREA IN 1999 |journal=NEJM |volume=344 |issue=24 |pages=1807-12 |year=2001 |pmid=11407341 |doi= }}</ref> with encephalitis reported in humans, dogs, cats, and horses, and the subsequent spread in the [[United States]] may be an important milestone in the evolving history of this virus. The American outbreak began in the [[New York City]] area (specifically, [[College Point, Queens]]) and was later seen in [[New Jersey]] and [[Connecticut]]; the virus is believed to have entered in an infected bird or mosquito, although there is no clear evidence.<ref>{{cite journal |author=Calisher CH |title=West Nile virus in the New World: appearance, persistence, and adaptation to a new econiche—an opportunity taken |journal=Viral Immunol. |volume=13 |issue=4 |pages=411–4 |year=2000 |pmid=11192287 |doi=10.1089/vim.2000.13.411 }}</ref> |
||
The US virus was very closely related to a lineage 1 strain found in Israel in 1998. Since the first North American cases in 1999, the virus has been reported throughout the United States, Canada, Mexico, the Caribbean and Central America. There have been human cases and equine cases, and many birds are infected. The [[Barbary macaque]], ''[[Macaca sylvanus]]'', was the first nonhuman [[primate]] to contract WNV.<ref>C. Michael Hogan. 2008. [http://globaltwitcher.auderis.se/artspec_information.asp?thingid=31757 Barbary Macaque: Macaca sylvanus, GlobalTwitcher.com]</ref> Both the US and Israeli strains are marked by high mortality rates in infected avian populations; the presence of dead birds—especially [[Corvidae]]—can be an early indicator of the arrival of the virus. |
|||
A [[media hype|high level of media coverage]] through 2001/2002 raised public awareness of WNV. This coverage was most likely the result of successive appearances of the virus in new areas, and had the unintended effect of increasing funding for research on this virus and related [[arthropod]]-borne viruses. Such research has expanded our understanding of viruses transmitted by mosquitoes. |
|||
[[Vertical transmission]] of West Nile virus from ''C. pipiens'' mosquitoes to their progeny has been demonstrated in the laboratory. It has not been suggested that vertically infected ''Culex'' mosquitoes could survive the winter to initiate a WNV [[amplification cycle]] the following spring. ''Culex'' mosquitoes spend the winter hibernating in protected structures such as root cellars, bank barns, caves, abandoned tunnels and other subterranean locations. The first overwintering adult mosquitoes to test positive for WNV were collected in New York in 2000. Since then, positive samples have been identified in New Jersey in 2003, and in Pennsylvania in 2003-05.<ref>{{cite journal | first = LM | last = Bugbee | coauthors = Forte LR | year = 2004 | month = September | title = The discovery of West Nile virus in overwintering Culex pipiens (Diptera: Culicidae) mosquitoes in Lehigh County, Pennsylvania | journal = Journal of the American Mosquito Control Association | volume = 20 | issue = 3 | pages = 326–7 | pmid = 15532939}}</ref> |
|||
==Overwintering mechanism== |
|||
[[Vertical transmission]] of West Nile Virus from ''Culex pipiens'' mosquitoes to their progeny has been demonstrated in the laboratory. It has not been suggested that vertically infected ''Culex'' could survive the winter to initiate a WNV [[amplification cycle]] the following spring. ''Culex'' mosquitoes spend the winter hibernating in protected structures such as root cellars, bank barns, caves, abandoned tunnels and other subterranean locations. The first overwintering adult mosquitoes to test positive for WNV were collected in New York, 2000. Since then, positive samples have been identified in New Jersey, 2003 and in Pennsylvania, 2003, 2004 and 2005.<ref>{{cite journal | first = LM | last = Bugbee | coauthors = Forte LR | year = 2004 | month = September | title = The discovery of West Nile virus in overwintering Culex pipiens (Diptera: Culicidae) mosquitoes in Lehigh County, Pennsylvania | journal = Journal of the American Mosquito Control Association | volume = 20 | issue = 3 | pages = 326–7 | pmid = 15532939}}</ref> |
|||
==Epidemiology== |
|||
[[File:West Nile Virus Map-United States 2012.jpg|thumb|300px|Cases reported to the [[Centers for Disease Control and Prevention|CDC]] of West Nile virus infection in the [[continental United States]] in 2012. (Current as of Aug. 28, 2012).]] |
|||
==Geographic distribution== |
|||
West Nile virus has been described in [[Africa]], [[Europe]], the [[Middle East]], west and central [[Asia]], [[Oceania]] (subtype [[Kunjin virus|Kunjin]]), and most recently, North America. |
West Nile virus has been described in [[Africa]], [[Europe]], the [[Middle East]], west and central [[Asia]], [[Oceania]] (subtype [[Kunjin virus|Kunjin]]), and most recently, North America. |
||
| Line 80: | Line 111: | ||
[[Epizootic]]s of disease in horses occurred in [[Morocco]] (1996), [[Italy]] (1998), the United States (1999 to 2001), and [[France]] (2000). In 2003, West Nile virus was found in horses in Mexico. |
[[Epizootic]]s of disease in horses occurred in [[Morocco]] (1996), [[Italy]] (1998), the United States (1999 to 2001), and [[France]] (2000). In 2003, West Nile virus was found in horses in Mexico. |
||
In 2011, West Nile |
In 2011, West Nile virus was found in horses in Sardinia (Italy). |
||
*'''United States''': From 1999 through 2001, the [[Centers for Disease Control and Prevention|CDC]] confirmed 149 West Nile virus infections, including 18 deaths. In 2002, a total of 4,156 cases were reported, including 284 fatalities. Thirteen cases in 2002 were contracted through blood transfusion. The cost of WNV-related health care in 2002 was estimated at $200 million. The first human West Nile disease in 2003 occurred in June, and one West Nile-infected blood transfusion was also identified that month.<ref>[http://www.cdc.gov/ncidod/dvbid/westnile/surv&control.htm CDC: West Nile Virus - Statistics, Surveillance, and Control<!-- Bot generated title -->]</ref> |
|||
[[Image:Wnv map MX.jpg|right|450px|]] |
|||
In the |
:In the 2003 outbreak, 9,862 cases and 264 deaths were reported by the CDC. At least 30% of those cases were considered severe, involving meningitis or encephalitis. In 2004, only 2,539 cases and 100 deaths were reported. In 2005, there was a slight increase in the number of cases, with 3,000 cases and 119 deaths reported. Cases increased in 2006, with 4,269 cases and 177 deaths. In 2007, the number of cases reported decreased to 3,623 and the number of deaths dropped to 124. In 2007, 1,227 cases of WNV neuroinvasion disease and 117 deaths occurred.<ref>[http://www.cdc.gov/ncidod/dvbid/westnile/surv&control.htm CDC: West Nile Virus - Statistics, Surveillance, and Control<!-- Bot generated title -->]</ref> |
||
:In 2008, West Nile surveillance data reported to CDC, a total of 28 states have reported 236 cases of human WNV illness. A total of 137 cases for which such data were available occurred in males, median age patients was 48 years. Dates of illness onset ranged from January 17 to August 14: Two cases were fatal.<ref>[http://www.cdc.gov/ncidod/dvbid/westnile/surv&control.htm CDC: West Nile Virus - Statistics, Surveillance, and Control<!-- Bot generated title -->]</ref> |
|||
==Recent outbreaks== |
|||
{{see also|Progress of the West Nile virus in the United States}} |
|||
'''United States''': From 1999 through 2001, the [[Centers for Disease Control and Prevention|CDC]] confirmed 149 West Nile virus infections, including 18 deaths. In 2002, a total of 4,156 cases were reported, including 284 fatalities. 13 cases in 2002 were contracted through blood transfusion. The cost of WNV-related health care in 2002 was estimated at $200 million. The first human West Nile disease in 2003 occurred in June and one West Nile-infected blood transfusion was also identified that month. In the 2003 outbreak, 9,862 cases and 264 deaths were reported by the CDC. At least 30% of those cases were considered severe involving meningitis or encephalitis. In 2004, there were only 2,539 reported cases and 100 deaths. In 2005, there was a slight increase in the number of cases, with 3,000 cases and 119 deaths reported. 2006 saw another increase, with 4,269 cases and 177 deaths. In 2007, the number of cases reported decreased to 3,623 and the number of deaths dropped to 124. In 2007, 1,227 cases of wnv neuroinvasion disease and 117 deaths occurred. In 2008, West Nile surveillance data reported to CDC, a total of 28 states have reported 236 cases of human WNV illness. A total of 137 cases for which such data were available occurred in males, median age patients was 48 years. Dates of illness onset ranged from January 17 to August 14: Two cases were fatal. In 2009, there were 663 cases. Three hundred and thirty-five of these cases were encephalitis or meningitis infections, a reaction to the virus that approximately one in 150 people who get the virus will show. Three hundred and two cases were filed for West Nile fever, the most likely symptom of the virus. Twenty-six cases were unspecified. The state of Texas had the most cases, with 104 total. The total mortality rate for 2009 was 32 deaths of the 663 reported serious cases. That is a 4.5% casualty rate, but only of the severe infections. Approximately 80% of cases have no symptoms, so the total casualty rate would be less than 1% of total infections in the U.S. These data and earlier years data are available from the [[Centers for Disease Control and Prevention]].<ref>[http://www.cdc.gov/ncidod/dvbid/westnile/surv&control.htm CDC: West Nile Virus - Statistics, Surveillance, and Control<!-- Bot generated title -->]</ref> |
|||
:In 2009, 663 cases were reported; of these, 335 were encephalitis or meningitis infections, a reaction to the virus that approximately one in 150 people who get the virus will show. Three hundred and two cases were filed for West Nile fever, the most likely symptom of the virus; 26 cases were unspecified. The state of Texas had the most cases, with 104 total. The total mortality rate for 2009 was 32 deaths of the 663 reported serious cases, a 4.5% casualty rate, but only of the severe infections. Approximately 80% of cases have no symptoms, so the total casualty rate would be less than 1% of total infections in the U.S. These and earlier years data are available from the [[Centers for Disease Control and Prevention]].<ref>[http://www.cdc.gov/ncidod/dvbid/westnile/surv&control.htm CDC: West Nile Virus - Statistics, Surveillance, and Control<!-- Bot generated title -->]</ref> |
|||
[[Image:WNVUSAMap.png|left|thumb|West Nile Virus Cases in the United States]] |
|||
:[[Dallas County, Texas]] health officials announced in the second week of August 2012 a death toll of nine so far in the county. County Judge Clay Jenkins declared a public health emergency for the county on August 9, 2012, due to the West Nile virus outbreak in the area.<ref>{{cite web|title=Dallas County declares state of emergency due to West Nile|url=http://www.wfaa.com/news/health/Dallas-County-declares-state-of-emergency-due-to-West-Nile-165675936.html|publisher=WFAA.com|accessdate=10 August 2012}}</ref><ref>{{cite web|last=Correll|first=Robin|title=Epidemic of West Nile Virus Hits Dallas County|url=http://www.healthmap.org/news/epidemic-west-nile-virus-hits-dallas-county-8912|publisher=The Disease Daily|accessdate=10 August 2012}}</ref> North Texas has been hit the hardest in 2012, with approximately 663 human cases and 18 deaths, as of August 23, 2012. During the same time, the CDC reported 1,118 human cases and 41 deaths nationwide.<ref>{{cite web|title=West Nile risk likely to run six more weeks, CDC says|url=http://www.bizjournals.com/dallas/blog/morning_call/2012/08/west-nile-risk-likely-to-run-six-more.html?ana=e_dal_rdup&s=newsletter&ed=2012-08-23|publisher=Dallas Morning News|accessdate=23 August 2012}}</ref> |
|||
'''Canada''': One human death occurred in 1999. In 2002, ten human deaths out of 416 confirmed and probable cases were reported by Canadian health officials. In 2003, 14 deaths and 1,494 confirmed and probable cases were reported. Cases were reported in 2003 in [[Nova Scotia]], [[Quebec]], [[Ontario]], [[Manitoba]], [[Saskatchewan]], [[Alberta]], [[British Columbia]], and the [[Yukon]]. In 2004, only 26 cases were reported and two deaths; however, 2005 saw 239 cases and 12 deaths. By October 28, 2006, 127 cases and no deaths had been reported. One case was asymptomatic and only discovered through a blood donation. In 2007, 445 Manitobans had confirmed cases of WNV and two people died with a third unconfirmed but suspected.<ref>[http://www.gov.mb.ca/health/wnv/index.html Province of Manitoba | Manitoba Health | West Nile virus<!-- Bot generated title -->]</ref> 17 people have either tested positive or are suspected of having the virus in Saskatchewan, and only one person has tested positive in Alberta.<ref>[http://www.mytelus.com/ncp_news/article.en.do?pn=regional/alberta&articleID=2734169 Error<!-- Bot generated title -->]</ref> |
|||
[[Saskatchewan]] has reported 826 cases of WNV plus three deaths.<ref>[http://www.ctv.ca/servlet/ArticleNews/story/CTVNews/20070824/west_nile_sask_070824/20070824?hub=Health CTV.ca | Sask. reports 339 cases of West Nile, one death]</ref> The spread of West Nile Virus infected mosquitoes to British Columbia for the first time was reported in 2009<ref>[http://www.cbc.ca/canada/british-columbia/story/2009/08/22/bc-west-nile-virus-mosquitoes.html=CBC News | West Nile virus found in BC mosquitoes]</ref> |
|||
'''Israel''': In the year 2000, the [[Centers for Disease Control and Prevention|CDC]] found that there were 417 confirmed cases with 326 hospitalizations. 33 of these people died. The main clinical presentations were encephalitis (57.9%), febrile disease (24.4%), and meningitis (15.9%).<ref>{{cite journal | first = MY | last = Chowers | coauthors = Lang R, Nassar F, Ben-David D, Giladi M, Rubinshtein E, Itzhaki A, Mishal J, Siegman-Igra Y, Kitzes R, Pick N, Landau Z, Wolf D, Bin H, Mendelson E, Pitlik SD, Weinberger M | year = 2001 | month = Jul–Aug | title = Clinical characteristics of the West Nile fever outbreak, Israel, 2000 | journal = Emerging Infectious Diseases | volume = 7 | issue = 4 | pages = 675–8 | pmid = 11585531 | url = http://www.cdc.gov/ncidod/eid/vol7no4/chowers.htm | accessdate = 2006-06-07 | doi = 10.3201/eid0704.010414 | pmc = 2631759}}</ref> |
|||
*'''Canada''': One human death occurred in 1999. In 2002, ten human deaths out of 416 confirmed and probable cases were reported by Canadian health officials. In 2003, 14 deaths and 1,494 confirmed and probable cases were reported. Cases were reported in 2003 in [[Nova Scotia]], [[Quebec]], [[Ontario]], [[Manitoba]], [[Saskatchewan]], [[Alberta]], [[British Columbia]], and the [[Yukon]]. In 2004, only 26 cases were reported and two deaths; however, 2005 saw 239 cases and 12 deaths. By October 28, 2006, 127 cases and no deaths had been reported. One case was asymptomatic and only discovered through a blood donation. In 2007, 445 Manitobans had confirmed cases of WNV and two people died with a third unconfirmed but suspected.<ref>[http://www.gov.mb.ca/health/wnv/index.html Province of Manitoba | Manitoba Health | West Nile virus<!-- Bot generated title -->]</ref> Summer of 2012 saw widespread viral penetration in Canada, coast to coast. <ref>[http://www.ctvnews.ca/video?clipId=745568]</ref> 17 people have either tested positive or are suspected of having the virus in Saskatchewan, and only one person has tested positive in Alberta.<ref>[http://www.mytelus.com/ncp_news/article.en.do?pn=regional/alberta&articleID=2734169 Error<!-- Bot generated title -->]</ref>[[Saskatchewan]] has reported 826 cases of WNV plus three deaths.<ref>[http://www.ctv.ca/servlet/ArticleNews/story/CTVNews/20070824/west_nile_sask_070824/20070824?hub=Health CTV.ca | Sask. reports 339 cases of West Nile, one death]</ref> The spread of West Nile Virus infected mosquitoes to British Columbia for the first time was reported in 2009<ref>[http://www.cbc.ca/canada/british-columbia/story/2009/08/22/bc-west-nile-virus-mosquitoes.html=CBC News | West Nile virus found in BC mosquitoes]</ref> |
|||
'''Romania''': In 1996–1997 about 500 cases occurred in Romania with a fatality rate of nearly 10%. In 2010 there were 34 confirmed cases and 3 fatalities.<ref>{{cite news|url=http://articles.cnn.com/2010-09-13/world/romania.west.nile_1_west-nile-virus-health-officials-romanian-health-ministry?_s=PM:WORLD|title=3 dead from West Nile virus in Romania|publisher=cnn.com|date=2010-09-13|accessdate=2010-12-21}}</ref> |
|||
*'''Israel''': In the year 2000, the [[Centers for Disease Control and Prevention|CDC]] found 417 confirmed cases with 326 hospitalizations; 33 of these people died. The main clinical presentations were encephalitis (57.9%), febrile disease (24.4%), and meningitis (15.9%).<ref>{{cite journal | first = MY | last = Chowers | coauthors = Lang R, Nassar F, Ben-David D, Giladi M, Rubinshtein E, Itzhaki A, Mishal J, Siegman-Igra Y, Kitzes R, Pick N, Landau Z, Wolf D, Bin H, Mendelson E, Pitlik SD, Weinberger M | year = 2001 | month = Jul–Aug | title = Clinical characteristics of the West Nile fever outbreak, Israel, 2000 | journal = Emerging Infectious Diseases | volume = 7 | issue = 4 | pages = 675–8 | pmid = 11585531 | url = http://www.cdc.gov/ncidod/eid/vol7no4/chowers.htm | accessdate = 2006-06-07 | doi = 10.3201/eid0704.010414 | pmc = 2631759}}</ref> |
|||
'''Greece''': In the summer of 2010 several cases were reported in [[northern Greece]]. In total there were 261 diagnosed cases and 34 fatalities.<ref>{{cite web|url=http://www.keel.org.gr/images/stories/keelpno/ios_ditikou_neilou/wnv_weekly_report_2010_11_12_english.pdf|title=Weekly Epidemiological Surveillance Report of cases of West Nile virus infection in Greece 12 November 2010|publisher=Hellenic Centre for Disease Control and Prevention (KEELPNO)|date=2010-11-12|accessdate=2010-12-21}}</ref> |
|||
*'''Romania''': In 1996–1997, about 500 cases occurred in Romania with a fatality rate of nearly 10%. In 2010, 34 cases were confirmed, with 3 fatalities.<ref>{{cite news|url=http://articles.cnn.com/2010-09-13/world/romania.west.nile_1_west-nile-virus-health-officials-romanian-health-ministry?_s=PM:WORLD|title=3 dead from West Nile virus in Romania|publisher=cnn.com|date=2010-09-13|accessdate=2010-12-21}}</ref> |
|||
==Surveillance methods== |
|||
West Nile virus can be sampled from the environment by the pooling of trapped mosquitoes, testing avian blood samples drawn from wild birds and dogs and sentinel monkeys, as well as testing brains of dead birds found by various animal control agencies and the public. Testing of the mosquito samples requires the use of [[RT-PCR]] to directly amplify and show the presence of virus in the submitted samples. When using the blood sera of wild bird and sentinel chickens, samples must be tested for the presence of WNV [[antibodies]] by use of [[immunohistochemistry]] (IHC)<ref>{{cite journal | first = M | last = Jozan | coauthors = Evans R, McLean R, Hall R, Tangredi B, Reed L, Scott J | year = 2003 | month = Fall | title = Detection of West Nile virus infection in birds in the United States by blocking ELISA and immunohistochemistry | journal = Vector-borne and Zoonotic Diseases | volume = 3 | issue = 3 | pages = 99–110 | pmid = 14511579 | doi = 10.1089/153036603768395799}}</ref> or Enzyme-Linked Immunosorbent Assay (ELISA).<ref>{{cite journal | first = RA | last = Hall | coauthors = Broom AK, Hartnett AC, Howard MJ, Mackenzie JS | year = 1995 | month = February | title = Immunodominant epitopes on the NS1 protein of MVE and KUN viruses serve as targets for a blocking ELISA to detect virus-specific antibodies in sentinel animal serum | journal = Journal of Virological Methods | volume = 51 | issue = 2–3 | pages = 201–10 | pmid = 7738140 | doi = 10.1016/0166-0934(94)00105-P}}</ref> |
|||
*'''Greece''': In the summer of 2010, several cases were reported mainly in [[northern Greece]], a countrywide total of 262 diagnosed cases and 35 fatalities.<ref>{{cite web|url=http://www.keelpno.gr/Portals/0/%CE%91%CF%81%CF%87%CE%B5%CE%AF%CE%B1/%CE%99%CF%8C%CF%82%20%CE%94%CF%85%CF%84%CE%B9%CE%BA%CE%BF%CF%8D%20%CE%9D%CE%B5%CE%AF%CE%BB%CE%BF%CF%85/ekthesi_epidimias_2010_revised.pdf|title=Έκθεση: Επιδημία Λοίμωξης από τον Ιό του Δυτικού Νείλου, 2010|publisher=Hellenic Centre for Disease Control and Prevention (KEELPNO)|year=2011|accessdate=2012-08-09|language=Greek}}</ref> In 2011 the virus spread to [[central Greece]] but with fewer cases, 101 diagnosed and 9 fatalities.<ref>{{cite web|url=http://www.keelpno.gr/Portals/0/%CE%91%CF%81%CF%87%CE%B5%CE%AF%CE%B1/%CE%99%CF%8C%CF%82%20%CE%94%CF%85%CF%84%CE%B9%CE%BA%CE%BF%CF%8D%20%CE%9D%CE%B5%CE%AF%CE%BB%CE%BF%CF%85/Report%202011_WNV_GR_final_07-5-2012.pdf|title=Έκθεση επιδημιολογικής επιτήρησης - Έτος 2011|publisher=Hellenic Centre for Disease Control and Prevention (KEELPNO)|date=2012-05-07|accessdate=2012-07-10}}</ref>. In 2012, 44 were diagnosed and 3 died.<ref>{{cite news|url=http://news.in.gr/greece/article/?aid=1231208673|title=Και τρίτος νεκρός από τον ιό του Δυτικού Νείλου|publisher=in.gr|date=2012-08-08|accessdate=2012-08-08|language=Greek}}</ref> The total number of people in Greece infected by the virus is estimated to 1,800.<ref>{{cite news|title=1800 άνθρωποι μολύνθηκαν από τον ιό του Δυτικού Νείλου στην Ελλάδα|url=http://www.sigmalive.com/news/greece/509232|date=2012-07-24|accessdate=2012-07-24|language=Greek|publisher=SigmaLive/news24.7}}</ref> |
|||
Dead birds, after [[necropsy]], have their various tissues tested for virus by either [[RT-PCR]] or immunohistochemistry, where virus shows up as brown stained tissue because of a substrate-[[enzyme]] reaction. |
|||
==Monitoring and control== |
|||
==Control== |
|||
[[File:CDC-LightTrap 200.jpg|thumb|left|A [[carbon dioxide]]-baited CDC [[light trap]] at [[National Park Service|NPS]] monitoring site. The highest individual light trap total for 2010 was from a trap located in a [[salt marsh]] in the [[Fire Island National Seashore]]: approximately 25,142 mosquitoes were collected during a 16-hour period on August 31.<ref>[http://www.nps.gov/fiis/parkmgmt/mosquito-management.htm [[National Park Service: Mosquito Management]</ref>]] |
|||
[[Image:West Nile Virus Warning -Sign.jpg|thumb|150px|right|West Nile virus warning sign in Southern California.]] |
|||
{{multiple image |
|||
West Nile control is achieved through [[mosquito control]], by elimination of mosquito breeding sites, larviciding active breeding areas and encouraging personal use of [[mosquito repellent]]s. The public is also encouraged to spend less time outdoors, wear long covering clothing, apply bug repellant that contains [[DEET]] and ensure that mosquitoes cannot enter buildings.<ref> |
|||
| align = right |
|||
{{cite web |
|||
| direction = horizontal |
|||
|url = http://www.osha.gov/dts/shib/shib082903b.html |
|||
| image1 = Culex sp larvae.png |
|||
|title = Workplace Precautions Against West Nile Virus |
|||
| width1 = 220 |
|||
|accessdate = 2007-11-21 |
|||
| caption1 = [[Insect egg|Eggs]] of permanent water [[mosquitos]] can hatch, and the [[larvae]] survive, in only a few ounces of water. Less than half the amount that may collect in a discarded coffee cup. Floodwater species lay their eggs on wet soil or other moist surfaces. Hatch time is variable for both types, under favorable circumstances, [[id est|i.e.]] warm weather; the eggs of some [[species]] may hatch in as little as 1-3 days after [[Oviposition|being laid]].<ref>[[Oklahoma State University]]: Mosquitos and West Nile virus</ref> |
|||
|work = Safety and Health Information Bulletins (SHIBs) |
|||
| image2 = Used tires.jpg |
|||
|publisher = U. S. Department of Labor Occupational Safety and Health Administration |
|||
| width2 = 200 |
|||
| caption2 = Used [[tires]] often hold [[stagnant water]] and are a breeding ground for many [[species]] of mosquitos. Some species such as the [[Aedes albopictus|Asian Tiger mosquito]] prefer manmade containers; such as tires, to lay their eggs in. The rapid spread of this aggressive daytime feeding species [[Invasive species|beyond their native range]] has been attributed to the used tire trade.<ref>University of Florida: Asian Tiger Mosquito, Aedes albopictus (Skuse) (Insecta: Diptera: Culicidae)</ref> |
|||
}} |
}} |
||
</ref> |
|||
[[Environmentalist]]s have condemned attempts to control the transmitting mosquitoes by spraying [[pesticide]], saying that the detrimental health effects of spraying outweigh the relatively few lives which may be saved, and that there are more environmentally friendly ways of controlling mosquitoes. They also question the effectiveness of insecticide spraying, as they believe mosquitoes that are resting or flying above the level of spraying will not be killed; the most common vector in the northeastern U.S., ''Culex pipiens'', is a [[Canopy (forest)|canopy]] feeder. |
|||
West Nile virus can be sampled from the environment by the pooling of trapped mosquitoes, testing avian blood samples drawn from wild birds, dogs and sentinel monkeys, as well as testing brains of dead birds found by various animal control agencies and the public. Testing of the mosquito samples requires the use of [[RT-PCR]] to directly amplify and show the presence of virus in the submitted samples. When using the blood sera of wild birds and sentinel chickens, samples must be tested for the presence of WNV [[antibodies]] by use of [[immunohistochemistry]] (IHC)<ref>{{cite journal | first = M | last = Jozan | coauthors = Evans R, McLean R, Hall R, Tangredi B, Reed L, Scott J | year = 2003 | month = Fall | title = Detection of West Nile virus infection in birds in the United States by blocking ELISA and immunohistochemistry | journal = Vector-borne and Zoonotic Diseases | volume = 3 | issue = 3 | pages = 99–110 | pmid = 14511579 | doi = 10.1089/153036603768395799}}</ref> or Enzyme-Linked Immunosorbent Assay (ELISA).<ref>{{cite journal | first = RA | last = Hall | coauthors = Broom AK, Hartnett AC, Howard MJ, Mackenzie JS | year = 1995 | month = February | title = Immunodominant epitopes on the NS1 protein of MVE and KUN viruses serve as targets for a blocking ELISA to detect virus-specific antibodies in sentinel animal serum | journal = Journal of Virological Methods | volume = 51 | issue = 2–3 | pages = 201–10 | pmid = 7738140 | doi = 10.1016/0166-0934(94)00105-P}}</ref> |
|||
The first effective horse vaccine, West Nile-INNOVATOR was introduced by Fort Dodge Animal Health ([[Wyeth]]). Shortly thereafter, a second, one-annual-dose vaccine called Prevenile was introduced by Intervet/Schering-Plough Animal Health ([[Merck & Co.|Merck]]),<ref>{{cite journal|url=http://www.thehorse.com/pdf/factsheets/west-nile-virus/wnv.pdf| title=West Nile Virus |author= Fact sheet|publisher=The Horse |date = 2009-11-23|DUPLICATE DATA: publisher=Blood-Horse Publications|accessdate=2010-02-10}}</ref> followed by a DNA-based vaccine, called Recombitek ([[Merial]]). In 2009, a new killed virus vaccine was introduced by Boehringer-Ingelheim, a privately held pharmaceutical company, incorporating an equine origin WNV strain (E159), representative of the more recent WNV strains impacting horses.<ref>{{cite journal|url=http://www.thehorse.com/ViewArticle.aspx?ID=15334| title=New West Nile Virus Vaccines for Horses Approved|author=Brown, Kimberly S.|publisher=The Horse |date = 2009-11-23|accessdate=2010-02-10}}</ref> |
|||
Dead birds, after [[necropsy]], have their various tissues tested for virus by either [[RT-PCR]] or IHC, where virus shows up as brown-stained tissue because of a substrate-[[enzyme]] reaction. |
|||
West Nile control is achieved through [[mosquito control]], by elimination of mosquito breeding sites, larviciding active breeding areas. |
|||
[[Environmentalist]]s have condemned attempts to control the transmitting mosquitoes by spraying [[pesticide]], saying the detrimental health effects of spraying outweigh the relatively few lives which may be saved, and more environmentally friendly ways of controlling mosquitoes are available. They also question the effectiveness of insecticide spraying, as they believe mosquitoes that are resting or flying above the level of spraying will not be killed; the most common vector in the northeastern US, ''Culex pipiens'', is a [[Canopy (forest)|canopy]] feeder. |
|||
==Prevention== |
|||
{{multiple image |
|||
| align = right |
|||
| direction = horizontal |
|||
| image1 = Mosquito Netting.jpg |
|||
| width1 = 180 |
|||
| caption1 = Low-cost, ceiling hung mosquito netting for a bed. |
|||
| image2 = Bedroom of Canopy Tower in Gamboa, Panama 01.jpg |
|||
| width2 = 180 |
|||
| caption2 = Ceiling hung mosquito netting when not in use. |
|||
}} |
|||
Personal protective measures can be taken to greatly reduce the risk of being bitten by an infected mosquito: |
|||
*Using insect repellent on exposed skin to repel mosquitoes. [[Environmental Protection Agency|EPA-registered]] repellents include products containing [[DEET]] (N,N-diethylmetatoluamide) and [[picaridin]] (KBR 3023). DEET concentrations of 30% to 50% are effective for several hours. Picaridin, available at 7% and 15 % concentrations, needs more frequent application. DEET formulations as high as 50% are recommended for both adults and children over 2 months of age. Protect infants less than 2 months of age by using a carrier draped with [[mosquito netting]] with an elastic edge for a tight fit. |
|||
*When using [[sunscreen]], apply sunscreen first and then repellent. Repellent should be washed off at the end of the day before going to bed. |
|||
*Wear long-sleeved shirts which should be tucked in, long pants, and hats to cover exposed skin. |
|||
*The application of [[permethrin]]-containing (e.g., Permanone) or other insect repellents to clothing, shoes, tents, mosquito nets, and other gear for greater protection. Permethrin is not labeled for use directly on skin. Most repellent is generally removed from clothing and gear by a single washing, but permethrin-treated clothing is effective for up to 5 washings. |
|||
*Be aware that most mosquitoes that transmit disease are most active during twilight periods (dawn and dusk or in the evening). A notable exception is the [[Asian Tiger mosquito]] which is a daytime feeder and is more apt to be found in, or on the periphery of, shaded areas with heavy vegetation. |
|||
*Staying in air-conditioned or well-[[Window screen|screened]] housing, and/ or sleeping under an insecticide treated [[Mosquito net|bed net. Bed nets should be tucked under mattresses and can be sprayed with a repellent if not already treated with an insecticide. |
|||
{{clear}} |
|||
==Treatment research== |
==Treatment research== |
||
WNV is one of the [[Japanese encephalitis]] antigenic [[Serotype|serocomplex]] of viruses. |
|||
Image reconstructions and [[cryoelectron microscopy]] reveal a 45–50 nm [[virion]] covered with a relatively smooth [[protein]] surface. This structure is similar to the [[dengue fever]] virus; both belong to the genus ''[[Flavivirus]]'' within the family ''[[Flaviviridae]]''. The genetic material of WNV is a [[Sense (molecular biology)|positive-sense]], single strand of [[RNA]], which is between 11,000 and 12,000 [[nucleotide]]s long; these [[genes]] encode seven nonstructural proteins and three structural proteins. The RNA strand is held within a nucleocapsid formed from 12-[[kDa]] protein blocks; the capsid is contained within a host-derived [[Biological membrane|membrane]] altered by two viral [[glycoprotein]]s. |
|||
[[AMD3100]], which had been proposed as an antiretroviral drug for HIV, has shown promise against West Nile encephalitis. [[Morpholino]] antisense oligos conjugated to [[cell penetrating peptide]]s have been shown to partially protect mice from WNV disease.<ref>{{cite journal | first = Tia S | last = Deas | coauthors = Bennett CJ, Jones SA, Tilgner M, Ren P, Behr MJ, Stein DA, Iversen PL, Kramer LD, Bernard KA, Shi PY | year = 2007 | month = May | title = In vitro resistance selection and in vivo efficacy of morpholino oligomers against West Nile virus | journal = Antimicrob Agents Chemother | pmid = 17485503 | doi = 10.1128/AAC.00069-07 | volume = 51 | pages = 2470–82 | issue = 7 | pmc = 1913242}}</ref> There have also been attempts to treat infections using [[ribavirin]], intravenous [[immunoglobulin]], or [[alpha interferon]].<ref>{{cite journal |author=Hayes EB, Sejvar JJ, Zaki SR, Lanciotti RS, Bode AV, Campbell GL |title=Virology, pathology, and clinical manifestations of West Nile virus disease |journal=Emerging Infect. Dis. |volume=11 |issue=8 |pages=1174–9 |year=2005 |pmid=16102303 |doi= |url=http://www.cdc.gov/ncidod/EID/vol11no08/05-0289b.htm |pmc=3320472}}</ref> GenoMed, a U.S. biotech company, has found that blocking angiotensin II can treat the "[[cytokine storm]]" of West Nile virus encephalitis as well as other viruses.<ref>{{cite journal |author=Moskowitz DW, Johnson FE |title=The central role of angiotensin I-converting enzyme in vertebrate pathophysiology |journal=Curr Top Med Chem |volume=4 |issue=13 |pages=1433–54 |year=2004 |pmid=15379656 |doi=10.2174/1568026043387818 }}</ref> |
[[AMD3100]], which had been proposed as an antiretroviral drug for HIV, has shown promise against West Nile encephalitis. [[Morpholino]] antisense oligos conjugated to [[cell penetrating peptide]]s have been shown to partially protect mice from WNV disease.<ref>{{cite journal | first = Tia S | last = Deas | coauthors = Bennett CJ, Jones SA, Tilgner M, Ren P, Behr MJ, Stein DA, Iversen PL, Kramer LD, Bernard KA, Shi PY | year = 2007 | month = May | title = In vitro resistance selection and in vivo efficacy of morpholino oligomers against West Nile virus | journal = Antimicrob Agents Chemother | pmid = 17485503 | doi = 10.1128/AAC.00069-07 | volume = 51 | pages = 2470–82 | issue = 7 | pmc = 1913242}}</ref> There have also been attempts to treat infections using [[ribavirin]], intravenous [[immunoglobulin]], or [[alpha interferon]].<ref>{{cite journal |author=Hayes EB, Sejvar JJ, Zaki SR, Lanciotti RS, Bode AV, Campbell GL |title=Virology, pathology, and clinical manifestations of West Nile virus disease |journal=Emerging Infect. Dis. |volume=11 |issue=8 |pages=1174–9 |year=2005 |pmid=16102303 |doi= |url=http://www.cdc.gov/ncidod/EID/vol11no08/05-0289b.htm |pmc=3320472}}</ref> GenoMed, a U.S. biotech company, has found that blocking angiotensin II can treat the "[[cytokine storm]]" of West Nile virus encephalitis as well as other viruses.<ref>{{cite journal |author=Moskowitz DW, Johnson FE |title=The central role of angiotensin I-converting enzyme in vertebrate pathophysiology |journal=Curr Top Med Chem |volume=4 |issue=13 |pages=1433–54 |year=2004 |pmid=15379656 |doi=10.2174/1568026043387818 }}</ref> |
||
In 2007 the [[World Community Grid]] launched the [[Discovering Dengue Drugs – Together]] project. This uses a distributed network of volunteers' computers via the [[Berkeley Open Infrastructure for Network Computing]] |
In 2007, the [[World Community Grid]] launched the [[Discovering Dengue Drugs – Together]] project. This uses a distributed network of volunteers' computers via the [[Berkeley Open Infrastructure for Network Computing]] to perform computer simulations of interacting molecules. Thousands of small molecules are screened for potential antiviral properties with respect to West Nile and related viruses. |
||
==See also== |
==See also== |
||
*[[West Nile virus in the United States]] |
|||
*[[CCR5]] |
*[[CCR5]] |
||
*''[[Psorophora howardii]]'' |
*''[[Psorophora howardii]]'' |
||
| Line 135: | Line 191: | ||
==External links== |
==External links== |
||
{{Commons|West Nile virus}} |
{{Commons|West Nile virus}} |
||
* [http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3311072/ Recent progress in West Nile virus diagnosis and vaccination] |
|||
* [http://www.encephalitis.info The Encephalitis Society] - a comprehensive resource on all aspects of Encephalitis and a source of support for those affected and their families. |
|||
* [http://www.cdc.gov/ncidod/dvbid/westnile/ West Nile Virus] - U.S. [[Centers for Disease Control and Prevention]] (CDC) page |
* [http://www.cdc.gov/ncidod/dvbid/westnile/ West Nile Virus] - U.S. [[Centers for Disease Control and Prevention]] (CDC) page |
||
* [http://www.cdc.gov/niosh/docs/2006-115/ Recommendations for Protecting Laboratory, Field, and Clinical Workers from West Nile Virus Exposure] |
* [http://www.cdc.gov/niosh/docs/2006-115/ Recommendations for Protecting Laboratory, Field, and Clinical Workers from West Nile Virus Exposure] |
||
| Line 145: | Line 201: | ||
* [http://www.viprbrc.org/brc/home.do?decorator=flavi Virus Pathogen Database and Analysis Resource (ViPR): Flaviviridae] |
* [http://www.viprbrc.org/brc/home.do?decorator=flavi Virus Pathogen Database and Analysis Resource (ViPR): Flaviviridae] |
||
* [http://www.invasivespeciesinfo.gov/microbes/westnile.shtml Species Profile- West Nile Virus (''Flavivirus'')], National Invasive Species Information Center, [[United States National Agricultural Library]]. Lists general information and resources for West Nile Virus. |
* [http://www.invasivespeciesinfo.gov/microbes/westnile.shtml Species Profile- West Nile Virus (''Flavivirus'')], National Invasive Species Information Center, [[United States National Agricultural Library]]. Lists general information and resources for West Nile Virus. |
||
*[http://www.pdbe.org/emsearch/west%20nile%20virus 3D macromolecular structures of the West Nile Virus archived in the EM Data Bank(EMDB)] |
|||
* [http://rad.usuhs.edu/medpix/parent.php3?mode=TFcase_thumbnails&case_prog=/medpix/cow_image.html&pt_id=14187&topic_id=4124&imageid=58164&quiz=no#top West Nile Virus] - West Nile Encephalitis Brain Scans |
|||
{{Zoonotic viral diseases}} |
{{Zoonotic viral diseases}} |
||
| Line 167: | Line 225: | ||
[[et:Lääne-Niiluse viirus]] |
[[et:Lääne-Niiluse viirus]] |
||
[[es:Virus del Nilo Occidental]] |
[[es:Virus del Nilo Occidental]] |
||
[[eo:Okcidentnila viruso]] |
|||
[[fr:Virus du Nil occidental]] |
[[fr:Virus du Nil occidental]] |
||
[[id:Virus West Nile]] |
[[id:Virus West Nile]] |
||
| Line 174: | Line 233: | ||
[[nl:West-Nijlvirus]] |
[[nl:West-Nijlvirus]] |
||
[[ja:ウエストナイル熱]] |
[[ja:ウエストナイル熱]] |
||
[[no:Vestnilfeber]] |
|||
[[pl:Gorączka Zachodniego Nilu]] |
[[pl:Gorączka Zachodniego Nilu]] |
||
[[pt:Vírus do oeste do Nilo]] |
[[pt:Vírus do oeste do Nilo]] |
||
| Line 180: | Line 240: | ||
[[simple:West Nile virus]] |
[[simple:West Nile virus]] |
||
[[sk:Západonílsky vírus]] |
[[sk:Západonílsky vírus]] |
||
[[sr:Вирус Западног Нила]] |
|||
[[sv:West Nile-feber]] |
[[sv:West Nile-feber]] |
||
[[tr:Batı Nil virüsü]] |
[[tr:Batı Nil virüsü]] |
||
Revision as of 08:45, 5 September 2012
| West Nile Virus | |
|---|---|

| |
| Virus classification | |
| Group: | Group IV ((+)ssRNA)
|
| Family: | |
| Genus: | |
| Species: | West Nile virus
|
| West Nile fever | |
|---|---|
| Specialty | Infectious diseases, neurology |
West Nile virus (WNV) is a mosquito-borne zoonotic arbovirus belonging to the genus Flavivirus in the family Flaviviridae. This flavivirus is found in temperate and tropical regions of the world. It was first identified in the West Nile sub-region in the East African nation of Uganda in 1937. Prior to the mid 1990's, WNV disease occurred only sporadically and was considered a minor risk for humans, until an outbreak in Algeria in 1994 with cases of WNV caused encephalitis, and the first large outbreak in Romania in 1996, with a high number of cases with neuroinvasive disease, WNV has now spread globally, with the first case in the Western Hemisphere being identified in New York City in 1999[1], over the next 5 years, the virus spread across the continental United States, north into Canada, and southward into the Caribbean Islands and Latin America. WNV also spread to Europe, beyond the Mediterranean Basin (a new strain of the virus was recently (2012) identified in Italy). WNV is now considered to be an endemic pathogen in Africa, Asia, Australia, the Middle East, Europe and in the United States which in 2012 has experienced one of its worst epidemics.
The main mode of WNV transmission is via various species of mosquitos which are the prime vector, with birds being the most commonly infected animal and serving as the prime reservoir host - especially passerines which are of the largest order (Passeriformes) of birds. WNV has been found in various species of ticks, but current research suggests they are not important vectors of the virus. WNV also infects various mammal species including humans and has been identified in reptilian species, including alligators and crocodiles, and also in amphibians.Not all animal species which are susceptible to WNV infection - humans included, and not all bird species - develop sufficient viral levels to transmit the disease to noninfected mosquitoes, and are thus not considered major factors in WNV transmission.[2][3]
Approximately 80 percent of West Nile virus infections in humans are subclinical, which cause no symptoms.[4]In the cases where symptoms do occur - termed West Nile Fever in cases without neurological disease - the time from infection to the appearance of symptoms (incubation period) is typically between 2-15 days. Symptoms may include fever, headaches, fatigue, muscle pain or aches, malaise, nausea, anorexia, vomiting, myalgias and rash. Less than 1% of the cases are severe and result in neurological disease when the central nervous system is affected. People of advanced age, the very young, or those with immunosuppression, either from medical induced such as those taking immunosupressive drugs or due to a preexisting medical condition such as HIV infection, are most susceptible. The specific neurological diseases which may occur are, West Nile encephalitis which causes inflammation of the brain, West Nile meningitis which causes inflammation of the meninges, which are the protective membranes that cover the brain and spinal cord, West Nile meningoencephalitis which causes inflammation of the brain and also the meninges surrounding it, and West Nile Poliomyelitis - spinal cord inflammation which results in a syndrome similar to polio, which may cause acute flaccid paralysis.
There is currently no vaccine against WNV infection. The best methods to reduce the rates of WNV infection are mosquito control on the part of municipalities, businesses and individual citizens to reduce breeding populations of mosquitoes in public, commercial and private areas via various means including eliminating standing pools of water where mosquitos breed, such as in old tires, buckets, disused swimming pools, etc. On an individual basis, the use of personal protective measures to avoid being bitten by an infected mosquito, via the use of mosquito repellent, window screens, avoiding areas where mosquitos are more prone to congregate, such as near marshes, areas with heavy vegetation etc., and being more vigilant from dusk to dawn when mosquitoes are most active offers the best defense. In the advent of being bitten by an infected mosquito, familiarity of the symptoms of WNV on the part of laypersons, physicians and allied health professionals affords the best chance of receiving timely medical treatment which may aid in reducing associated possible complications and also appropriate palliative care.
Virus transmission
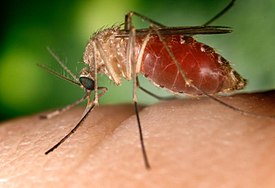
The virus is transmitted through mosquito vectors, which bite and infect birds. The birds are amplifying hosts, developing sufficient viral levels to transmit the infection to other biting mosquitoes which go on to infect other birds (in the Western Hemisphere, the American robin and the American crow are the most common carriers) and also humans. The infected mosquito species vary according to geographical area; in the US, Culex pipiens (Eastern US), Culex tarsalis (Midwest and West), and Culex quinquefasciatus (Southeast) are the main sources.[5]
In mammals, the virus does not multiply as readily (i.e. does not develop high viremia during infection), and mosquitoes biting infected mammals are not believed to ingest sufficient virus to become infected,[6] making mammals so-called dead-end infections.
A 2004 paper in Science reported that Culex pipiens mosquitoes existed in two populations in Europe, one that bites birds and one that bites humans. In North America, 40% of C. pipiens mosquitoes were found to be hybrids of the two types which bite both birds and humans, providing a vector for WNV. This is argued to provide an explanation of why the West Nile disease has spread more quickly in North America than Europe.[7] However, these conclusions have been disputed.[8] In 2010, the Greek Center for Disease Control and Prevention verified that C. pipiens was responsible for an outbreak of the virus in northern Greece.[9]
Direct human-to-human transmission initially was believed to be caused only by occupational exposure,[10] or conjunctive exposure to infected blood.[11] The US outbreak identified additional transmission methods through blood transfusion,[12] organ transplant,[13] intrauterine exposure,[14] and breast feeding.[15] Since 2003, blood banks in the US routinely screen for the virus among their donors.[16] As a precautionary measure, the UK's National Blood Service initially ran a test for this disease in donors who donate within 28 days of a visit to the United States, Canada or the northeastern provinces of Italy and the Scottish National Blood Transfusion Service[17] asks prospective donors to wait 28 days after returning from North America or the northeastern provinces of Italy before donating.
The more severe outcomes of WNV infection are clearly associated with advancing age[18] and a patient history of organ transplantation[19] and diabetes[citation needed]. A genetic factor also appears to increase susceptibility to West Nile disease. A mutation of the gene CCR5 gives some protection against HIV but leads to more serious complications of WNV infection. Carriers of two mutated copies of CCR5 made up 4.0 to 4.5% of a sample of West Nile disease sufferers, while the incidence of the gene in the general population is only 1.0%.[20][21]
Recently, the potential for mosquito saliva to impact the course of WNV disease was demonstrated.[22][23][24] Mosquitoes inoculate their saliva into the skin while obtaining blood. Mosquito saliva is a pharmacological cocktail of secreted molecules, principally proteins, that can affect vascular constriction, blood coagulation, platelet aggregation, inflammation, and immunity. It clearly alters the immune response in a manner that may be advantageous to a virus.[25][26][27][28] Studies have shown it can specifically modulate the immune response during early virus infection,[29] and mosquito feeding can exacerbate WNV infection, leading to higher viremia and more severe forms of disease.[22][23][24] It is unknown what benefit, if any, the mosquito receives by assisting the virus in this manner, so it is likely the virus is simply exploiting the preexisting qualities of mosquito saliva developed for other purposes.[according to whom?]
There is no vaccine for humans. A vaccine for horses (ATCvet code: QI05AA10 (WHO)) based on killed viruses exists; some zoos have given this vaccine to their birds, although its effectiveness is unknown. Dogs and cats show few if any signs of infection. There have been no known cases of direct canine-human or feline-human transmission; although these pets can become infected, it is unlikely they are, in turn, capable of infecting native mosquitoes and thus continuing the disease cycle.[30]
Avoiding mosquito bites is the most straightforward means to avoid infection;[31] remaining indoors (while preventing mosquitoes from entering) at dawn and dusk, wearing light-colored clothing that covers arms and legs, as well as the trunk, and using insect repellents on both skin and clothing (such as DEET, picaradin, or oil of lemon eucalyptus for skin and permethrin for clothes) are effective.[32] If one becomes infected, generally, treatment is purely supportive: analgesia for the pain of neurological diseases, and rehydration for nausea, vomiting, or diarrhea; encephalitis may also require airway protection and seizure management.
Reported cases in the U.S. in 2005 exceeded those in 2004, and cases in 2006 exceeded 2005's totals. On August 19, 2006, the LA Times reported the expected incidence rate of WNV was dropping as the local population becomes exposed to the virus. "In countries like Egypt and Uganda, where West Nile was first detected, people became fully immune to the virus by the time they reached adulthood", federal health officials said.[33] However, just days later, the CDC said WNV cases could reach a three-year high because hot temperatures had allowed a larger brood of mosquitoes.[34] Although currently no West Nile virus vaccine is available for humans, many scientists are working on this issue, and hopefully a vaccine will become available in the next few years.[35]
Symptoms
The incubation period for WNV - the amount of time from infection to symptom onset - is typically from between 2-15 days. Multiple somatic complaints, tremor, and abnormalities in motor skills and executive functions are common long-term problems among patients who have had West Nile virus infection. Patients with milder illness are just as likely as patients with more-severe illness to experience adverse outcomes.
- West Nile Fever (WNF), which occurs in 20 percent of cases, is a febrile syndrome which causes flu-like symptoms.[39]The incubation period - the amount of time from infection to symptom onset is typically from between 2-15 days. Most characterizations of WNF generally describe it as a mild, acute syndrome lasting 3 - 6 days after symptom onset. Systematic follow-up studies of patients with WNF had not previously been done, so this information is largely anecdotal. In addition to a high fever, headache, chills, excessive sweating, weakness, fatigue, swollen lymph nodes, drowsiness, pain in the joints and symptoms like those of influenza. Occasionally, some patients experience a short-lived truncal rash or gastrointestinal symptoms, including nausea, vomiting, loss of appetite, or diarrhea.
- West Nile Neuroinvasive Disease (WNND), which occurs in less than 1 percent of cases, is when the virus infects the central nervous system resulting in meningitis, encephalitis, meningoencephalitis or poliomyelitis.[40][medical citation needed]
- West Nile virus encephalitis (WNVE) is characterized by similar early symptoms, but also a decreased level of consciousness, sometimes approaching near-coma. Deep tendon reflexes are hyperactive at first, later diminished. There are also extrapyramidal symptoms. Recovery is marked by a long convalescence with fatigue. One study found that neuroinvasive WNV infection was associated with an increased risk for subsequent kidney disease.[41][42]
- More recent outbreaks have resulted in a deeper study of the disease and other, rarer, outcomes have been identified. The spinal cord may be infected, marked by anterior myelitis with or without encephalitis.[43] WNV-associated Guillain-Barré syndrome has been identified[44] and other rare effects include multifocal chorioretinitis (which has 100% specificity for identifying WNV infection in patients with possible WNV encephalitis),[45] hepatitis, myocarditis, nephritis, pancreatitis, and splenomegaly.[46][47][48]
Diagnosis

Preliminary diagnosis is often based on the patient's clinical symptoms, places and dates of travel (if patient is from a non-endemic country or area), activities, and epidemiologic history of the location where infection occurred. A recent history of mosquito bites and an acute febrile illness associated with neurologic signs and symptoms should cause clinical suspicion of WNV. Diagnosis of West Nile Virus infections is generally accomplished by serologic testing of blood serum or cerebrospinal fluid (CSF), which is obtained via a lumbar puncture. Typical findings of WNV infection include lymphocytic pleocytosis, elevated protein level, reference glucose and lactic acid levels, and no erythrocytes. Definitive diagnosis is of WNV is obtained through detection of virus-specific antibody Immunoglobulin M, (IgM) and neutralizing antibodies. Cases of West Nile virus meningitis and encephalitis which have been serologically confirmed produce similar degrees of CSF pleocytosis and are often associated with substantial CSF neutrophilia.[49]
Four FDA-cleared WNV IgM ELISA kits from different manufacturers are commercially available in the U.S. According to the package inserts, each of these kits is indicated for use on serum to aid in the presumptive laboratory diagnosis of WNV infection in patients with clinical symptoms of meningitis or encephalitis. The package inserts also state that all positive results obtained with any of the commercially-available WNV test kits should be confirmed by additional testing at a state health department laboratory or CDC.
In fatal cases, nucleic acid amplification, histopathology with immunohistochemistry and virus culture of autopsy tissues can also be useful. Only a few state laboratories or other specialized laboratories, including those at CDC, are capable of doing this specialized testing
Prognosis
While the general prognosis is favorable, current studies indicate that West Nile Fever can often be more severe than previously recognized, with studies of various recent outbreaks indicating that it may take as long as 60 -90 days to recover.[50][51]Patients with milder WNF are just as likely as those with more severe manifestations of neuroinvasive disease to experience multiple long term (>1+ years) somatic complaints such as tremor, and dysfunction in motor skills and executive functions.[52]
History
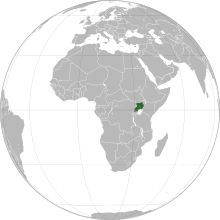
Studies of phylogenetic lineages determined WNV emerged as a distinct virus around 1000 years ago.[53] This initial virus developed into two distinct lineages, lineage 1 and its multiple profiles is the source of the epidemic transmission in Africa and throughout the world. Lineage 2 was considered an Africa zoonose. However, in 2008, lineage 2, previously only seen in horses in sub-Saharan Africa and Madagascar, began to appear in horses in Europe, where the first known outbreak affected 18 animals in Hungary in 2008.[54] Lineage 1 West Nile virus was detected in South Africa in 2010 in a mare and her aborted fetus; previously, only lineage 2 West Nile virus had been detected in horses and humans in South Africa.[55] A 2007 fatal case in a killer whale in Texas broadened the known host range of West Nile virus to include cetaceans.[56]
WNV has been posited as one of the possible causes of Alexander the Great's early death based on reports of avian deaths before his illness period.[57]
WNV was first isolated from a feverish 37-year-old woman at Omogo in the West Nile District of Uganda in 1937 during research on yellow fever virus.[58] A series of serosurveys in 1939 in central Africa found anti-WNV positive results ranging from 1.4% (Congo) to 46.4% (White Nile region, Sudan). It was subsequently identified in Egypt (1942) and India (1953), a 1950 serosurvey in Egypt found 90% of those over 40 years in age had WNV antibodies. The ecology was characterized in 1953 with studies in Egypt[59] and Israel.[60] The virus became recognized as a cause of severe human meningoencephalitis in elderly patients during an outbreak in Israel in 1957. The disease was first noted in horses in Egypt and France in the early 1960s and found to be widespread in southern Europe, southwest Asia and Australia.
The first appearance of WNV in the Western Hemisphere was in 1999[61] with encephalitis reported in humans, dogs, cats, and horses, and the subsequent spread in the United States may be an important milestone in the evolving history of this virus. The American outbreak began in the New York City area (specifically, College Point, Queens) and was later seen in New Jersey and Connecticut; the virus is believed to have entered in an infected bird or mosquito, although there is no clear evidence.[62]
The US virus was very closely related to a lineage 1 strain found in Israel in 1998. Since the first North American cases in 1999, the virus has been reported throughout the United States, Canada, Mexico, the Caribbean and Central America. There have been human cases and equine cases, and many birds are infected. The Barbary macaque, Macaca sylvanus, was the first nonhuman primate to contract WNV.[63] Both the US and Israeli strains are marked by high mortality rates in infected avian populations; the presence of dead birds—especially Corvidae—can be an early indicator of the arrival of the virus.
Vertical transmission of West Nile virus from C. pipiens mosquitoes to their progeny has been demonstrated in the laboratory. It has not been suggested that vertically infected Culex mosquitoes could survive the winter to initiate a WNV amplification cycle the following spring. Culex mosquitoes spend the winter hibernating in protected structures such as root cellars, bank barns, caves, abandoned tunnels and other subterranean locations. The first overwintering adult mosquitoes to test positive for WNV were collected in New York in 2000. Since then, positive samples have been identified in New Jersey in 2003, and in Pennsylvania in 2003-05.[64]
Epidemiology

West Nile virus has been described in Africa, Europe, the Middle East, west and central Asia, Oceania (subtype Kunjin), and most recently, North America.
Recent outbreaks of West Nile virus encephalitis in humans have occurred in Algeria (1994), Romania (1996 to 1997), the Czech Republic (1997), Congo (1998), Russia (1999), the United States (1999 to 2009), Canada (1999–2007), Israel (2000) and Greece (2010).
Epizootics of disease in horses occurred in Morocco (1996), Italy (1998), the United States (1999 to 2001), and France (2000). In 2003, West Nile virus was found in horses in Mexico. In 2011, West Nile virus was found in horses in Sardinia (Italy).
- United States: From 1999 through 2001, the CDC confirmed 149 West Nile virus infections, including 18 deaths. In 2002, a total of 4,156 cases were reported, including 284 fatalities. Thirteen cases in 2002 were contracted through blood transfusion. The cost of WNV-related health care in 2002 was estimated at $200 million. The first human West Nile disease in 2003 occurred in June, and one West Nile-infected blood transfusion was also identified that month.[65]
- In the 2003 outbreak, 9,862 cases and 264 deaths were reported by the CDC. At least 30% of those cases were considered severe, involving meningitis or encephalitis. In 2004, only 2,539 cases and 100 deaths were reported. In 2005, there was a slight increase in the number of cases, with 3,000 cases and 119 deaths reported. Cases increased in 2006, with 4,269 cases and 177 deaths. In 2007, the number of cases reported decreased to 3,623 and the number of deaths dropped to 124. In 2007, 1,227 cases of WNV neuroinvasion disease and 117 deaths occurred.[66]
- In 2008, West Nile surveillance data reported to CDC, a total of 28 states have reported 236 cases of human WNV illness. A total of 137 cases for which such data were available occurred in males, median age patients was 48 years. Dates of illness onset ranged from January 17 to August 14: Two cases were fatal.[67]
- In 2009, 663 cases were reported; of these, 335 were encephalitis or meningitis infections, a reaction to the virus that approximately one in 150 people who get the virus will show. Three hundred and two cases were filed for West Nile fever, the most likely symptom of the virus; 26 cases were unspecified. The state of Texas had the most cases, with 104 total. The total mortality rate for 2009 was 32 deaths of the 663 reported serious cases, a 4.5% casualty rate, but only of the severe infections. Approximately 80% of cases have no symptoms, so the total casualty rate would be less than 1% of total infections in the U.S. These and earlier years data are available from the Centers for Disease Control and Prevention.[68]
- Dallas County, Texas health officials announced in the second week of August 2012 a death toll of nine so far in the county. County Judge Clay Jenkins declared a public health emergency for the county on August 9, 2012, due to the West Nile virus outbreak in the area.[69][70] North Texas has been hit the hardest in 2012, with approximately 663 human cases and 18 deaths, as of August 23, 2012. During the same time, the CDC reported 1,118 human cases and 41 deaths nationwide.[71]
- Canada: One human death occurred in 1999. In 2002, ten human deaths out of 416 confirmed and probable cases were reported by Canadian health officials. In 2003, 14 deaths and 1,494 confirmed and probable cases were reported. Cases were reported in 2003 in Nova Scotia, Quebec, Ontario, Manitoba, Saskatchewan, Alberta, British Columbia, and the Yukon. In 2004, only 26 cases were reported and two deaths; however, 2005 saw 239 cases and 12 deaths. By October 28, 2006, 127 cases and no deaths had been reported. One case was asymptomatic and only discovered through a blood donation. In 2007, 445 Manitobans had confirmed cases of WNV and two people died with a third unconfirmed but suspected.[72] Summer of 2012 saw widespread viral penetration in Canada, coast to coast. [73] 17 people have either tested positive or are suspected of having the virus in Saskatchewan, and only one person has tested positive in Alberta.[74]Saskatchewan has reported 826 cases of WNV plus three deaths.[75] The spread of West Nile Virus infected mosquitoes to British Columbia for the first time was reported in 2009[76]
- Israel: In the year 2000, the CDC found 417 confirmed cases with 326 hospitalizations; 33 of these people died. The main clinical presentations were encephalitis (57.9%), febrile disease (24.4%), and meningitis (15.9%).[77]
- Romania: In 1996–1997, about 500 cases occurred in Romania with a fatality rate of nearly 10%. In 2010, 34 cases were confirmed, with 3 fatalities.[78]
- Greece: In the summer of 2010, several cases were reported mainly in northern Greece, a countrywide total of 262 diagnosed cases and 35 fatalities.[79] In 2011 the virus spread to central Greece but with fewer cases, 101 diagnosed and 9 fatalities.[80]. In 2012, 44 were diagnosed and 3 died.[81] The total number of people in Greece infected by the virus is estimated to 1,800.[82]
Monitoring and control

West Nile virus can be sampled from the environment by the pooling of trapped mosquitoes, testing avian blood samples drawn from wild birds, dogs and sentinel monkeys, as well as testing brains of dead birds found by various animal control agencies and the public. Testing of the mosquito samples requires the use of RT-PCR to directly amplify and show the presence of virus in the submitted samples. When using the blood sera of wild birds and sentinel chickens, samples must be tested for the presence of WNV antibodies by use of immunohistochemistry (IHC)[86] or Enzyme-Linked Immunosorbent Assay (ELISA).[87]
Dead birds, after necropsy, have their various tissues tested for virus by either RT-PCR or IHC, where virus shows up as brown-stained tissue because of a substrate-enzyme reaction.
West Nile control is achieved through mosquito control, by elimination of mosquito breeding sites, larviciding active breeding areas.
Environmentalists have condemned attempts to control the transmitting mosquitoes by spraying pesticide, saying the detrimental health effects of spraying outweigh the relatively few lives which may be saved, and more environmentally friendly ways of controlling mosquitoes are available. They also question the effectiveness of insecticide spraying, as they believe mosquitoes that are resting or flying above the level of spraying will not be killed; the most common vector in the northeastern US, Culex pipiens, is a canopy feeder.
Prevention
Personal protective measures can be taken to greatly reduce the risk of being bitten by an infected mosquito:
- Using insect repellent on exposed skin to repel mosquitoes. EPA-registered repellents include products containing DEET (N,N-diethylmetatoluamide) and picaridin (KBR 3023). DEET concentrations of 30% to 50% are effective for several hours. Picaridin, available at 7% and 15 % concentrations, needs more frequent application. DEET formulations as high as 50% are recommended for both adults and children over 2 months of age. Protect infants less than 2 months of age by using a carrier draped with mosquito netting with an elastic edge for a tight fit.
- When using sunscreen, apply sunscreen first and then repellent. Repellent should be washed off at the end of the day before going to bed.
- Wear long-sleeved shirts which should be tucked in, long pants, and hats to cover exposed skin.
- The application of permethrin-containing (e.g., Permanone) or other insect repellents to clothing, shoes, tents, mosquito nets, and other gear for greater protection. Permethrin is not labeled for use directly on skin. Most repellent is generally removed from clothing and gear by a single washing, but permethrin-treated clothing is effective for up to 5 washings.
- Be aware that most mosquitoes that transmit disease are most active during twilight periods (dawn and dusk or in the evening). A notable exception is the Asian Tiger mosquito which is a daytime feeder and is more apt to be found in, or on the periphery of, shaded areas with heavy vegetation.
- Staying in air-conditioned or well-screened housing, and/ or sleeping under an insecticide treated [[Mosquito net|bed net. Bed nets should be tucked under mattresses and can be sprayed with a repellent if not already treated with an insecticide.
Treatment research
WNV is one of the Japanese encephalitis antigenic serocomplex of viruses. Image reconstructions and cryoelectron microscopy reveal a 45–50 nm virion covered with a relatively smooth protein surface. This structure is similar to the dengue fever virus; both belong to the genus Flavivirus within the family Flaviviridae. The genetic material of WNV is a positive-sense, single strand of RNA, which is between 11,000 and 12,000 nucleotides long; these genes encode seven nonstructural proteins and three structural proteins. The RNA strand is held within a nucleocapsid formed from 12-kDa protein blocks; the capsid is contained within a host-derived membrane altered by two viral glycoproteins.
AMD3100, which had been proposed as an antiretroviral drug for HIV, has shown promise against West Nile encephalitis. Morpholino antisense oligos conjugated to cell penetrating peptides have been shown to partially protect mice from WNV disease.[88] There have also been attempts to treat infections using ribavirin, intravenous immunoglobulin, or alpha interferon.[89] GenoMed, a U.S. biotech company, has found that blocking angiotensin II can treat the "cytokine storm" of West Nile virus encephalitis as well as other viruses.[90]
In 2007, the World Community Grid launched the Discovering Dengue Drugs – Together project. This uses a distributed network of volunteers' computers via the Berkeley Open Infrastructure for Network Computing to perform computer simulations of interacting molecules. Thousands of small molecules are screened for potential antiviral properties with respect to West Nile and related viruses.
See also
Notes
- ^ Nash D, Mostashari F; et al. (2001). "THE OUTBREAK OF WEST NILE VIRUS INFECTION IN THE NEW YORK CITY AREA IN 1999". NEJM. 344 (24): 1807–12. PMID 11407341.
{{cite journal}}: Explicit use of et al. in:|author=(help) - ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 12899140, please use {{cite journal}} with
|pmid=12899140instead. - ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 15663852, please use {{cite journal}} with
|pmid= 15663852instead. - ^ "West Nile Virus: What You Need to Know CDC Fact Sheet". www.CDC.gov. Retrieved 2012.04.09.
{{cite web}}: Check date values in:|accessdate=(help) - ^ Hayes EB, Komar N, Nasci RS, Montgomery SP, O'Leary DR, Campbell GL (2005). "Epidemiology and transmission dynamics of West Nile virus disease". Emerging Infect. Dis. 11 (8): 1167–73. PMID 16102302.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Taylor RM, Hurlbut HS, Dressler HR, Spangler EW, Thrasher D (1953). "Isolation of West Nile virus from Culex mosquitoes". J Egypt Med Assoc. 36 (3): 199–208. PMID 13084817.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Fonseca DM; et al. (2004). "Emerging vectors in the Culex pipiens complex". Science. 303 (5663): 1535–8. doi:10.1126/science.1094247. PMID 15001783.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help) - ^ Spielman A; et al. (2004). "Outbreak of West Nile Virus in North America". Science. 306 (5701): 1473–5. doi:10.1126/science.306.5701.1473c. PMID 15567836.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help) - ^ "Virus culprit tracked down". Kathimerini. 2010-08-21. Retrieved 2010-08-21.
- ^ Centers for Disease Control and Prevention (CDC) (2002). "Laboratory-acquired West Nile virus infections—United States, 2002". MMWR Morb. Mortal. Wkly. Rep. 51 (50): 1133–5. PMID 12537288.
- ^ Fonseca K, Prince GD, Bratvold J; et al. (2005). "West Nile virus infection and conjunctive exposure". Emerging Infect. Dis. 11 (10): 1648–9. PMID 16355512.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Centers for Disease Control and Prevention (CDC) (2002). "Investigation of blood transfusion recipients with West Nile virus infections". MMWR Morb. Mortal. Wkly. Rep. 51 (36): 823. PMID 12269472.
- ^ Centers for Disease Control and Prevention (CDC) (2002). "West Nile virus infection in organ donor and transplant recipients—Georgia and Florida, 2002". MMWR Morb. Mortal. Wkly. Rep. 51 (35): 790. PMID 12227442.
- ^ Centers for Disease Control and Prevention (CDC) (2002). "Intrauterine West Nile virus infection—New York, 2002". MMWR Morb. Mortal. Wkly. Rep. 51 (50): 1135–6. PMID 12537289.
- ^ Centers for Disease Control and Prevention (CDC) (2002). "Possible West Nile virus transmission to an infant through breast-feeding—Michigan, 2002". MMWR Morb. Mortal. Wkly. Rep. 51 (39): 877–8. PMID 12375687.
- ^ Centers for Disease Control and Prevention (CDC) (2003). "Detection of West Nile virus in blood donations—United States, 2003". MMWR Morb. Mortal. Wkly. Rep. 52 (32): 769–72. PMID 12917583.
- ^ SNBTS position on West Nile virus
- ^ Panthier R, Hannoun C, Beytout D, Mouchet J (1968). "[Epidemiology of West Nile virus. Study of a center in Camargue.]". Ann Inst Pasteur, Paris (in French). 115 (3): 435–45. PMID 5711530.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Kumar D, Drebot MA, Wong SJ; et al. (2004). "A seroprevalence study of west nile virus infection in solid organ transplant recipients". Am. J. Transplant. 4 (11): 1883–8. doi:10.1111/j.1600-6143.2004.00592.x. PMID 15476490.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Glass, WG (2005). "Chemokine receptor CCR5 promotes leukocyte trafficking to the brain and survival in West Nile virus infection". Journal of Experimental Medicine. 202 (8): 1087–98. doi:10.1084/jem.20042530. PMC 2213214. PMID 16230476.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help); Unknown parameter|month=ignored (help) - ^ Glass, WG (2006). "CCR5 deficiency increases risk of symptomatic West Nile virus infection". Journal of Experimental Medicine. 203 (1): 35–40. doi:10.1084/jem.20051970. PMC 2118086. PMID 16418398.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help); Unknown parameter|month=ignored (help) - ^ a b Schneider BS, McGee CE, Jordan JM, Stevenson HL, Soong L, Higgs S (2007). "Prior exposure to uninfected mosquitoes enhances mortality in naturally-transmitted west nile virus infection". PLoS ONE. 2 (11): e1171. doi:10.1371/journal.pone.0001171. PMC 2048662. PMID 18000543.
{{cite journal}}: CS1 maint: multiple names: authors list (link) CS1 maint: unflagged free DOI (link) - ^ a b Styer LM, Bernard KA, Kramer LD (2006). "Enhanced early West Nile virus infection in young chickens infected by mosquito bite: effect of viral dose". Am. J. Trop. Med. Hyg. 75 (2): 337–45. PMID 16896145.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Schneider BS, Soong L, Girard YA, Campbell G, Mason P, Higgs S (2006). "Potentiation of West Nile encephalitis by mosquito feeding". Viral Immunol. 19 (1): 74–82. doi:10.1089/vim.2006.19.74. PMID 16553552.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Wasserman HA, Singh S, Champagne DE (2004). "Saliva of the Yellow Fever mosquito, Aedes aegypti, modulates murine lymphocyte function". Parasite Immunol. 26 (6–7): 295–306. doi:10.1111/j.0141-9838.2004.00712.x. PMID 15541033.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Limesand KH, Higgs S, Pearson LD, Beaty BJ (2003). "Effect of mosquito salivary gland treatment on vesicular stomatitis New Jersey virus replication and interferon alpha/beta expression in vitro". J. Med. Entomol. 40 (2): 199–205. doi:10.1603/0022-2585-40.2.199. PMID 12693849.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Wanasen N, Nussenzveig RH, Champagne DE, Soong L, Higgs S (2004). "Differential modulation of murine host immune response by salivary gland extracts from the mosquitoes Aedes aegypti and Culex quinquefasciatus". Med. Vet. Entomol. 18 (2): 191–9. doi:10.1111/j.1365-2915.2004.00498.x. PMID 15189245.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Zeidner NS, Higgs S, Happ CM, Beaty BJ, Miller BR (1999). "Mosquito feeding modulates Th1 and Th2 cytokines in flavivirus susceptible mice: an effect mimicked by injection of sialokinins, but not demonstrated in flavivirus resistant mice". Parasite Immunol. 21 (1): 35–44. doi:10.1046/j.1365-3024.1999.00199.x. PMID 10081770.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Schneider BS, Soong L, Zeidner NS, Higgs S (2004). "Aedes aegypti salivary gland extracts modulate anti-viral and TH1/TH2 cytokine responses to sindbis virus infection". Viral Immunol. 17 (4): 565–73. doi:10.1089/vim.2004.17.565. PMID 15671753.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ CDC
- ^ Hayes EB, Gubler DJ (2006). "West Nile virus: epidemiology and clinical features of an emerging epidemic in the United States". Annu. Rev. Med. 57: 181–94. doi:10.1146/annurev.med.57.121304.131418. PMID 16409144.
- ^ Fradin MS, Day JF (2002). "Comparative efficacy of insect repellents against mosquito bites". N. Engl. J. Med. 347 (1): 13–8. doi:10.1056/NEJMoa011699. PMID 12097535.
- ^ CDC
- ^ Yahoo
- ^ CDC: West Nile Virus - Questions and Answers
- ^ Gorsche R, Tilley P. CMAJ. The rash of West Nile virus infection. 2005 May 24;172(11):1440. Gorsche R, Tilley P. PMID 15911857
- ^ Weiss D, Carr D, Kellachan J, et al. Clinical findings of West Nile virus infection in hospitalized patients, New York and New Jersey, 2000. Emerg Infect Dis. 2001 Jul-Aug;7(4):654-8. PMID 11589170
- ^ Pepperell C, Rau N, Krajden S, et al. West Nile virus infection in 2002: morbidity and mortality among patients admitted to hospital in southcentral Ontario. CMAJ. 2003 May 27;168(11):1399-405. PMID 12771068
- ^ Olejnik E (1952). "Infectious adenitis transmitted by Culex molestus". Bull Res Counc Isr. 2: 210–1.
- ^ Smithburn KC, Jacobs HR (1942). "Neutralization-tests against neurotropic viruses with sera collected in central Africa". Journal of Immunology. 44: 923.
- ^ Melissa S. Nolan; et al. (6 July 2012). "Prevalence of Chronic Kidney Disease and Progression of Disease Over Time among Patients Enrolled in the Houston West Nile Virus Cohort". PLoS One. PLOS. Retrieved 26 August 2012.
{{cite journal}}: Explicit use of et al. in:|author=(help) - ^ "New Study Reveals: West Nile virus is far more menacing & harms far more people". The Guardian Express. The Guardian Express. 26 August 2012. Retrieved 26 August 2012.
- ^ Sejvar JJ, Haddad MB, Tierney BC; et al. (2003). "Neurologic manifestations and outcome of West Nile virus infection". JAMA. 290 (4): 511–5. doi:10.1001/jama.290.4.511. PMID 12876094.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Ahmed S, Libman R, Wesson K, Ahmed F, Einberg K (2000). "Guillain-Barré syndrome: An unusual presentation of West Nile virus infection". Neurology. 55 (1): 144–6. PMID 10891928.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Abroug F, Ouanes-Besbes L, Letaief M; et al. (2006). "A cluster study of predictors of severe West Nile virus infection". Mayo Clin. Proc. 81 (1): 12–6. doi:10.4065/81.1.12. PMID 16438473.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Perelman A, Stern J (1974). "Acute pancreatitis in West Nile Fever". Am. J. Trop. Med. Hyg. 23 (6): 1150–2. PMID 4429184.
- ^ Omalu BI, Shakir AA, Wang G, Lipkin WI, Wiley CA (2003). "Fatal fulminant pan-meningo-polioencephalitis due to West Nile virus". Brain Pathol. 13 (4): 465–72. doi:10.1111/j.1750-3639.2003.tb00477.x. PMID 14655752.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Mathiot CC, Georges AJ, Deubel V (1990). "Comparative analysis of West Nile virus strains isolated from human and animal hosts using monoclonal antibodies and cDNA restriction digest profiles". Res. Virol. 141 (5): 533–43. doi:10.1016/0923-2516(90)90085-W. PMID 1703658.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Tyler KL, Pape J, Goody RJ, et al. CSF findings in 250 patients with serologically confirmed West Nile virus meningitis and encephalitis. Neurology. 2006 Feb 14;66(3):361-5. Epub 2005 Dec 28. PMID 16382032
- ^ Watson J., Patel P. Jones R., et al. Clinical Characteristics and Functional Outcomes of West Nile Fever[66.39.42.45/topics/west_nile/pdf/wnv_clinical_characteristics.pdf]
- ^ Carson1 P, Konewko P., Wold k., et al. Long-Term Clinical and Neuropsychological Outcomes of West Nile Virus Infection. Oxford Journals Medicine Clinical Infectious Diseases Volume 43, Issue 6 Pp. 723-730.
- ^ Klee AL, Maidin B, Edwin B, et al. Long-term prognosis for clinical West Nile virus infection. Emerg Infect Dis. 2004 Aug;10(8):1405-11. PMID 15496241
- ^ Galli M, Bernini F, Zehender G (2004). "Alexander the Great and West Nile virus encephalitis". Emerging Infect. Dis. 10 (7): 1330–2, author reply 1332–3. PMID 15338540.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ West, Christy (2010-02-08). "Different West Nile Virus Genetic Lineage Evolving?". The Horse, online edition. Retrieved 2010-02-10.
{{cite journal}}: Cite journal requires|journal=(help); Invalid|nopp=from statements by Orsolya Kutasi, DVM, of the Szent Istvan University, Hungary at the 2009 American Association of Equine Practitioners Convention, December 5–9 , 2009(help); Unknown parameter|nopp=ignored (|no-pp=suggested) (help) - ^ Venter, M., S. Human, S. van Niekerk, J. Williams, C. van Eeden, and F. Fatal neurologic disease and abortion in mare infected with lineage 1 West Nile virus, South Africa. Emerging Infectious Diseases, 2011 Aug [July 27, 2011], http://www.cdc.gov/EID/content/17/8/101794.htm.
- ^ St. Leger, J., G. Wu, M. Anderson, L. Dalton, E. Nilson, and D. Wang. 2011. West Nile virus infection in killer whale, Texas, USA, 2007. Emerging Infectious Diseases, 2011 Aug [July 27, 2011]. http://www.cdc.gov/EID/content/17/8/101979.htm.
- ^ "Alexander the Great and West Nile Virus Encephalitis". Centers for Disease Control and Prevention. Retrieved on 2009-04-26
- ^ Smithburn KC, Hughes TP, Burke AW, Paul JH (1940). "A Neurotropic Virus Isolated from the Blood of a Native of Uganda". Am. J. Trop. Med. 20 (1): 471–92.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Work TH, Hurlbut HS, Taylor RM (1953). "Isolation of West Nile virus from hooded crow and rock pigeon in the Nile delta". Proc. Soc. Exp. Biol. Med. 84 (3): 719–22. PMID 13134268.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Bernkopf H, Levine S, Nerson R (1953). "Isolation of West Nile virus in Israel". J. Infect. Dis. 93 (3): 207–18. PMID 13109233.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Nash D, Mostashari F; et al. (2001). "THE OUTBREAK OF WEST NILE VIRUS INFECTION IN THE NEW YORK CITY AREA IN 1999". NEJM. 344 (24): 1807–12. PMID 11407341.
{{cite journal}}: Explicit use of et al. in:|author=(help) - ^ Calisher CH (2000). "West Nile virus in the New World: appearance, persistence, and adaptation to a new econiche—an opportunity taken". Viral Immunol. 13 (4): 411–4. doi:10.1089/vim.2000.13.411. PMID 11192287.
- ^ C. Michael Hogan. 2008. Barbary Macaque: Macaca sylvanus, GlobalTwitcher.com
- ^ Bugbee, LM (2004). "The discovery of West Nile virus in overwintering Culex pipiens (Diptera: Culicidae) mosquitoes in Lehigh County, Pennsylvania". Journal of the American Mosquito Control Association. 20 (3): 326–7. PMID 15532939.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help); Unknown parameter|month=ignored (help) - ^ CDC: West Nile Virus - Statistics, Surveillance, and Control
- ^ CDC: West Nile Virus - Statistics, Surveillance, and Control
- ^ CDC: West Nile Virus - Statistics, Surveillance, and Control
- ^ CDC: West Nile Virus - Statistics, Surveillance, and Control
- ^ "Dallas County declares state of emergency due to West Nile". WFAA.com. Retrieved 10 August 2012.
- ^ Correll, Robin. "Epidemic of West Nile Virus Hits Dallas County". The Disease Daily. Retrieved 10 August 2012.
- ^ "West Nile risk likely to run six more weeks, CDC says". Dallas Morning News. Retrieved 23 August 2012.
- ^ Province of Manitoba | Manitoba Health | West Nile virus
- ^ [1]
- ^ Error
- ^ CTV.ca | Sask. reports 339 cases of West Nile, one death
- ^ News | West Nile virus found in BC mosquitoes
- ^ Chowers, MY (2001). "Clinical characteristics of the West Nile fever outbreak, Israel, 2000". Emerging Infectious Diseases. 7 (4): 675–8. doi:10.3201/eid0704.010414. PMC 2631759. PMID 11585531. Retrieved 2006-06-07.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help); Unknown parameter|month=ignored (help) - ^ "3 dead from West Nile virus in Romania". cnn.com. 2010-09-13. Retrieved 2010-12-21.
- ^ "Έκθεση: Επιδημία Λοίμωξης από τον Ιό του Δυτικού Νείλου, 2010" (PDF) (in Greek). Hellenic Centre for Disease Control and Prevention (KEELPNO). 2011. Retrieved 2012-08-09.
- ^ "Έκθεση επιδημιολογικής επιτήρησης - Έτος 2011" (PDF). Hellenic Centre for Disease Control and Prevention (KEELPNO). 2012-05-07. Retrieved 2012-07-10.
- ^ "Και τρίτος νεκρός από τον ιό του Δυτικού Νείλου" (in Greek). in.gr. 2012-08-08. Retrieved 2012-08-08.
- ^ "1800 άνθρωποι μολύνθηκαν από τον ιό του Δυτικού Νείλου στην Ελλάδα" (in Greek). SigmaLive/news24.7. 2012-07-24. Retrieved 2012-07-24.
- ^ [[National Park Service: Mosquito Management
- ^ Oklahoma State University: Mosquitos and West Nile virus
- ^ University of Florida: Asian Tiger Mosquito, Aedes albopictus (Skuse) (Insecta: Diptera: Culicidae)
- ^ Jozan, M (2003). "Detection of West Nile virus infection in birds in the United States by blocking ELISA and immunohistochemistry". Vector-borne and Zoonotic Diseases. 3 (3): 99–110. doi:10.1089/153036603768395799. PMID 14511579.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help); Unknown parameter|month=ignored (help) - ^ Hall, RA (1995). "Immunodominant epitopes on the NS1 protein of MVE and KUN viruses serve as targets for a blocking ELISA to detect virus-specific antibodies in sentinel animal serum". Journal of Virological Methods. 51 (2–3): 201–10. doi:10.1016/0166-0934(94)00105-P. PMID 7738140.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help); Unknown parameter|month=ignored (help) - ^ Deas, Tia S (2007). "In vitro resistance selection and in vivo efficacy of morpholino oligomers against West Nile virus". Antimicrob Agents Chemother. 51 (7): 2470–82. doi:10.1128/AAC.00069-07. PMC 1913242. PMID 17485503.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help); Unknown parameter|month=ignored (help) - ^ Hayes EB, Sejvar JJ, Zaki SR, Lanciotti RS, Bode AV, Campbell GL (2005). "Virology, pathology, and clinical manifestations of West Nile virus disease". Emerging Infect. Dis. 11 (8): 1174–9. PMC 3320472. PMID 16102303.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Moskowitz DW, Johnson FE (2004). "The central role of angiotensin I-converting enzyme in vertebrate pathophysiology". Curr Top Med Chem. 4 (13): 1433–54. doi:10.2174/1568026043387818. PMID 15379656.
External links
- Recent progress in West Nile virus diagnosis and vaccination
- West Nile Virus - U.S. Centers for Disease Control and Prevention (CDC) page
- Recommendations for Protecting Laboratory, Field, and Clinical Workers from West Nile Virus Exposure
- West Nile Virus Resource Guide—National Pesticide Information Center
- Vaccine Research Center (VRC)—Information concerning WNV vaccine research studies
- Nature news article on West Nile paralysis
- CBC News Coverage of West Nile in Canada
- Gene mutation turned West Nile virus into killer disease among crows
- Virus Pathogen Database and Analysis Resource (ViPR): Flaviviridae
- Species Profile- West Nile Virus (Flavivirus), National Invasive Species Information Center, United States National Agricultural Library. Lists general information and resources for West Nile Virus.
- 3D macromolecular structures of the West Nile Virus archived in the EM Data Bank(EMDB)
- West Nile Virus - West Nile Encephalitis Brain Scans




