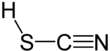Content deleted Content added
Updating {{chembox}} (changes to verified and watched fields - updated 'KEGG_Ref', 'Verifiedfields', 'Watchedfields') per Chem/Drugbox validation (report errors or bugs) |
Saehrimnir (talk | contribs) No edit summary Tag: Visual edit |
||
| Line 1: | Line 1: | ||
{{Chembox |
{{Chembox |
||
|Verifiedfields = changed|Watchedfields = changed|verifiedrevid = 470607041|Reference = <ref>''[[Merck Index]]'', 11th Edition, '''9257'''.</ref>|ImageFileL1 = Thiocyanic acid.png|ImageFileL1_Ref = {{chemboximage|correct|??}}|ImageNameL1 = Skeletal formula of thiocyanic acid|ImageFileR1 = Thiocyansäure.png|ImageFileR1_Ref = {{chemboximage|correct|??}}|ImageNameR1 = Skeletal formula of thiocyanic acid with the explicit hydrogen added|ImageFile2 = Thiocyanic-acid-3D-vdW.png|ImageFile2_Ref = {{chemboximage|correct|??}}|ImageSize2 = 100|ImageName2 = Spacefill model of thiocyanic acid|IUPACName = Nitridosulfanidocarbon<ref>{{Cite web|title=thiocyanic acid (CHEBI:29200)|url=https://www.ebi.ac.uk/chebi/searchId.do?chebiId=29200|work=Chemical Entities of Biological Interest|publisher=European Bioinformatics Institute|accessdate=5 June 2012|location=USA|date=18 October 2009|at=Main.}}</ref>|OtherNames = {{Unbulleted list|Hydrogen thiocyanate{{Citation needed|date=June 2012}}|Sulfocyanic acid{{Citation needed|date=June 2012}}|Sulfocyanide{{Citation needed|date=June 2012}} |
|||
| Verifiedfields = changed |
|||
| ⚫ | |||
| Watchedfields = changed |
|||
| verifiedrevid = 470607041 |
|||
| Reference = <ref>''[[Merck Index]]'', 11th Edition, '''9257'''.</ref> |
|||
| ImageFileL1 = Thiocyanic acid.png |
|||
| ImageFileL1_Ref = {{chemboximage|correct|??}} |
|||
| ImageNameL1 = Skeletal formula of thiocyanic acid |
|||
| ImageFileR1 = Thiocyansäure.png |
|||
| ImageFileR1_Ref = {{chemboximage|correct|??}} |
|||
| ImageNameR1 = Skeletal formula of thiocyanic acid with the explicit hydrogen added |
|||
| ImageFile2 = Thiocyanic-acid-3D-vdW.png |
|||
| ImageFile2_Ref = {{chemboximage|correct|??}} |
|||
| ImageSize2 = 100 |
|||
| ImageName2 = Spacefill model of thiocyanic acid |
|||
| IUPACName = Nitridosulfanidocarbon<ref>{{Cite web|title=thiocyanic acid (CHEBI:29200)|url=https://www.ebi.ac.uk/chebi/searchId.do?chebiId=29200|work=Chemical Entities of Biological Interest|publisher=European Bioinformatics Institute|accessdate=5 June 2012|location=USA|date=18 October 2009|at=Main.}}</ref> |
|||
| OtherNames = {{Unbulleted list|Hydrogen thiocyanate{{Citation needed|date=June 2012}}|Sulfocyanic acid{{Citation needed|date=June 2012}}|Sulfocyanide{{Citation needed|date=June 2012}} |
|||
}} |
|||
| ⚫ | |||
| CASNo = 463-56-9 |
| CASNo = 463-56-9 |
||
| CASNo_Ref = {{cascite|correct|CAS}} |
| CASNo_Ref = {{cascite|correct|CAS}} |
||
| Line 39: | Line 23: | ||
| StdInChIKey = ZMZDMBWJUHKJPS-UHFFFAOYSA-N |
| StdInChIKey = ZMZDMBWJUHKJPS-UHFFFAOYSA-N |
||
| StdInChIKey_Ref = {{stdinchicite|correct|chemspider}} |
| StdInChIKey_Ref = {{stdinchicite|correct|chemspider}} |
||
| ⚫ | |||
}} |
|||
| ⚫ | |||
| C = 1 |
| C = 1 |
||
| H = 1 |
| H = 1 |
||
| Line 48: | Line 31: | ||
| Odor = pungent |
| Odor = pungent |
||
| Solubility = Miscible |
| Solubility = Miscible |
||
| |
| SolubleOther = soluble in [[ethanol]], [[diethyl ether]] |
||
| LogP = 0.429 |
| LogP = 0.429 |
||
| pKa = 0.926 |
| pKa = 0.926 |
||
| Line 54: | Line 37: | ||
| Density = 2.04 g/cm<sup>3</sup> |
| Density = 2.04 g/cm<sup>3</sup> |
||
| MeltingPtC = 5 |
| MeltingPtC = 5 |
||
| ⚫ | |||
}} |
|||
| ⚫ | |||
| EUIndex = 615-003-00-8 |
| EUIndex = 615-003-00-8 |
||
| EUClass = {{Hazchem Xn}} |
| EUClass = {{Hazchem Xn}} |
||
| RPhrases = {{R20/21/22}}, {{R32}}, {{R52/53}} |
| RPhrases = {{R20/21/22}}, {{R32}}, {{R52/53}} |
||
| SPhrases = {{S2}}, {{S13}} |
| SPhrases = {{S2}}, {{S13}} |
||
| ⚫ | |||
}} |
|||
| ⚫ | |||
| Function = alkanenitriles |
| Function = alkanenitriles |
||
| OtherFunctn = {{Unbulleted list|[[Hydrogen cyanide]]|[[Cyanogen iodide]]|[[Cyanogen bromide]]|[[Cyanogen chloride]]|[[Cyanogen fluoride]]|[[Acetonitrile]]|[[Aminoacetonitrile]]|[[Glycolonitrile]]|[[Cyanogen]]}} |
| OtherFunctn = {{Unbulleted list|[[Hydrogen cyanide]]|[[Cyanogen iodide]]|[[Cyanogen bromide]]|[[Cyanogen chloride]]|[[Cyanogen fluoride]]|[[Acetonitrile]]|[[Aminoacetonitrile]]|[[Glycolonitrile]]|[[Cyanogen]]}} |
||
}} |
}}}} |
||
}} |
|||
'''Thiocyanic acid''' is a [[chemical compound]] with the formula HSCN that exists as a mixture with the [[isomer]]ic compound isothiocyanic acid (HNCS).<ref>Holleman, A. F.; Wiberg, E. ''Inorganic Chemistry'' Academic Press: San Diego, 2001. ISBN 0-12-352651-5.</ref> It is the sulfur analog of [[cyanic acid]] (HOCN). |
'''Thiocyanic acid''' is a [[chemical compound]] with the formula HSCN that exists as a mixture with the [[isomer]]ic compound isothiocyanic acid (HNCS).<ref>Holleman, A. F.; Wiberg, E. ''Inorganic Chemistry'' Academic Press: San Diego, 2001. ISBN 0-12-352651-5.</ref> It is the sulfur analog of [[cyanic acid]] (HOCN). |
||
Revision as of 03:28, 11 July 2013
| |||

| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Nitridosulfanidocarbon[2]
| |||
Other names
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 3DMet | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.006.672 | ||
| EC Number |
| ||
| 25178 | |||
| KEGG | |||
| MeSH | thiocyanic+acid | ||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| CHNS | |||
| Molar mass | 59.09 g·mol−1 | ||
| Appearance | colorless, oily liquid | ||
| Odor | pungent | ||
| Density | 2.04 g/cm3 | ||
| Melting point | 5 °C (41 °F; 278 K) | ||
| Miscible | |||
| Solubility | soluble in ethanol, diethyl ether | ||
| log P | 0.429 | ||
| Acidity (pKa) | 0.926 | ||
| Basicity (pKb) | 13.071 | ||
| Related compounds | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Thiocyanic acid is a chemical compound with the formula HSCN that exists as a mixture with the isomeric compound isothiocyanic acid (HNCS).[3] It is the sulfur analog of cyanic acid (HOCN).
It is a weak acid, bordering on strong, with a pKa of 1.1 at 20°C and extrapolated to zero ionic strength.[4]
HSCN is predicted to have a triple bond between carbon and nitrogen. It has been observed spectroscopically but has not been isolated as a pure substance.[5]
The salts and esters of thiocyanic acid are known as thiocyanates. The salts are composed of the thiocyanate ion (-SCN) and a suitable metal cation (e.g., potassium thiocyanate, KSCN). The esters of thiocyanic acid have the general structure R-SCN.
References
- ^ Merck Index, 11th Edition, 9257.
- ^ "thiocyanic acid (CHEBI:29200)". Chemical Entities of Biological Interest. USA: European Bioinformatics Institute. 18 October 2009. Main. Retrieved 5 June 2012.
- ^ Holleman, A. F.; Wiberg, E. Inorganic Chemistry Academic Press: San Diego, 2001. ISBN 0-12-352651-5.
- ^ Martell, A. E.; Smith, R. M.; Motelaitis, R. J. NIST Database 46 National Institute of Standards and Technology: Gaithersburg, MD, 2001.
- ^ Wierzejewska, M.; Mielke, Z. (2001). "Photolysis of Isothiocyanic Acid HNCS in Low-Temperature Matrices. Infrared Detection of HSCN and HSNC Isomers". Chemical Physics Letters. 349: 227–234. doi:10.1016/S0009-2614(01)01180-0.
{{cite journal}}: CS1 maint: multiple names: authors list (link)