→Propose closing as is: collapse |title=More detailed talk on easy closure |
Mdaniels5757 (talk | contribs) ew. close per WP:ANRFC |
||
| Line 237: | Line 237: | ||
== RFC: Should the default form of the periodic table be changed to put Lu and Lr in group 3, rather than La and Ac? == |
== RFC: Should the default form of the periodic table be changed to put Lu and Lr in group 3, rather than La and Ac? == |
||
{{atop|Per consensus to abandon this trainwreck of a thread as-is. —[[User:Mdaniels5757|Mdaniels5757]] ([[User talk:Mdaniels5757|talk]]) 20:23, 6 August 2020 (UTC)}} |
|||
Should the default appearance of the periodic table on Wikipedia be changed to what it was in 2016, i.e. to have lutetium (<sub>71</sub>Lu) and lawrencium (<sub>103</sub>Lr) in group 3 rather than lanthanum (<sub>57</sub>La) and actinium (<sub>89</sub>Ac)? Or should it simply have markers below yttrium where all the lanthanides and actinides are supposed to go in? [[User:Double sharp|Double sharp]] ([[User talk:Double sharp|talk]]) 10:08, 20 July 2020 (UTC) |
Should the default appearance of the periodic table on Wikipedia be changed to what it was in 2016, i.e. to have lutetium (<sub>71</sub>Lu) and lawrencium (<sub>103</sub>Lr) in group 3 rather than lanthanum (<sub>57</sub>La) and actinium (<sub>89</sub>Ac)? Or should it simply have markers below yttrium where all the lanthanides and actinides are supposed to go in? [[User:Double sharp|Double sharp]] ([[User talk:Double sharp|talk]]) 10:08, 20 July 2020 (UTC) |
||
| Line 3,432: | Line 3,432: | ||
::::Stop it. Stop this thread. By any means. Again stating "GF" arguments is the problem. -[[User:DePiep|DePiep]] ([[User talk:DePiep|talk]]) 23:42, 2 August 2020 (UTC) |
::::Stop it. Stop this thread. By any means. Again stating "GF" arguments is the problem. -[[User:DePiep|DePiep]] ([[User talk:DePiep|talk]]) 23:42, 2 August 2020 (UTC) |
||
:::::{{ping|DePiep}} But it is already stopped? I simply meant to say "go to the bottom for the new thread". [[User:Double sharp|Double sharp]] ([[User talk:Double sharp|talk]]) 02:30, 3 August 2020 (UTC) |
:::::{{ping|DePiep}} But it is already stopped? I simply meant to say "go to the bottom for the new thread". [[User:Double sharp|Double sharp]] ([[User talk:Double sharp|talk]]) 02:30, 3 August 2020 (UTC) |
||
{{abot}} |
|||
== Caption wording == |
== Caption wording == |
||
Revision as of 20:23, 6 August 2020
 | Periodic table is a featured article; it (or a previous version of it) has been identified as one of the best articles produced by the Wikipedia community. Even so, if you can update or improve it, please do so. | |||||||||||||||||||||||||||
| This article appeared on Wikipedia's Main Page as Today's featured article on February 28, 2004, and on January 8, 2018. | ||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||
| This article is of interest to the following WikiProjects: | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Index
|
|||||||||||||||||
|
This page has archives. Sections older than 60 days may be automatically archived by Lowercase sigmabot III when more than 4 sections are present. |
Mendeleev Tabli first edition
According to the final chronology of the first publications of the Mendeleev’s Periodic Table (Druzhinin, 2020[1]), the first Medneleev ‘s Table published March 26-27 [O.S. March 14-15] 1869 in the Mendeleev’s The Principles of Chemistry 1st Edition, and around March 17 [O.S. March 29] 1869 Mendeleev printed separate broadsheets with Periodic table - Attempt - to be sent to overseas scientists; April 17/19 for the first time the Mendeleev’ Periodic table was published in Europe in Journal für Praktische Chemie (in German)[2]
References
- ^ Druzhinin, Petr (2020). "The First Publication of Mendeleev's Periodic System of Elements: A New Chronology". Historical Studies in the Natural Sciences. 50: 129–182.
- ^ Mendeleev, Dmitri (1869). "Versuche eines Systems der Elemente nach ihren Atomgewichten und chemischen Functionen" [System of Elements according to their Atomic Weights and Chemical Functions]. Journal für Praktische Chemie. 106: 251.
A relevant argument, unfortunately incomplete in sources
I wrote this paragraph, but then deleted it:
| “ | Because of the almost completely regular variation of lattice and solvation energies across the lanthanide series, the most important factor determining redox stability of lanthanide compounds is the third ionisation energy. This almost always involves the loss of one f electron, for which ligand field stabilisation energies are negligible as the f shells are deeply buried; that is distinct from the behaviour of the d transition elements. In almost all cases this ionisation is M2+ (4fn) → M3+ (4fn−1); although lanthanum (La2+ [Xe]5d1) and gadolinium (Gd2+ [Xe]4f75d1) are exceptions, the regular configurations [Xe]4f1 and [Xe]4f8 are very low-lying excited states (energies 0.8881 eV and 0.295 eV) respectively that negligibly affect the ionisation energy trend on being subtracted (the third ionisation energies of La and Gd respectively are 19.1773 eV and 20.63 eV). Similar processes are observed for example in the condensation of the lanthanides, which generally involves the transition 4fn6s2 → 4fn−15d16s2 and so varies in energy similarly to the above 4fn → 4fn−1 ionisations. In condensed phases the configurations of the Ln2+ may also change due to smaller destabilisation of the 5d orbitals compared to the 4f by ligand field effects. Energy variation of a process across the lanthanide series can be predicted from how the number of 4f electrons changes during its reactions. However, lutetium cannot be considered to be of this type as an f shell cannot hold the 15 electrons that would be required. Similarly, if we consider the processes where it is the relative stability of M3+ and M4+ that is at stake, then lanthanum cannot be considered as getting it to the +4 state would require breaking open the xenon core. (cite Johnson from the article) | ” |
The problem is that this argument, so far as it is taken in the source, does not clearly support either La-Yb or Ce-Lu as the 4f row. (Of course, there are lots of arguments about why La-Yb is the right answer and why the arguments for Ce-Lu are focusing on chemical irrelevancies in the literature. I know it probably doesn't look like the periodic table in your textbook, but (1) that textbook is probably not focusing on group 3 and (2) if you can cite Seaborg, as well as Landau and Lifshitz, on your side, like I have, it's definitely not a fringe view. XD) In order to make it complete, you would have to note that physical properties universally support the +2 to +3 process' double periodicity, but so far I haven't seen that being noted. Which is admittedly a bit puzzling, because it is absolutely obvious from the cited values of melting and boiling points: minima occur at Eu and Yb, just as they do at Mn and Zn. Double sharp (talk) 04:44, 18 May 2020 (UTC)
- Deleting it was a good move.
- Johnson: The relevance of his reference to the third ionisation energy involving the loss of one f electron in the free atom, is puzzling.
- The Ln metals, as opposed to the free atoms, are mostly f1-14d1s2. In their most stable trivalent cationic forms they are f1-14. Here, Ce3+ is f1, Pr3+ is f2, ending with Yb3+ f13 and Lu3+ f14. Thus, the f-block metals and their cations run from Ce-Lu. La is not include in the particular set, since La3+ is [Xe] i.e. it has no f electron.
- As Seaborg wrote, "it is the electronic structure of the ions and compounds that we are chiefly concerned with in chemistry".
- Seaborg again: An examination of Seaborg's article on the development of the actinide hypothesis shows he consistently distinguished between La-Ac, and Ce-Lu, Th-Lr. See figures 2, and 4-6 (pp. 52, 54-56).
- The right answer: There are no arguments about why La-Yb is the "right" answer. Rather, there are some arguments in the literature suggesting La-Yb looks better in some cases. Many of these are one-shots based on a single property. Jensen had a red-hot go at corralling these arguments in 1982, in support of La-Yb, but his effort failed to gain traction. He was criticized by Scerri, (chair of the IUPAC Group 3 project group) and Parsons (2018, p. 143), for being too selective in his arguments. There was Holden (1985) who looked at seven properties but estimated the argument of Landau & Ligshitz (1958) to be the strongest. For a discussion as to the unreliability of Landau and Lifshitz, see here.
- A survey by the IUPAC Group 3 project team found that the majority of textbook and other periodic tables in the 18-column format show the elements of group 3 as scandium, yttrium, lanthanum and actinium. The survey did not include a Google image search, since such results are plagued by bias arising from the false impression that the IUPAC *-** table is “official”, and unreliable sources.
- Mathias (1969) grumbled about La in group 3 being the most popular form. Myers, Oldham and Tocci (2004, p. 130) found La and Ac to be the most popular form of periodic table, a sentiment echoed by Clarke and White (2008); and Lavelle (2008; 2009).
- Clark RW & White GD 2008, "The flyleaf periodic table", Journal of Chemical Education, vol. 85, no. 4, p. 497
- Holden NE 1985, Status of the lanthanides and actinides in the periodic table, accessed 24 November 2016
- Jensen WB 1982, The positions of lanthanum (actinium) and lutetium (lawrencium) in the periodic table, Journal of Chemical Education vol. 59, pp. 634−636
- Lavelle L 2008, "Lanthanum (La) and actinium (Ac) should remain in the d-block", Journal of Chemical Education, vol. 85, no. 11, pp. 1482–1483, doi: 10.1021/ed085p1482
- —— 2009, "Response to misapplying the periodic law", Journal of Chemical Education, vol. 86, no. 10, p. 1187, doi: 10.1021/ed086p1187
- Mathias BT 1969, "Systematics of superconductivity", in PR Wallace (ed.), "Superconductivity: Proceedings of the Advanced Summer Study Institute on Superconductivity", McGill University, Montreal, vol. 1, Gordon and Breach, New York, pp. 225−294
- Myers RT, Oldham KB & Tocci S 2004, Holt chemistry, Holt, Rinehart and Winston, Orlando
- Scerri ER & Parsons W 2018, "What elements belong in Group 3 of the periodic table?". In E Scerri & G Restrepo (eds), Mendeleev to Oganesson: A multidisciplinary perspective on the periodic table, Oxford University Press, New York, pp. 140–151
- Chemical irrelevancies: There are no arguments in the literature, to my knowledge, saying Ce-Lu is focusing on "chemical irrelevancies".
- Double periodicity: As to Eu and Yb, and Mn and Zn, it is more relevant, from a chemistry based view, rather than a physical property-based view, to note double periodicity for the 4f and 3d metals pivots around Gd and Lu, and Mn and Zn.
- Shchukarev (1974, p. 118), a well-regarded Russian chemist, supported -La-Ac on the grounds that the 4f shell does not start filling until cerium and that (effectively) the filling sequence—which runs from cerium to lutetium—is periodic, with two periods. Thus, after the occurrence of a half-full 4f shell at europium and gadolinium, the filling sequence repeats with the occurrence of a full shell at ytterbium and lutetium (Rokhlin 2003, pp. 4–5). A similar, but weaker, periodicity (Wiberg 2001, p. 1643–1645) is seen in the actinoids, with a half full 5f shell at americium and curium, and a full shell at nobelium and lawrencium.
- Placing lutetium and lawrencium under yttrium obscures the start of the filling of the f-block (it would appear to start at lanthanum) and visually truncates its double periodicity (it would be cut off at ytterbium whereas it would actually end in the d-block). In Shchukarev’s words, the "correctness of placing of imitators before gadolinium and curium as well as lutetium and lawrencium, would be lost. The exceptional uniqueness of Gd and Cm, akin to that of Mn and Zn, would also be unclear."
- Here is what a lanthanum table looks like:
+4 +2 | +4 +2
Ce Pr Nd Pm Sm Eu Gd | Tb Dy Ho Er Tm Yb Lu
½f ½f | f f
|
+4 +2 | +4 +2
Th Pa U Np Pu Am Cm | Bk Cf Es Fm Md No Lr
½f ½f f f
- Shchukarev’s imitators are Eu2+ and Yb2+ which like to attain the Gd3+ and Lu3+ cores. Then there is Ce4+, which likes to attain the core of its lanthanoid progenitor namely La3+; and Tb4+ attaining the same configuration as Gd3+.
- Europium, the most reactive of the lanthanoids, is associated with the place of a halogen, and gadolinium is associated with place of a noble gas, although to a much lesser degree. See the Rare-earth metal long term air exposure test (Hamric 2007). Terbium to lutetium are much less reactive, and the Group 17/18 analogy is not seen here.
- The f-block contraction starts with Ce3+ and culminates in Lu3+.
- The lutetium option is less regular:
+4 +2 | +4 +2
La Ce Pr Nd Pm Sm Eu | Gd Tb Dy Ho Er Tm Yb
½f | ½f f
|
+4 +2 | +4 +2
Ac Th Pa U Np Pu Am | Cm Bk Cf Es Fm Md No
½f ½f f
- The ½-filled and filled f- sub-shell regularity is lost; the correspondence of europium and gadolinium to Group 17 and 18 metals is lost; the f-block contraction starts with Ce3+ and awkwardly finishes in the d-block.
- Shchukarev’s approach is consistent with the observation of a progressively delayed start of filling of the f-shell as one goes down the periodic table i.e. at cerium for 4f; at protactinium for 5f although some 5f involvement may be possible for thorium (Edelstein & Kot 1993, p. 86); and at E123−125(?) for 5g (Eugen Schwarz, pers. comm. 8 Dec 2019).
- This double periodicity had been noted by Klemm (1929; 1930) on the basis of atomic structure (Remy 1956, p. 492); and by Sobolev (2000, pp. 44–45).
- Edelstein NM and Kot WK 1993, "Spectroscopic and magnetic studies of tetravalent Pa and trivalent Th compounds", Journal of Alloys and Compounds, vol. 193. pp. 82–87
- Hamric D 2007, "Rare-earth metal long term air exposure test", Metallium, Inc., viewed 7 March 2020
- Horovitz O & Sârbu C 2005, "Characterisation and classification of lanthanides by multivariate-analysis methods", Journal of Chemical Education, vol. 82 no. 3, pp. 473–483, doi: 10.1021/ed082p473
- Klemm W 1929, "Measurements on divalent and quadrivalent rare earth compounds. II. A system of rare earths based on periodic property changes of their ions", Zeitschrift für anorganische und allgemeine Chemie, vol. 184, p. 345
- —— 1930, "Measurements on divalent and quadrivalent rare earth compounds. IV. Supplement to the communication: A system of rare earths", Zeitschrift für anorganische und allgemeine Chemie, vol. 184, p. 29
- Remy H 1956, Treatise on inorganic chemistry, vol 2, Elsevier, Amsterdam
- Rokhlin LL 2003, Magnesium alloys containing rare earth metals: Structure and properties, Taylor & Francis, London
- Sobolev BP 2000, The rare earth trifluorides: The high temperature chemistry of the rare earth trifluorides, vol. 1, Institut d'Estudis Catalans, Barcelona, pp. 44–45
- Ternström T 1976, "Subclassification of lanthanides and actinides", Journal of Chemical Education, vol. 53, no. 10, pp. 629–631
- Wiberg N 2001, Inorganic Chemistry, Academic Press, San Diego
- Physical properties: Concluding with these, lutetium resembles the 5d transition metals more than is the case for lanthanum. That said, lutetium resembles closely erbium and holmium, and while it melts at a slightly higher temperature and is essentially non-magnetic, the details of producing, purifying and fabricating it are almost identical to that for holmium (Spedding et al. 1968).
- Spedding FH & Beadry BJ 1968, "Lutetium", in CA Hampel (ed.), The encyclopedia of the chemical elements, Reinhold Book Corporation, New York, pp. 374–378
Recent attempts to change the periodic table (Scerri 2020)
Here's a link to the latest article by Eric Scerri, a world authority on the periodic table.
Here are some extracts from, and my commentary on, this interesting piece:
Philosophy v pragmatism?
The focus of the paper is philosophy rather than pragmatism. (p.2)
I suggest a need to focus on philosophy and pragmatism.
Focusing on just one results in needless arguments, including of the kind I used to make.
Insisting on one PT
Scerri writes:
- "There is no need to insist on the periodic table having a format that is suited mainly for the purposes of the chemical community and for chemical educators." (p. 6)
As far as I know, no one has made such an insistence. It is rather a case of people insisting on such a table within a particular sub-context or interest dependence, and usually not making this clear enough.
Who owns the PT?
Scerri writes:
- "The periodic table has now become as much the property of physicists, geologists, astronomers and others as it is of its chemical originators." (p. 7)
This is a contentious generalisation.
I suggest the periodic table, in the first instance, remains the organising icon of chemistry. Thus, here is what Scerri said in the 2nd (2020) edition of his Red Book:
- "…it helps to remember that, when all is said and done, the periodic table remains primarily in the domain of chemistry, although the relationship between chemistry and the underlying explanation from physics remains as the underlying theme… (p. ix)"
Rather than becoming a shared commodity, the periodic table concept has been borrowed, adapted, tailored and presented in various different guises—including the 15-element wide f-block version—by the physicists, geologists, astronomers and others.
As Scerri rightly says:
- "It becomes increasingly clear that there may not be any such thing as one optimal table in a purely objective sense. The question seems to depend on what criteria are considered and, most importantly perhaps, on whether one favours chemical or physical criteria or general didactic considerations." (p. 12)
To this commendable end, he goes on:
- "We should accept that a degree of convention must be used in selecting a periodic table that can be presented as perhaps the best possible table that combines objective factors as well as interest dependence." (p. 14)
Quite so, having regard to the priorities of each interest group.
The Madelung Rule
- "In any case, it is interesting to see that Pyykkö admits that it is surprising, in view of the relativistic effects, that the Madelung rule survives so well all the way up to atomic number 172." [!] (p. 8)
As far as the MR anomalies/symmetry breaking we observe in real life are concerned, the analogy is to an aeroplane experiencing turbulence. The flight path always returns to normal, after each turbulence episode.
The MR can also be regarded as the "spine" underlying the pattern of free atom electron configurations. The spine has bumps, dips, and knobbly bits on it, but still runs "true", so to speak.
Group 3
- "While the majority of textbook and other periodic tables in the 18-column format show the elements of this group as scandium, yttrium, lanthanum and actinium, a significant number of more recent tables feature the last two elements as lutetium and lawrencium instead." (p. 10)
Serving the largest audience
Scerri seems to go off-message in his conclusion, with references to:
- "…the format of the periodic table that serves the greatest number of periodic table users including students, instructors and practising chemists alike"; and
- "…how the periodic table is presented to the widest possible audience of users." (p. 15)
Surely the result of these notions will be a periodic table that attempts to cater to everyone but pleases no one?
Going off-message at this point is peculiar, since he earlier wrote:
- "It becomes increasingly clear that there may not be any such thing as one optimal table in a purely objective sense. The question seems to depend on what criteria are considered and, most importantly perhaps, on whether one favours chemical or physical criteria or general didactic considerations." (p. 12)
Accordingly, give me a tailor-made PT anytime, whether that is 14CeTh, 15LaAc, or 14LaAc, Adomah, AAE, Janet or some other variation, as long as the applicable context is set out.
The bugaboo of the split s-block
Scerri notes the periodic table is generally depicted with helium in group 18, and this splits the s-block (p. 11).
This is one of those things—the split s-block—that effectively all chemists (to a first approximation) do not lose any sleep over.
Same goes for the split d-block, which is less visible.
That said, better chemists keep both of these interesting aspects of the PT in mind.
It is like what Jones says:
- "Scientists should not lose sleep over the hard cases. As long as a classification system is beneficial to economy of description, to structuring knowledge and to our understanding, and hard cases constitute a small minority, then keep it. If the system becomes less than useful, then scrap it and replace it with a system based on different shared characteristics." Jones 2010, Pluto: Sentinel of the outer solar system, Oxford University Press, p. 171).
The anomalous first period
- "Another attractive feature of the left-step table is that it restores regularity and perhaps even balance to the otherwise awkwardly shaped traditional periodic table representation. More significantly than such aesthetic considerations, this table provides greater regularity in depicting every single-period length as repeating once as, 2,2,8,8,18,18,32,32. Meanwhile, the traditional table features an anomalous first period that, unlike all subsequent ones, does not repeat in length to give a sequence of 2,8,8,18,18,32,32. (p. 13)
I don't understand why the lack of repetition of the length of the first period is regarded as anomalous. There is no first principles derivation being breached here, as far as I know. It only means that, from a chemistry perspective, it is more meaningful to break the periods after the end of the noble gases. At the same time, the left step table is still good for its particular uses.
Thorium
- "Needless to say, the characterization of these blocks of the periodic table is only approximate, just as the assignment of electronic configurations to atoms represents an approximation. Moreover, one may readily concede that an element such as thorium does not actually possess any f-orbital electrons and yet it is classified as being among the f-block elements even in all four of the periodic table representations shown in figures 10 to 13.
--- Sandbh (talk) 00:44, 19 July 2020 (UTC)
I finally got around to reading this fascinating piece, and offer here my comments, which I am also emailing to Mr. Scerri.
- Page 2, section 2, paragraph 2, sentence 1: "an eight column" should be "an eighth column"
- Page 13, paragraph 1 & 2. In the phrases "clearly illogical situation" and "restores logic to the situation", it seems to me that this is not an issue of violating and restoring "logic" in the sense of deduction, but rather an issue of an "irregularity" violating a simplistic pattern which turns out to be perfectly "regular" according to a slightly more complex pattern.
- Page 13, paragraph 2, last sentence. In "completely redundant in Schwartz's view", the term "redundant" doesn't seem to be quite correct, at least to my ears. It isn't "redundant" in the sense of "repeating something else", but rather it is "superfluous" or "unnecessary". But perhaps this is merely a difference between my American dialect and a more British one where an employee becomes "redundant" when they are no longer needed, whether or not their work duplicates that of another employee.
- Page 18, paragraph 1, lines 1-2: " evolution spoils any hope ... bound to change as time evolves". I don't think anyone would contend that time itself "evolves". Rather, time passes by as other things may or may not change.
- General comment: Just as the 50-column table provides insight into the construction of a 32-column table, and the 50- and 32-column tables provide insight into the construction of an 18-column table, it might be interesting to apply insights from 50-, 32-, and 18- column table to consider what might be the "best" 8-column table, and then consider what its deficiencies might be that make an 8-column format impractical. This is treated in one of the footnotes; that might be all it deserves, but I would find a more extended discussion interesting.
— YBG (talk) 03:42, 1 August 2020 (UTC)
RFC: Should the default form of the periodic table be changed to put Lu and Lr in group 3, rather than La and Ac?
The following discussion is closed. Please do not modify it. Subsequent comments should be made on the appropriate discussion page. No further edits should be made to this discussion.
Should the default appearance of the periodic table on Wikipedia be changed to what it was in 2016, i.e. to have lutetium (71Lu) and lawrencium (103Lr) in group 3 rather than lanthanum (57La) and actinium (89Ac)? Or should it simply have markers below yttrium where all the lanthanides and actinides are supposed to go in? Double sharp (talk) 10:08, 20 July 2020 (UTC)
- Note for further clarification: this does not propose to treat any form as the only one, because that would contradict WP:NPOV. On this article we must of course discuss both forms and treat them neutrally but briefly, that goes without saying. I simply mean to discuss the appearance of our templates like
{{Periodic table}},{{Compact periodic table}}, the one in the infoboxes like{{infobox helium}}, etc. These normally appear as navigation aids in articles where the group 3 dispute is simply irrelevant, therefore unless someone has a better solution we seem to need to pick a default form for those. Double sharp (talk) 12:44, 20 July 2020 (UTC)
- Current RFC withdrawn for a last try at drafting something both Sandbh and I will find acceptable. Double sharp (talk) 08:51, 21 July 2020 (UTC)
The three forms
Lu and Lr below Y. The form I propose, and which we had before 2016.
18-column:
| H | He | |||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | |||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | |||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | |
| Cs | Ba | * | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | ** | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| * | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | ||||
| ** | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No |
32-column:
| H | He | ||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
La and Ac below Y. The current form.
18-column:
| H | He | |||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | |||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | |||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | |
| Cs | Ba | La | * | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac | ** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| * | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| ** | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
32-column:
| H | He | ||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
Markers below Y. The compromise form. IUPAC shows it on its periodic table, but they also plan to change it to one of the other forms. Which one has not been decided.
18-column:
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | * | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | ** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| * | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||
| ** | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
32-column (note the stretching of scandium and yttrium necessary):
| H | He | ||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
Background information by Double sharp
I spent some time making this issue understandable for any bystanders who may want to join in, helped by feedback from User:Dreigorich. As such, here is an explanation in the form of a dialogue. I have tried to keep answers very short. (The first version was a huge wall of text. Now it is not.)
The issue had its most recent reignition on Wikipedia in December 2019 and raged on through Wikipedia talk:WikiProject Elements/Archive 42, Wikipedia talk:WikiProject Elements/Archive 44, Wikipedia talk:WikiProject Elements/Archive 46, and until a few days ago Wikipedia talk:WikiProject Elements. I say "until a few days ago" because that's when I gave up the endeavour as fruitless: nobody seems about to have a change in opinion, and the arguments are raging in circles, and civility has been thrown out the window (and I realise now that I'm partly responsible for that, which I'm sorry for).
At that point we had exactly, among eight participants:
- one (1) editor in favour of La (Sandbh);
- five (5) in favour of Lu (myself, Droog Andrey, Officer781, Dreigorich, ComplexRational);
- and two (2) who at least found the arguments being made in favour of La questionably convincing (DePiep, R8R).
Since the issue was previously decided in an RFC back at Template talk:Periodic table, discussion between me and R8R concluded that going to another RFC would be the best way to propose it be changed back to how it was before the first RFC. Given that that RFC was over three years ago, and one of the main La proponents in it was me, and I've learnt enough new information since then that makes me think that the arguments I was propounding were wrong-headed, I think starting a new RFC is also most reasonable.
In theory one should get a neutral editor to write the background information. However, the problem is that this issue has something in common with many of the disputes at Wikipedia:Lamest edit wars. It is impossible to find a neutral editor to write background information because as soon as she looks at the issue enough to understand the background information, she takes a side herself and is no longer neutral. That is quite unusual given that Wikipedia:Lamest edit wars does not have a category for natural science, much less chemistry. Indeed, the main positive difference between the issue here and the ones over there is that so far there has not been any actual edit warring. Some uncivil remarks when people on either side get fed up, yes, and I am sorry that I have made some of them. But, thankfully, no edit warring.
So I wrote it. Even if I obviously have an opinion and am obviously supporting it with arguments here, I'm at least referring to reliable sources focusing on the issue to do it. If that is still not suitable, then this may at least be considered as a long explanation of my !vote rather than background information.
Questions 1 to 5
Q1: What are you arguing about?
Well, it turns out that somewhere near the bottom-left of the periodic table there's a bit of a dispute about which elements should go where.
Q2: Really? But I thought the periodic table was ancient history chemically speaking.
Yes, Mendeleev's first table dates from 1869. We still haven't sorted everything out, though. There are a bunch of elements that are rather difficult to place.
Q3: That sounds interesting. Tell me more.
In the words of Eric Scerri, "there's trouble at the beginning, middle and end of the periodic table". This RFC is about the middle: the rare earth elements.
Q4: So are you going to tell us what it is already?
It comes down to whether lutetium (Lu) or lanthanum (La) is a better fit for the position under yttrium (Y) in the periodic table. There's also a third form which has all fifteen lanthanides under yttrium, which we did have on WP a while ago, but not anymore.
Q5: Why is this so terribly important? If an element fits well in two places, why not duplicate it in both positions?
This is not generally done by reliable sources, except maybe when making a point. Therefore on Wikipedia we should not start doing it.
Questions 6 to 10
Q6: What about the "markers below yttrium" form? Isn't it a reasonable compromise?
It looks like one, but it is also a side in itself.
I do not support it for two reasons. One is technical. We sometimes use 32 column templates because they fit better as footers, e.g. {{compact periodic table}}, and then this approach becomes really problematic: scandium and yttrium have to stretch to cover 15 columns.
The other is chemical. This form claims that the f-block is a degenerate branch of the d-block and thus by extension that the f orbitals are a part of the valence core, which is simply false for almost all the elements involved (all but Lu and Lr, in fact). Every redox-capable f element (which is almost all of them) is obviously using its f electrons, and even those which are not seem to have some f contribution anyway (see Q22). See Jensen for a discussion. Maybe all the lanthanides are similar enough to fit below yttrium, but can anyone really claim that for the actinides?
Q7: Well, shouldn't we follow NPOV and show all three options all the time then?
That makes sense, and is what I support, when the group 3 controversy is the topic at hand. But usually it is not. Then you have to pick one to avoid going off on a tangent which is not relevant. Below I argue for Lu under Y.
For reasons of brevity, I will hence forth shorten the options' names to:
- Lu table: Lu and Lr below Y.
- La table: La and Ac below Y.
- * table: Markers * and ** below Y.
Q8: The things are called lanthanides and actinides. Shouldn't lanthanum and actinium continue to be used as the placeholders?
They're not placeholders. The * table and La table are different. Moreover, the whole point of the categories "lanthanide" and "actinide" is that those elements are similar to La and Ac. It seems quite weird to put La and Ac away from the rest of them. It is less weird for Lu (whose properties among the lanthanides are furthest from those of La) and not weird at all for Lr (which is quite unlike the other late actinides Es-No in properties).
Q9: IUPAC is the relevant source here, surely? What do they have to say?
They show their table as a * table, but they also state:
| “ | While IUPAC has no recommendation for a specific form of the periodic table, i.e. 18-column or 32-column format, the version here presented is in the conventional long form and is yours to use. | ” |
| — IUPAC Periodic Table website | ||
So it is not clear if that is an actual IUPAC recommendation for the * form or not.
IUPAC also organised a project to resolve precisely this issue, as can be seen from number 7 on their periodic table page. That project considers the La and Lu alternatives, but not the * one that they currently show. It started in 2015 and still has not concluded anything. Since it has both strong La and Lu proponents on it I also doubt it will conclude anything soon.
All we can conclude for now is that they seem to intend to deprecate the * form they currently show, thus leaving us without a firm guide in the meantime.
Q10: Why aren't we waiting for them then?
Because (1) I doubt that their work will be finished anytime soon, (2) we don't always follow IUPAC (we follow it for spelling aluminium but not for phosphane), and (3) any way you look at it, we have to show something in the meantime, see Q7.
I argue that we should look at the consensus of reliable sources that focus on the matter. Because an article focusing on group 3 and explaining why the authors think La or Lu should be there, I argue, outweighs in terms of reliability somebody who just draws a periodic table and gives no clue as to why he or she has drawn it that way. I claim that's in favour of Lu.
Questions 11 to 15
Q11: What do most reliable sources say?
Depends really. If you do a Google Image survey on "periodic table", * below Y dominates with proportions La:Lu:* of 1:1:4. The IUPAC project organised a survey that seems to show that La below Y dominates in textbooks (with La:Lu:* proportions of 4:1:1). However usually those textbooks have * in the same cell as La with a group 3 header above, therefore also implicitly claiming that the other lanthanides go there. So there is, at least, an ambiguity. Also, all these dominances are just 2/3 majorities, it's not a rare thing at all to find a textbook or periodic table poster giving Lu below Y.
I claim it's more relevant to look at what articles specifically focusing on this issue tell us, because it's common for textbooks to repeat standard errors on issues like this (e.g. claims that d orbitals are involved for phosphorus in PCl5, which are false but ubiquitous in textbooks), and most of these textbooks are hardly focusing on these heavy 4f and 5d elements that the dispute is all about. Those have a majority supporting the Lu form. I tallied them with Sandbh in an old submission to IUPAC we made on this matter in 2016; I've updated the list to 2020 below. Back then we agreed that La is better; then I changed my mind in response to new evidence from Droog Andrey that Lu is better, so we started arguing.
| Advocates Lu | Advocates La | Advocates * |
|---|---|---|
| Bury (1921) Shemyakin (1932) Landau and Lifshitz (1958) Hamilton (1965) Merz and Ulmer (1967) Chistyakov (1968) Mathias (1971) Wittig (1973) Jensen (1982 and again 2015) Holden (1985) Fang et al. (2000) Horovitz and Sârbu (2005) Wulfsberg (2006) Ouyang et al. (2008) Scerri (2012) Nelson (2013) Settouti and Aouragi (2014) Alvarez (2020) |
Smith (1927) Trifonov (1970) Shchukarev (1974) Atkins (2006) Lavelle (2008) Restrepo (2017) Cao et al. (2020) |
Xu and Pyykkö (2016) |
Q12: Aren't you essentially claiming that a whole lot of eminent scientists are wrong?
It's a controversy. No matter which side you pick you're going to be claiming that a whole lot of eminent scientists are wrong. La advocates like Sandbh have Sergey Shchukarev on their side, Lu advocates like me have Landau and Lifshitz, so we can see that both options have had respected adherents. That's why I claim we have to look at the articles focusing on this issue and also consider the chemistry involved.
Q13: Many chemists have never heard that there was such a debate. Are you sure you're not just another fringe theory peddler?
Landau and Lifshitz are hardly "fringe". Neither is Clayden et al.'s Organic Chemistry, which has a Lu table. And Jensen, before his 1982 article, had not heard of this debate either.
Q14: What is the status of that interminable thread at Wikipedia talk:WikiProject Elements, stretching from last December to this July?
We have 5-1 for Lu and an extra two who at least think the La arguments proffered are problematic.
- Well, I guess it goes without saying that I support the Lu form, yes?
- "La arguments are totally local, while Lu arguments are pretty regular. That exactly matches Ptolemy vs. Copernicus. The history just repeats itself. Nothing more to say." - Droog Andrey
- "Cool, thanks. This makes perfect sense. Well said. Team Lu for me!" - Dreigorich
- "I'd really just want to get rid of the Scandium/Yttrium overhang in the long periodic table. It is ugly and very artificial IMO." - Officer781
- "I am more convinced by the Lu arguments I've read, and given the support of the scientific community and lack of consensus against it here (no "battleship" as you describe), I'll join the RfC once it's underway." - ComplexRational
- "Even worse, a publication may be biased towards a preference for more simple or elegant or 'by authority' (IUPAC) form, which is a beauty contest not science. I understand Sandbh does argue for such criteria seriously, but that still does not convince me—FWIW." - DePiep
- "I have noted in the very beginning of this discussion, which started with an article you [Sandbh] wrote, that it did not appear to me that pro-La-Ac and pro-Lu-Lr arguments were given the same weight, and I said, perhaps not as explicitly but to the same meaning, that it looked like this was done so deliberately so that one option is favored over the other. I am afraid that what I've read so far reinforces this thinking within me." - R8R
versus a single one:
- "Ahead of the recommendation of the IUPAC project I see no case for change." - Sandbh
However since we carried out the last change in an RFC, I feel (after discussion with R8R) that a proposed reversal should also go through an RFC. It seems only fair.
Q15: Have you addressed Sandbh's arguments there?
Yes, I responded to everything new he raised. Though not always in the most polite way after a few months of arguing, for which I am sorry.
Questions 16 to 20
Q16: Exactly why were you arguing this fiercely with Sandbh?
Well, he was writing an article, to be published in Foundations of Chemistry, that supports La under Y, and brought it to WT:ELEM for peer review last December. I and Droog Andrey disagreed with his arguments on what seemed to me to be logical grounds. Eventually it got to this:
| “ | When you get as old as me, you'll learn there is much more to the world than logic. ...
You keep asking for a falsifiable theory. There isn't one! |
” |
| — Sandbh, 07:49, 12 May 2020 (UTC) | ||
I found this quite surprising and responded in a rather too harsh manner, which I'm sorry for. Nevertheless I still cannot agree with this approach. Unless we use logic as a base, I don't see how we can judge the strength of arguments.
Q17: So why did it last so long?
Arguing with him was very helpful to sharpen my own understanding. But let's be honest: "someone is wrong on the Internet" also had something to do with it on my part, and I should really wean myself off of that.
Q18: But you've not been a complete angel yourself dealing with him.
That is true. I'm sorry. I will undertake to be more civil in future.
Q19: What's the history of this neverending argument anyway?
Mendeleev more or less gave up on placing the lanthanides, mostly leaving it to his Czech colleague Bohuslav Brauner. After some other attempts, eventually a form with all lanthanides under yttrium (basically the * form) was settled on. However even during this time tables that quite clearly don't put La under Y had already appeared (Henry Bassett 1892, Alfred Werner 1905). Actually Werner's form (yes, that's the father of coordination chemistry we're talking about) is incredibly close to the modern 32-column Lu form (with the exception that Be and Mg go over Zn instead of Ca). The only reason I do not call it a Lu table is because Lu had not yet been discovered(!).
In the 1920s and 1930s some chemists assigned Lu under Y because the chemistry of Y is more similar to Lu than La (Sc, Y, and Lu separate in the yttrium group of rare earths, but La and Ac in the cerium group).
The current dominance of the La form seems to stem, according to Jensen, from wrong electron configurations from the 1940s in which the lanthanides were assumed to be fn-1ds2 instead of fns2. These were later corrected, but by that time it seems the La form had stuck. We should not forget that these electron configurations are for the gas-phase ground state and are not really relevant for chemistry anyway (in chemical environments they change).
Meanwhile articles supporting Lu have appeared since 1921 and have been going on until the present; there have not been as many articles supporting La. Some authors have changed their periodic tables, others have not, there is still no consensus. IUPAC started a project in 2015 to look into it, they still have not made a decision.
It is possible that things will change. Be-Mg-Zn was present in Werner's periodic table of 1905, B-Al-Sc was once shown by Pauling(!), and prior to Seaborg it was generally accepted that the actinides known (Ac, Th, Pa, U) form a fourth d block row with uranium being eka-tungsten. Now you don't see any of these.
Q20: Should tradition count for something here?
There's not much of a tradition when chemists still argue about it. I prefer that we take the option that most of them who really analyse the issue support when we're not discussing the issue.
Questions 21 to 25
Q21: Would it not simply work if we just added footnotes like for helium where we explain that the other option is also common?
For the infobox, for the footer, you do not want to emphasise the issue where it is irrelevant, e.g. for the article on helium where group 3 matters not at all. The "overhang" the La table has in the 32 column form draws the eye's attention. The "stretchy scandium and yttrium" of the * table draws even more of the eye's attention. In order to make them look good, you have to use an 18 column instead of a 32 column form, which strikes me as not terribly good considering that those are supposed to be the same thing (just with the footnote glued in or not). So for these purposes I claim the Lu table is better: it preserves the symmetry. Moreover there is even scientific basis for preserving that symmetry, as the periodic table is explained by the valence orbitals and thus quantum mechanics, the Madelung rule having derived in the 1950s by Klechkovsky already.
Of course, whenever it is relevant (i.e. article of lanthanum, article of lutetium, and the period 7 congeners), we will add a note like there is for helium (about helium over beryllium being supported by some chemists). That goes without saying, for WP:NPOV reasons. But I think the reader would be better served with a Lu under Y default for when the situation does not matter.
Q22: OK, so are you going to get on to the scientific basis of this change or not?
Here are eight short arguments and the evidence behind them.
- Per Jensen (1982) Lu under Y matches the trends in the d block better.
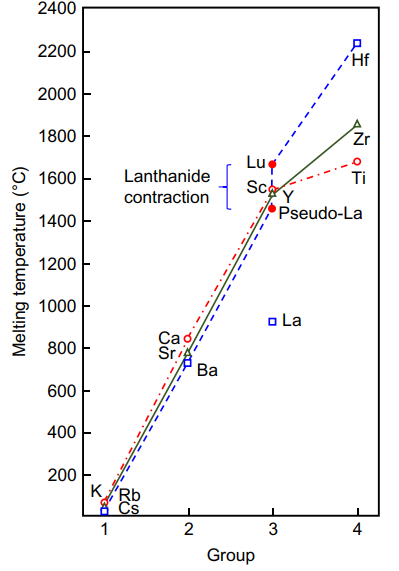
- Per Gschneidner (2016) La has 4f involvement in the metal that impacts its melting point, making it 450 °C lower than expected. The figure to the right from his paper shows "pseudo-La" for the expected melting point of La if it didn't have this 4f hybridisation. Such 4f involvement also ties together lots of properties of lanthanum: it explains melting point, heat of sublimation, high coordination numbers around 12 (difficult to explain without f orbitals), cubic molecular geometries of lanthanum complexes (difficult to explain without f orbitals giving the symmetry), superconductivity (it is superconductive, but Sc, Y, Lu are not), quite possibly also its crystal structure (which matches the early f elements but is different from Sc, Y, and Lu). Putting La under Y misses the point that the first element for which the 4f orbitals are non-hydrogenic and can participate chemically is lanthanum, not cerium.
- Lutetium has no involvement of the f electrons in chemistry. All they do is contribute incomplete shielding effects which is exactly like the effect of the lanthanide contraction on hafnium through mercury. On those grounds it is surely a d element.
- Properties of yttrium are closer to those of lutetium than lanthanum.
- The only precedent for taking an element out of its block is helium, which is obviously much closer in properties to neon than beryllium. Why do that for lanthanum when yttrium is already closer to lutetium in all ways?
- Taking La out of the f block on the grounds of its ground-state gas-phase electron configuration lacking a 4f electron is irrelevant (these configurations change in chemical environments) and inconsistent with thorium. Thorium is in the f block, yet its ground-state gas-phase configuration lacks a 5f electron. Besides the periodic table is not even based on ground-state gas-phase electron configurations in the first place, as evidenced by the fact that no one rips nickel [Ar]3d84s2, palladium [Kr]4d105s0, and platinum [Xe]4f145d96s1 apart from each other. Rather it is based on the number of valence electrons and which orbitals they may enter across all chemical environments.
- Examining properties reveals that La fits well with f elements, but is an outlier in the d elements. Same is true for Ac. Lu is OK either way (mostly), but Lr seems to fit well in the d elements but is an outlier in the f elements (we mostly only have predictions for that one of course).
- When examining properties, Eu and Yb show the properties expected and known from Mn and Zn (half-filled and filled subshell); Gd and Lu do not. That supports La-Yb as the f block. In fact a lot of analogies with the d block strongly support La-Yb as the f block for exactly this reason (standard electrode potentials, electronegativity, ionisation energies, melting and boiling points).
Everything said of the lanthanides above is also true or suspected of the corresponding actinides.
Evidence
For the 7th period elements no one has ever made more than a few atoms of, calculated properties have been used.

In general properties of Sc-Y-Lu match the d block trend better. Sc-Y-La matches the s block trend, but remember that group 3 is a d block group, and the s block groups are in fact anomalous: they break the trend of having no even-odd periodicity. (Even-period elements usually are more electronegative and more oxidising in higher oxidation states; odd-period elements act the opposite way. From 5d onwards relativistic effects make it not quite right, as 5d is electronegative but prefers higher oxidation states, and 7p is electropositive but prefers lower oxidation states. But mostly it is still correct as a generalisation.) That's because there's no contraction:
1s period I 2s 2p period II 3s 3p period III 4s 3d 4p period IV 5s 4d 5p period V 6s 4f 5d 6p period VI 7s 5f 6d 7p period VII
From the build-up of the periodic table we see that for non-s blocks, even periods either have the first orbital of a given angular momentum (which is smaller than expected), or they have suffered a new contraction that wipes out the expected increase of radius and basicity (3p-4p increase wiped out by 3d, 4d-5d increase wiped out by 4f, 5p-6p increase wiped out both by 4f, 5d, and relativistic contraction of 6s). The s block is an exception: it always sits right above the noble gas core and never has any incomplete shielding effects (except a tiny bit for 2s vs 3s, and the obvious massive exception of 1s). There's no reason why any other group should follow them.
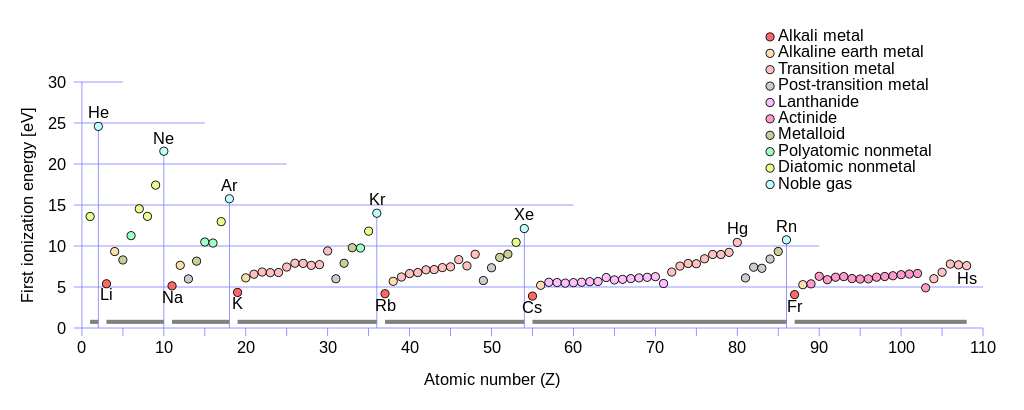
1st ionisation energies of the elements. Lu and especially Lr fall off the trends of the lanthanides and actinides respectively, and fit better with the trends of the succeeding 5d and 6d transition metals.

3rd ionisation energies of the transition elements (makes sense, having ionised the s electrons away already, and now probing the energies to remove a d or an f electron). This is not perfect, but when the configuration is "wrong", the "right" one is usually so close that it doesn't matter. La2+ is [Xe]5d1, but the expected [Xe]4f1 is about 0.89 eV up in energy; Gd2+ is [Xe]4f75d1, but the expected [Xe]4f8 is about 0.30 eV up in energy. I hope we agree that this is small potatoes compared to the differences between neighbouring elements.
Natural families La-Eu and Gd-Yb are supported, following Sc-Mn and Fe-Zn; the half-full and full subshell gives a local maximum in energy, because the next electron going in is either paired (after half-full) or in a higher-energy subshell (after full) and is then easier to remove. The effect weakens for higher rows (as you can see from the 1st IE chart as well; N-O is an obvious blip downward, P-S is not so obvious, by Sb-Te it doesn't go down anymore), but you can still see it.
| property | La | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg |
|---|---|---|---|---|---|---|---|---|---|---|---|
| m.p. (K) | 1193 | 1925 | 2506 | 3290 | 3695 | 3459 | 3306 | 2719 | 2041.4 | 1337.33 | 234.43 |
| b.p. (K) | 3737 | 3675 | 4876 | 5731 | 5828 | 5869 | 5285 | 4701 | 4098 | 3129 | 629.88 |
| specific heat capacity (J/(g*K)) | .195 | .154 | .144 | .14 | .132 | .137 | .13 | .131 | .133 | .129 | .14 |
| EN (Pauling) | 1.1 | 1.27 | 1.3 | 1.5 | 2.36 | 1.9 | 2.2 | 2.2 | 2.28 | 2.54 | 2.0 |
| EN (Kulsha-Kolevich) | 1.11 | 1.31 | 1.38 | 1.46 | 1.54 | 1.55 | 1.67 | 1.75 | 1.84 | 1.93 | 1.81 |
| Density | 6.145 | 9.84 | 13.31 | 16.654 | 19.25 | 21.02 | 22.61 | 22.56 | 21.46 | 19.282 | 13.5336 |
| Young's modulus | 36.6 | 68.6 | 78 | 186 | 411 | 463 | ??? | 528 | 168 | 78 | ??? |
| Bulk modulus | 27.9 | 47.6 | 110 | 200 | 310 | 370 | 462 | 320 | 230 | 180 | 25 |
| Resistivity (nΩm, close to r.t.) | 615 | 582 | 331 | 131 | 52.8 | 193 | 81 | 47.1 | 105 | 22.14 | 960 |
| Brinell hardness (MPa) | 350-400 | 893-1300 | 1450-2100 | 441-3430 | 2000-4000 | 1320-2500 | 3920-4000 | 1670 | 310-500 | 188-245 | ??? |
| Heat of fusion (kJ/mol) | 6.20 | 22 | 27.2 | 36.57 | 52.31 | 60.43 | 57.85 | 41.12 | 22.17 | 12.55 | 2.29 |
In most properties Lu matches the properties of the 5d elements better than La. That is also true for chemical properties (Lu is less basic and less big as a cation, matching 5d elements better), but those are harder to show as data. ^_^
| property | Ac | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn |
|---|---|---|---|---|---|---|---|---|---|---|---|
| m.p. (K) | 1323 | 1900 | 2400 | ??? | ??? | ??? | ??? | ??? | ??? | ??? | 283 |
| EN (Kulsha-Kolevich) | 0.97 | 1.29 | 1.34 | 1.41 | 1.49 | 1.59 | 1.72 | 1.83 | 1.92 | 1.99 | 1.91 |
| Density (g/cm3) | 10.07 | 15.6 | 23.2 | 29.3 | 35.0 | 37.1 | 40.7 | 37.4 | 34.8 | 28.7 | 14.0 |
| 1st IE (kJ/mol) | 499 | 470 | 580 | 665 | 757 | 740 | 730 | 800 | 960 | 1020 | 1155 |
| 3rd IE (kJ/mol) | 1900 | 2228 | 2300 | 2378 | 2484 | 2570 | 2830 | 2900 | 3030 | 3080 | 3160 |
The little known and predicted about Ac vs Lr-Cn seems to show the effect is even stronger. All bulk properties of Lr-Cn are of course predictions as nobody has ever made enough. Kulsha-Kolevich electronegativity was used because no other scale actually seems to have values for elements as heavy as copernicium.
| property | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1st IE (kJ/mol) | 499 | 587 | 568 | 598 | 605 | 585 | 578 | 581 | 601 | 608 | 619 | 627 | 635 | 642 | 470 |
| 3rd IE (kJ/mol) | 1900 | 1978 | 1814 | 1900 | 1997 | 2084 | 2132 | 2026 | 2152 | 2267 | 2334 | 2363 | 2470 | 2643 | 2228 |
| m.p. (K) | 1323 | 2115 | 1841 | 1405 | 917 | 913 | 1449 | 1613 | 1259 | 1173 | 1133 | 1125 | 1100 | 1100 | 1900 |
| EN (Kulsha-Kolevich) | 0.97 | 1.01 | 1.04 | 1.06 | 1.08 | 1.12 | 1.07 | 1.18 | 1.22 | 1.27 | 1.32 | 1.36 | 1.39 | 1.37 | 1.29 |
| Density (g/cm3) | 10.07 | 11.72 | 15.37 | 18.95 | 20.45 | 19.84 | 13.69 | 13.51 | 14.79 | 15.1 | 8.84 | 9.7 | 10.3 | 9.9 | 15.6 |
Lawrencium's properties, as far as they are predicted, seem to be a quite bad match for the late actinides. (Data for Fm through Lr bulk properties is predicted, no one has ever made enough.)
| M | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| mp (°C) | 842 | 1541 | 1668 | 1910 | 1907 | 1246 | 1538 | 1495 | 1455 | 1085 | 420 | 30 | 938 |
| bp (°C) | 1484 | 2836 | 3287 | 3407 | 2482 | 2061 | 2861 | 2927 | 2730 | 2562 | 907 | 2400 | 2833 |
| M | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| mp (°C) | 727 | 920 | 795 | 935 | 1024 | 1042 | 1072 | 826 | 1312 | 1356 | 1407 | 1461 | 1529 | 1545 | 824 | 1652 | 2233 |
| bp (°C) | 1633 | 3464 | 3443 | 3130 | 3074 | 3000 | 1900 | 1529 | 3000 | 3123 | 2567 | 2600 | 2868 | 1950 | 1430 | 3402 | 4603 |
Double periodicity in melting and boiling points likewise supports La-Eu and Gd-Yb as natural f-block subfamilies. (When we get to a half-filled or filled subshell, delocalisation of those electrons becomes less favourable.) The effect is weaker for succeeding rows as usual.
| M | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| M3+/M2+ potential | very high | −2.3 | −0.9 | −0.255 | −0.42 | +1.56 | +0.771 | +1.92 | +2.3 | +2.4 | very high | −0.8 |
| M | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In |
| M3+/M2+ potential | very high | −2.8 | ??? | −0.9 | −0.2 | +0.3 | +0.24 | +0.7 | ??? | +1.8 | very high | −0.49 |
| M | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| M3+/M2+ potential | very high | −3.1 | −3.2 | −3.1 | −2.7 | −2.6 | −1.55 | −0.35 | −3.9 | −3.7 | −2.6 | −2.8 | −3.0 | −2.2 | −1.05 | −2.7 |
| M | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
| M3+/M2+ potential | very high | −4.9 | −4.9 | −5.0 | −4.7 | −4.7 | −3.5 | −2.3 | −3.7 | −2.8 | −1.6 | −1.3 | −1.1 | −0.1 | +1.4 | −2.6 |
The standard reduction potential trends across the two subfamilies La-Eu and Gd-Yb are smooth, like those of Sc-Mn and Fe-Zn; they reach maxima at the half-filled and filled subshell elements as expected as then it is more difficult to ionise an extra electron past the half-filled or filled subshell. Same goes for 3rd ionisation energies, and same goes for melting and boiling points (more difficult to delocalise those electrons).
We use +2 oxidation states as a baseline to compare like with like: this way the s electrons are ionised, the configurations involved are (almost always!) dn and fn, when they are not the difference in energy to that is usually small enough to ignore, and we can probe the stabilising effect of the real half-filled and filled shell. Of course you may artificially shift to the +3 state and get different results, but it has about as much meaning as plotting 2nd ionisation energies to "prove" that the periods should go from group 2 to group 1:
| element | He | Li | Be | B | C | N | O | F | Ne | Na | Mg | Al | Si | P | S | Cl | Ar | K | Ca |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2nd IE (kJ/mol) | 5250.5 | 7298.1 | 1757.1 | 2427.1 | 2352.6 | 2856 | 3388.3 | 3374.2 | 3952.3 | 4562 | 1450.7 | 1816.7 | 1577.1 | 1907 | 2252 | 2298 | 2665.8 | 3052 | 1145.4 |
I hope we can agree that this is self-evidently absurd.
Conclusion
For these reasons I claim that the La form has no scientifically sound basis. It is not a good chemist's periodic table because yttrium is closer to lutetium than lanthanum, and because lutetium and lawrencium are far more like transition metals than lanthanum and actinium. It is not a good physicist's periodic table, because it goes away from the blocks that underlie the periodic table. And clearly teaching the Madelung rule is going to be pedagogically better than teaching the Madelung rule and then giving an exception for La that doesn't even match reality, given its 4f involvement.
This matches the criteria for putting elements in the periodic table as stated by Jensen, particularly arguments 2 and 3 (4f in La and its complete lack in Lu).
| “ | In actual fact, when assigning an element to a position in the periodic table the above factors are not simply applied as a haphazard empirical blend or combination, rather they are applied in a strictly hierarchical order, consisting of four steps or stages (Jensen 2009):
|
” |
| — William B. Jensen (2015), The positions of lanthanum (actinium) and lutetium (lawrencium) in the periodic table: an update | ||
Q23: Are there any other La arguments that you haven't refuted above?
- Differentiating electrons
Sandbh seems to believe in the importance of the "differentiating electron" of an element, which is the electron that differentiates it in its ground-state gas-phase configuration for the previous element. For example, since titanium is [Ar]3d24s2 (ground-state gas-phase), and vanadium is [Ar]3d34s2 (same), vanadium has a 3d differentiating electron.
I do not see why this should be important, given that we are not generally in the business of building elements up by adding one proton and one electron from the previous one. Also differentiating electrons are for the gas phase, and we've already discussed above that this is not relevant for chemistry because atoms usually have a different configuration when chemically bound from what they have sitting alone by themselves. Neither is it that clear all the time what exactly the differentiating electron ought to be. Passing from vanadium [Ar]3d34s2 to chromium [Ar]3d54s1 we have added two 3d electrons and subtracted one 4s one.
But all right, suppose we grant that. A usual argument Sandbh gives for why this should supposedly be important is looking at silver. It has a 5s differentiating electron as opposed to the 5d one of its heavier homologue gold, and this supposedly explains why the chemistry of silver is more main-group-like than gold:
| “ | [Double sharp has argued that] Differentiating electrons make no difference to the chemistry of the elements and are therefore irrelevant. In fact, a counterexample is silver, the d/e of which is expected to be a d-type, but in fact turns out to be an s-type. Silver then acts predominately as a main-group metal rather than a transition metal. ... --- Sandbh (talk) 03:01, 15 July 2020 (UTC) | ” |
On the other hand, technetium also has a 5s differentiating electron as opposed to the 5d one of its heavier homologue rhenium. Judging by how happy technetium is to interconvert between its many oxidation states like the transition metal it really is, this does not seem to do anything at all to make it more main-group-like at all.
- Group divides
If you look at KCl, CaCl2, ScCl3, and TiCl4, the first three are ionic solids and the last one is a volatile liquid. So group 3 has to be cleft from group 4.
I used to believe this, but it doesn't really work. We may equally well observe the melting points of Cs2O, BaO, La2O3 or Lu2O3, HfO2, Ta2O5, WO3, Re2O7, OsO4. These are all high melting-point compounds until WO3, and the melting points keep increasing (showing ionic character) until HfO2. And we can multiply other counterexamples like Na2O, MgO, Al2O3, SiO2 (melting points increase till MgO).
Any way you look at it, this doesn't support any natural group divide at all. Mostly because there is no such thing outside the fundamental one (noble gas | alkali metal).
A related argument states that group 3 is more similar to group 2 than group 4 because group 3 acts like a trivalent version of the main group metals. But for group 4 you can find aqueous cations with noble gas configurations that make them like a tetravalent version. Simply make the pH low enough. −1 will do, you'll easily find things like Zr4+ and Hf4+, maybe even Ti4+, then. Well, look at Fajans' rules again. Cation polarising power, hence acidity, depends on charge and radius. Following Gary Wulfsberg's Principles of Descriptive Inorganic Chemistry, the usual dependence is on charge squared divided by ionic radius. We can then draw his table:
| Z2/r ratio | Pauling EN | Category | pKa | Examples |
|---|---|---|---|---|
| 0.00–0.01 | <1.8 | Nonacidic cations | 14–15 | Most +1 ions in s block |
| 0.01 | >1.8 | Feebly acidic cations | 11.5–14 | Tl+ |
| 0.01–0.04 | <1.8 | Feebly acidic cations | 11.5–14 | Most +2 ions in s, f blocks |
| 0.01–0.04 | >1.8 | Weakly acidic cations | 6–11.5 | Most +2 ions in d block |
| 0.04–0.10 | <1.8 | Weakly acidic cations | 6–11.5 | All +3 ions in f block |
| 0.04–0.10 | >1.8 | Moderately acidic cations | 1–6 | Most +3 ions in d block |
| 0.10–0.16 | <1.8 | Moderately acidic cations | 1–6 | Most +4 ions in f block |
| 0.10–0.16 | >1.8 | Strongly acidic cations | (−4)–1 | Most +4 ions in d block |
| 0.16–0.22 | <1.8 | Strongly acidic cations | (−4)–1 | |
| >0.16 | >1.8 | Very strongly acidic cations | <−4 | |
| >0.22 | <1.8 | Very strongly acidic cations | <−4 |
And indeed, we find that while the group 4 cations are indeed strongly acidic, they are not so acidic that they react irreversibly with water: the pKa is not quite low enough. Sure, you'll soon get a precipitate if their cations are introduced to aqueous solution at all but pretty low pH. But you can redissolve that precipitate in concentrated hydrochloric acid, at least if it hasn't aged too much. It's cations like Nb5+ and Ta5+ that are "very strongly acidic": too acidic to exist in water at all. So, apparently the group divide is really between groups 4 and 5 instead.
But probably the clearest way to rebut this whole idea is to look at what happens in the p block. Group 13 metals Ga, In, Tl can form aqueous cations in their group oxidation state. Group 14 metals Ge, Sn, Pb cannot, they are too acidic. (Remember what I said before? Elements in higher oxidation states are more electronegative and hence more acidic, whence how Pd2+ is more acidic than Be2+.) So this seems to also support B-Al-Sc if you apply it consistently. I think that is not a good sign.
The chemistry of group 3 is not more similar to that of group 2 to group 4, so that justification is also out. Observe the aqueous chemistry of group 4 and the organometallic chemistry of group 3. Not to mention that the standard "transition metal" property everyone knows from high school is forming coloured paramagnetic compounds with incomplete d orbitals. If you think scandium should be disqualified, because you will have a tough time finding stable lower-oxidation state compounds of Zr-Hf-Rf in group 4 and Nb-Ta-Db in group 5, those suddenly are also in danger of being disqualified. And as for the other s-block property of forming hard, class-A cations – the elements up to group 6 show that. Everything is a continuum here, and transition properties are no exception: they go in slowly, they come out slowly. All peripheral groups in blocks have properties similar to their block neighbours, there's no reason why to emphasise the property this much for group 3.
- Ions
According to this argument, lanthanum cannot start the f block because La3+ fails to have an f electron, and there is complete regularity down Ce3+ to Lu3+ in showing f1 to f14. But such a common stable oxidation state for everybody only exists in the f block, and actually only the 4f elements, not the 5f ones. So this argument is about as local as you can get and doesn't work as part of the periodic law.
I also note that by this logic La cannot be in the d block either, since La3+ is d0. In fact it cannot be anywhere in the periodic table either, since La3+ is also s0 and p0. And there are no s block elements either except helium (the least s-block-like of them), because in their common oxidation states (+1 for the column H-Li-Na-K-Rb-Cs-Fr, +2 for Be-Mg-Ca-Sr-Ba-Ra) they are all s0. Why should we use criteria that only apply to one block?
Finally, La had been [Xe]4f16s2, all this argument would surely never have started: a La-Yb f block (i.e. a Lu table) would be absolutely standard. But it would still lose the f electron in the +3 state anyway, so this argument would still argue for Ce-Lu anyway. That seems hard to accept.
- A Pair Out of Place
According to this argument, advanced by Lavelle, La and Ac cannot be put in the f block because they would represent the only case of two elements going into a block with no outer electrons in common with their block.
Of course, this is predicated on the gas-phase ground-state configurations, which is not that relevant. Also, how does this deal with Lu and Lr? Neither of them have any outer f electrons, they also form a pair out of place. So the argument is as much for the Lu table as it is for the La table.
Q24: Weren't you on the other side of this last time?
Indeed. I learnt more, I changed my mind. All thanks to Droog Andrey (= A. V. Kulsha). You may enjoy the periodic table poster he and one of his colleagues created too (link is to version of 2019). ^_^
Q25: So what's wrong with the old arguments that you were using then?
I've addressed them all above.
Questions 26 to 30
Q26: So how do I know you won't be fervently arguing to change it back again in four years?
I don't, that's because I believe in the scientific method. If I find out something falsifying my stand, I change my mind. You can argue it if you want... ;)
Q27: Why now? Why not wait for IUPAC?
It's not possible to be neutral. Sc-Y-* is also a side. I also think Sc-Y-* is chemically just weird (those f electrons are not core electrons, and while asking La-Lu to share one flat makes some sense, it's really weird for Ac-Lr), and Jensen agrees.
Q28: Why start it now and not earlier?
Because my ideas about this were not clear enough till this round of argument with Sandbh started. Now they are.
Q29: Do you have nothing better to do than write long screeds here about this?
Well, at R8R's suggestion it is now behind a collapse box, and most of the long screeds have been replaced by very short answers.
Q30: Do you have anything against Sandbh personally?
No, I just find myself in disagreement with his stand on logic and falsifiability.
Mass summoning
@ComplexRational, DePiep, Droog Andrey, Officer781, R8R, Sandbh, Дрейгорич, AzaToth, Tazerdadog, Maproom, CuriousMind01, Silvio1973, and Timtempleton: Pinging all megathread and previous RFC participants. And also AnthonyDu0122 who raised it above. ^_^
Unacceptable behaviour
I object, in the strongest possible terms, to the way Double sharp initiated this RFC.
I allege that Double sharp's background to the RFC is biased; incomplete; out-of-context; contains inappropriate content; and—at 33,000 words—is too long for anyone to gain an informed understanding of the issue at hand. This follows other unacceptable behaviour alluded to on his part, including his hack work on our periodic table article; removing some of my citation supported content; slandering me; swearing; and effectively demanding I provide a falsifiable hypothesis when I was under no obligation to do so.
The context for my objection is my long-term participation in the Group 3 debate, including two submissions to IUPAC on the matter, one with Double sharp (which he has now dissociated himself from).
My other objection is that an article written by me will soon appear in the journal Foundations of Chemistry, entitled "The location and composition of Group 3 of the periodic table". I have the proofs in hand. The article has been reviewed by three PhDs and accepted for publication by Eric Scerri, a world authority of the periodic table. Scerri is also the chair of the IUPAC Group 3 project.
Double sharp knows all this, yet chooses to go ahead with the RFC. So much for taking into account "reliable sources that focus on the matter." So much for extending me the courtesy of professional respect. So much for working collegiately within WP:ELEMENTS.
Here again is a continuation of his pattern of unacceptable behaviour.
Most of the rest of the editors who have voiced their opinions for Lu, it seems to me, do so on the basis of a personal preference or a very limited grasp of the full extent of the arguments and literature involved in this case.
I'm not holier than anyone. I do claim, along with Double sharp, to have maintained a depth of familiarity and interest in this question since at least 2016. In my case I've been arguing the question with Scerri, on and off, since 2008. And along the way, Double sharp and I have changed our minds about Group 3 as we both learnt new things along the way.
As Jones (2010) has written:
- "Scientists should not lose sleep over the hard cases. As long as a classification system is beneficial to economy of description, to structuring knowledge and to our understanding, and hard cases constitute a small minority, then keep it. If the system becomes less than useful, then scrap it and replace it with a system based on different shared characteristics."
- Jones 2010, Pluto: Sentinel of the outer solar system, Oxford University Press, p. 171
As Schwerdtfeger, Smits & Pyykkö (2020) wrote:
- "Fuzzy concepts like chemical similarity often lead to unnecessary disputes concerning the PTE."
- Schwerdtfeger P, Smits OR & Pyykkö P, 2020, "The periodic table and the physics that drives it". Nature Reviews Chemistry, vol. 4, pp. 359–380
That is why we have an IUPAC Group 3 project team, to assess the literature and address these unnecessary disputes.
And the IUPAC project team has reported that, based on a survey of chemistry textbooks and other sources, that the La form dominates by a 4:1:1 margin, where the two 1's are the *-** form and the Lu form.
As Scerri wrote:
- "…the majority of textbook and other periodic tables in the 18-column format show the elements of this group as scandium, yttrium, lanthanum and actinium…"
- Scerri ER 2020, "Recent attempts to change the periodic table", Philosophical Transactions A, here
As WP:Elements member R8R wrote:
- "…I'll say that the 4:1:1 consideration will be very important for the upcoming RfC, especially given that it's something that can be found on IUPAC's official website. I agree that consistency with literature is of the uttermost importance for Wikipedia if literature itself goes shows such a consistency; Wikipedia is meant to be a tertiary source. It'll be the thing that will hold me from supporting the motion on WP right now…--R8R (talk) 16:51, 19 July 2020 (UTC)"
Precisely.
The way ahead, as I see it, is (1) cancel the RFC until my article appears on line; and (2) Double sharp and I to agree on a balanced introduction to a new RFC, should Double sharp wish to proceed with another RFC. Failing this I will take up the matter on WP:AN. --- Sandbh (talk) 03:27, 21 July 2020 (UTC)
- The supposed consistency of the literature has been addressed above by noting that most reliable sources focusing on the issue support Lu, and that the 4:1:1 for La:Lu:* is of questionable relevance due to (1) Google results showing instead 1:1:4 for periodic tables, (2) general ambiguity resulting from * appearing in the same cell as La, and the fact that (3) there are some things that have been demonstrated many times in reliable sources but tend not to be taken up by textbooks, e.g. the refutation of d orbital involvement for hypervalent molecules. Incidentally, in the article Sandbh links to, Scerri supports Lu under Y.
| “ | Given these points I would like to propose that although there is no objective way to distinguish the tables shown in figures 10 and 12 [Lu and La respectively] one should nevertheless opt for the former of these as the official IUPAC periodic table. The factors that might lead us to favoring figure 10 have already been mentioned. They are that the d-block of the periodic table remains unified and intact, whereas the table in figure 12 requires that the d-block be separated into two highly uneven portions consisting of one and nine groups respectively. | ” |
| — Scerri ER 2020, "Recent attempts to change the periodic table", Philosophical Transactions A, here | ||
- I have added a thread at WP:AN#Dispute on an RFC. Double sharp (talk) 04:17, 21 July 2020 (UTC)
- To address Sandbh's concern about length, I have removed the long bottom-up explanation of periodicity, leaving only the arguments. Since we have been arguing about this for nearly seven months now at WT:ELEM, I think trying to create a balanced introduction to a new RFC that will please both of us may well be futile because our approaches diverge very early: many arguments that one of us consider decisive are not considered so by the other. However, I have absolutely no objection to him adding his own view of the background next to mine. Double sharp (talk) 04:58, 21 July 2020 (UTC)
On the "supposed" consistency of the literature
Here is what Double sharp neglects to mention.
Mathias (1969) grumbled about La in group 3 being the most popular form. Myers, Oldham and Tocci (2004, p. 130) found La and Ac to be the most popular form of periodic table, a sentiment echoed by Clarke and White (2008); and Lavelle (2008; 2009). My article (Vernon 2020) will reiterate this.
- Clark RW & White GD 2008, "The flyleaf periodic table", Journal of Chemical Education, vol. 85, no. 4, p. 497
- Lavelle L 2008, "Lanthanum (La) and actinium (Ac) should remain in the d-block", Journal of Chemical Education, vol. 85, no. 11, pp. 1482–1483, doi: 10.1021/ed085p1482
- —— 2009, "Response to misapplying the periodic law", Journal of Chemical Education, vol. 86, no. 10, p. 1187, doi: 10.1021/ed086p1187
- Mathias BT 1969, "Systematics of superconductivity", in PR Wallace (ed.), "Superconductivity: Proceedings of the Advanced Summer Study Institute on Superconductivity", McGill University, Montreal, vol. 1, Gordon and Breach, New York, pp. 225−294
- Myers RT, Oldham KB & Tocci S 2004, Holt chemistry, Holt, Rinehart and Winston, Orlando
- Vernon RE 2020, "The location and composition of Group 3 of the periodic table", Foundations of Chemistry, open access (in press)
As noted, IUPAC and Scerri have confirmed this. Double sharp knows it but neglects to mention it. There is no support in the literature for his WP:OR interpretation.
Scerri has noted (pers. comm.) that he distinguishes between his personal view as a chemist, and his role as the chair of the IUPAC Group 3 committee. Once again Double sharp demonstrates that he does not know what is going on here.
I reject Double sharp's assertion that it would not be possible for is to reach agreement on a balanced approach to an RFC. Once again he resorts to unwarranted—act of desperation—escalation i.e. 1. the hack work on our periodic table; 2, this RFC; 3. unilateral escalation to WP:AN. 08:05, 21 July 2020 (UTC)
Last try at being reasonable
@Double sharp: So, are we going to cancel this RFC and come up with an NPOV version, suitable for another RFC post online publication of my article, or are you going to waste everyone's time and some poor WP:ADMIN's? Sandbh (talk) 08:19, 21 July 2020 (UTC)
- @Sandbh: RFC tag removed (I understand this withdraws the RFC).
- Now I have stepped back and thought about it. I have gone overboard, and I apologise for it. I am still not at all convinced by your arguments and believe the Lu form is significantly better. However, since I also believe helium over beryllium is significantly better, yet recognise that that approach has no chance to become the default on WP, I am willing to let this one go provided we add notes to the relevant articles like what I did on helium explaining that there is a dispute. So, I have decided not to start a new RFC even after your article is published.
- I do not wish to discuss the issue further on WP at present. Everything relevant has already been said far too many times, and it seems unlikely that either of us are going to change our minds if it is said more times. It is also true that the fact that most textbooks discuss Sc, Y, La, and Ac together is, for Wikipedia, probably definitive at this moment. So, let's drop the issue until and unless IUPAC decides eventually on Lu under Y. In that circumstance, I think no one will argue that an RFC will be justified, but we do not have it at the moment.
- Respectfully, Double sharp (talk) 08:51, 21 July 2020 (UTC)
Discussion
- Strong Support for change to Lu form, per what I wrote above. Double sharp (talk) 10:08, 20 July 2020 (UTC)
- Also, per Michael D. Turnbull, below: I strongly support that periodic table article should show both forms and briefly discuss the difference. When I suggest changing to the Lu form, it is in the context where we simply show the periodic table as a template, e.g. for navigation like the infobox on neon or other elements. Double sharp (talk) 12:13, 20 July 2020 (UTC)
- Comment I think the reader would be aided massively if there were a one-line response to each question and the rest of the answer were hidden in a collapsed (but uncollapsible) box.--R8R (talk) 10:30, 20 July 2020 (UTC)
- @R8R: You raise a good point. I'll do it soon. Double sharp (talk) 11:22, 20 July 2020 (UTC)
- @R8R:
 Done Double sharp (talk) 12:40, 20 July 2020 (UTC)
Done Double sharp (talk) 12:40, 20 July 2020 (UTC)
- @R8R:
- @R8R: You raise a good point. I'll do it soon. Double sharp (talk) 11:22, 20 July 2020 (UTC)
- I will assume that I do not need to read this giant thing before giving my input? < Atom (Anomalies) 11:33, 20 July 2020 (UTC)
- @AnomalousAtom: I am working on adding one-line responses and collapsing the big ones per R8R's suggestion. In the meantime there is a summary. Double sharp (talk) 12:13, 20 July 2020 (UTC)
- @AnomalousAtom: It is now significantly less giant since all the detail is now in collapse boxes. Double sharp (talk) 12:40, 20 July 2020 (UTC)
- @Double sharp: Thank you. It is a little bit too long but I will take a look. The Request for Comment should be short though, because that is what Wikipedia:Requests for comment#Statement should be neutral and brief says. < Atom (Anomalies) 07:47, 22 July 2020 (UTC)
- @AnomalousAtom: You're welcome, but I withdrew the RFC. The problem is that the main argument doesn't really work. While it's true that the articles focusing on the issue tend to favour Lu under Y, this may be because La under Y was considered too obvious to need supporting until recently; the fractions from the 2000s onward are more balanced. Because if you look at articles discussing the placement of helium, for the same reason they tend to favour helium over beryllium (the unusual form) because the usual form above neon is I suppose considered too obvious to need defending. So I do not propose this change anymore right now, but prefer to wait and see if IUPAC decides in its favour – I hope it does. Double sharp (talk) 07:51, 22 July 2020 (UTC)
- @Double sharp: Understood, thank you. < Atom (Anomalies) 21:40, 22 July 2020 (UTC)
- @AnomalousAtom: You're welcome, but I withdrew the RFC. The problem is that the main argument doesn't really work. While it's true that the articles focusing on the issue tend to favour Lu under Y, this may be because La under Y was considered too obvious to need supporting until recently; the fractions from the 2000s onward are more balanced. Because if you look at articles discussing the placement of helium, for the same reason they tend to favour helium over beryllium (the unusual form) because the usual form above neon is I suppose considered too obvious to need defending. So I do not propose this change anymore right now, but prefer to wait and see if IUPAC decides in its favour – I hope it does. Double sharp (talk) 07:51, 22 July 2020 (UTC)
- @Double sharp: Thank you. It is a little bit too long but I will take a look. The Request for Comment should be short though, because that is what Wikipedia:Requests for comment#Statement should be neutral and brief says. < Atom (Anomalies) 07:47, 22 July 2020 (UTC)
- @AnomalousAtom: It is now significantly less giant since all the detail is now in collapse boxes. Double sharp (talk) 12:40, 20 July 2020 (UTC)
- @AnomalousAtom: I am working on adding one-line responses and collapsing the big ones per R8R's suggestion. In the meantime there is a summary. Double sharp (talk) 12:13, 20 July 2020 (UTC)
- I will assume that I do not need to read this giant thing before giving my input? < Atom (Anomalies) 11:33, 20 July 2020 (UTC)
- Neutral on the change but Strong support that the periodic table article should show both forms and give some (brief!) discussion of why some reliable sources use La and others Lu. There is no such thing as the periodic table, it has been presented in numerous forms over the years and Wikipedia should reflect that. However there is only one periodic table article and we need to put a summary there reflecting WP:NPOV on the various forms. Michael D. Turnbull (talk) 11:37, 20 July 2020 (UTC)
- Now Against the change after the useful clarification that we are really only talking about the appearance of Wikipedia templates like
{{Periodic table}}(See top of this RFC thread). We should stick to the same version as IUPAC currently endorse, i.e. IUPAC (2018-12-01). "PERIODIC TABLE OF ELEMENTS". since that's what most readers would expect. (Even British chemists don't use sulphur now, only sulfur.) Michael D. Turnbull (talk) 13:49, 20 July 2020 (UTC)- Actually it is not quite true that IUPAC endorses the form. They write on their website "While IUPAC has no recommendation for a specific form of the periodic table, i.e. 18-column or 32-column format, the version here presented is in the conventional long form and is yours to use."
- Now I can see the argument for using * under Y as that's what IUPAC shows on their webpage, even if they seem to be planning to change it away from * under Y eventually. But it does make things awkward for the 32-column forms encountered in
{{Compact periodic table}}etc. Will scandium and yttrium have to be stretched over 15 columns? The only way it becomes less awkward is if we change those to the 18-column form too. Which is a bit odd as the 18 and 32 column forms are supposed to be saying the same thing in a different layout. Double sharp (talk) 13:56, 20 July 2020 (UTC)
- Strong support on changing to Lu as per arguments above. I was involved in the discussion itself as a minor participant. ― Дрейгорич / Dreigorich Talk 12:29, 20 July 2020 (UTC)
- Support change to Lu per arguments above; I also was a minor participant in the group 3 megathread. I would also like to comment that the IUPAC standard is intended to be deprecated (
that is why this RfC does not consider Sc-Y-*-*;(not originally considered here but later added) it is possibly even more incorrect to place all 30 inner transition metals below Sc and Y, and it still doesn't address how to structure the 32-column table), and the remaining arguments strongly suggest that Lu is the better choice. ComplexRational (talk) 14:11, 20 July 2020 (UTC)- @ComplexRational: BTW, I just added Sc-Y-*-** as an option since Michael D. Turnbull is in favour of it. So now it is not accurate anymore that the RFC does not consider the option. It is however correct that I am against that option. ^_^ Double sharp (talk) 14:18, 20 July 2020 (UTC)
- Extremely strong oppose (I have to make this prominent to balance and counteract the supports above) Just about all the arguments are irelevant and we should just use the traditional form with La and Ac under Y. If needed we can use the place holder * or **. Graeme Bartlett (talk) 07:52, 21 July 2020 (UTC)
- Illegitimate RFC due to breaching Neutral point of view, Verifiability, No original research, and Lack of civility. Sandbh (talk) 08:15, 21 July 2020 (UTC)
- @Sandbh: RFC tag removed (I understand this withdraws the RFC).
- Now I have stepped back and thought about it. I have gone overboard, and I apologise for it. I am still not at all convinced by your arguments and believe the Lu form is significantly better. However, since I also believe helium over beryllium is significantly better, yet recognise that that approach has no chance to become the default on WP, I am willing to let this one go provided we add notes to the relevant articles like what I did on helium explaining that there is a dispute. So, I have decided not to start a new RFC even after your article is published.
- I do not wish to discuss the issue further on WP at present. Everything relevant has already been said far too many times, and it seems unlikely that either of us are going to change our minds if it is said more times. It is also true that the fact that most textbooks discuss Sc, Y, La, and Ac together is, for Wikipedia, probably definitive at this moment. So, let's drop the issue until and unless IUPAC decides eventually on Lu under Y. In that circumstance, I think no one will argue that an RFC will be justified, but we do not have it at the moment.
- Respectfully, Double sharp (talk) 08:51, 21 July 2020 (UTC)
How about discussing navigation aids? The present one, seen on the information box for every element (look at Neon for example) is arguably too wide for easy use. It has 32 columns and as a result provides only tiny boxes for people to use to link to other elements that they may wish to jump to next. I'd like to make a proposal for a new navigation aid of 18 columns, as below. Astute readers will realise that this also has something to do with the debate we have recently been having on this page!
- New version 1
Michael D. Turnbull (talk) 09:49, 21 July 2020 (UTC)
- But isn't this clearly a Lu under Y table? And surely that will mean the arguments opposers have used above are still valid against this one? Double sharp (talk) 09:54, 21 July 2020 (UTC)
- For the avoidance of any doubt, should we use a 18-column table, could we have a small gap where lanthanides are supposed to be, like in {{periodic table}}?--R8R (talk) 10:00, 21 July 2020 (UTC)
- OK, so we could have "a nothing at all under Y" table, as version 2. Personally, I never understood why the Wikipedia tables have the lanthanides and actinides as a separate strip but without including those elements in that strip! Version 2 is more like IUPAC.
- New version 2
Michael D. Turnbull (talk) 10:42, 21 July 2020 (UTC)
- Yeah, I think this * form is best for Wikipedia. It is at least as close to neutral as you'll get, and it has the argument "well, that's what IUPAC shows now; we know they plan to change it, so we will change it only when they do". We can sweep the 32-column problem under the rug by simply not using the 32-column form much. What you say about La and Ac is indeed one scientific objection I have to the La form, but, again, the important thing on WP is probably not the scientific arguments. Double sharp (talk) 11:24, 21 July 2020 (UTC)
- I think we should keep our -La-Ac form there. For one reason that trumps all other possible reasons combined: we need to be consistent about our PT layout. If our layout is -La-Ac, or -*-**, or -Lu-Lr, then so be it.--R8R (talk) 18:43, 21 July 2020 (UTC)
- I agree with Michael D. Turnbull that the current 32-column form is too wide and provides only tiny boxes. I agree with R8R that we should keep our -La-Ac form there. Sandbh (talk) 00:56, 22 July 2020 (UTC)
- I think we should keep our -La-Ac form there. For one reason that trumps all other possible reasons combined: we need to be consistent about our PT layout. If our layout is -La-Ac, or -*-**, or -Lu-Lr, then so be it.--R8R (talk) 18:43, 21 July 2020 (UTC)
Ah, now we are getting somewhere! What we were all forgetting is that a wiki page is not the same as a printed page. We have the luxury of being able to link cells in the navigation table to anything we like AND give a hover-over message that's appropriate. This is very useful when filling in the contentious cells that IUPAC places asterisks in. We can do better..... see my third and final suggestion below. For the avoidance of doubt, I've added back the usual table legend at the bottom and, yes, I would add a blank row between the main body and the Lanthanide/Actinide strip, if only I could work out how to do so. Note what happens when you click on the cells now below element Y: it's not the same as the click on the words "Lanthanides" or "Actinides" at lower left.
- New version 3
I don't think we need any more full versions to discuss the issue and find a consensus for this navigation table to replace {{Compact periodic table}}. We just need to agree a) the colours - I see no need to change; b) the short text where I've currently got La em-dash Lu; c) the Hover-text (I think this has to be the page name that the click will link to) and d) the correct wikilink-on-click Michael D. Turnbull (talk) 09:16, 22 July 2020 (UTC)
- I still consider this best for Wikipedia. It will have the argument "IUPAC gives it" behind it in case anyone complains. It will also be neutral in the sense that neither Sc-Y-La aficionados nor Sc-Y-Lu aficionados will be terribly happy. Then a footnote can be added in saying "The elements of group 3 below scandium and yttrium are disputed. Some tables put lanthanum (La) and actinium (Ac) in these positions, while others put lutetium (Lu) and lawrencium (Lr) there. Hence we show a compromise form with all lanthanides and actinides placed under yttrium, as does the current IUPAC periodic table. A IUPAC project was started in 2015 to decide the question."
- Now, it does make the scientific statement that group 3 has 32 elements in it. That is indeed scientifically a pretty strange view given the properties of the actinides. However, that is precisely what was done in the JWP (IUPAC-IUPAP) report on the discoveries of Nh, Mc, and Ts.
| “ | The technique employed was claimed to distinguish between Group 3 elements (lanthanides and actinides) and combined Groups 4 and 5. ... No carrier-free actinide tracers were employed despite the extremely complex oxidation chemistry and adsorption quirks of those Group 3 elements in contrast to lanthanide behavior. | ” |
| — That JWP report | ||
- The evidence is clear: IUPAC in its publications supports this view. People rarely do, of course. But as long as the debate rages and IUPAC has not decided on a form, I support Michael D. Turnbull's proposal: Sc-Y-* and 18-column until IUPAC makes up its mind. Maybe it is not neutral because Sc-Y-* is itself a side, like I said. But you will never find anything more neutral than to follow IUPAC when chemists cannot agree. Double sharp (talk) 10:13, 22 July 2020 (UTC)
- At Michael D. Turnbull's request on my talk page, I have added my proposed footnote above as hovertext for the cells reading "La–Lu" and "Ac–Lr". Also a blank row between the main body and the footnote. Double sharp (talk) 11:46, 22 July 2020 (UTC)
- Could we please please please stick a single layout and use it consistently in en.wiki? As far as I am concerned, the question of whether it should be -La-Ac, -*-**, or -Lu-Lr is less important than whether we use a consistent format or not (we definitely should). If there really is that much appetite for using a different format than what we use at {{periodic table}}, then please let us discuss a change that concerns all periodic tables, not just the one in element infoboxes.--R8R (talk) 14:57, 22 July 2020 (UTC)
- I absolutely agree! The template for
{{periodic table}}is, however, enormous and I hesitated to inflict it on this thread. You may not be aware that the template used in the infoboxes at each element page is very much based on what we're discussing here, which as you know goes at the foot of each article. It gets some clever tweaks that highlight the element in question in the periodic table when used in the infobox. So, once we have settled the current discussion and found a competent volunteer who is familiar with template documentation and won't mess up the implementations (that ain't me!) all relevant en.wiki templates should be done. If you feel a separate discussion is needed for{{periodic table}}, please start one, or sandbox ideas to link here (the template has its own sandbox for that purpose). Michael D. Turnbull (talk) 15:13, 22 July 2020 (UTC) - R&R kindly sandboxed something like version 3 at this link but for the moment I don't think we need to take this further. When the dust settles on the current discussion and moves to implementation, we'll certainly keep consistency between templates. Michael D. Turnbull (talk) 17:14, 23 July 2020 (UTC)
There are several things to consider here. [start of comment by Sandbh 23:42, 23 July 2020 (UTC)]
1. There is no such thing as an IUPAC recommended or endorsed periodic table. The table that appears on their website is used within IUAPC. A spokesperson has made it clear that IUPAC does not recommended or endorse any form of periodic table (Leigh 2009).
- Leigh, J.: Periodic tables and IUPAC. Chem. Int., 31, 4–6 (2009)
- Not worth fighting over this but Leigh seems to have changed his mind by 2019, in preparation for IYPT. "In conclusion, IUPAC was not involved in the development of the Periodic Table, but .... it has been the prime international authority for developing and adopting changes in its content ...." see Leigh, G.J. (2019). "IUPAC and the Periodic Table". Chemistry International. 41: 6–9. doi:10.1515/ci-2019-0102. S2CID 126911180.. Maybe in 2019 he only meant content, not the format but if so he didn't make that clear in his article. Michael D. Turnbull (talk) 17:33, 23 July 2020 (UTC)
- @Michael D. Turnbull: The development and adoption of changes Leigh was referring to included, "new elements, and their atomic numbers, names, symbols, and atomic weights."
- Not worth fighting over this but Leigh seems to have changed his mind by 2019, in preparation for IYPT. "In conclusion, IUPAC was not involved in the development of the Periodic Table, but .... it has been the prime international authority for developing and adopting changes in its content ...." see Leigh, G.J. (2019). "IUPAC and the Periodic Table". Chemistry International. 41: 6–9. doi:10.1515/ci-2019-0102. S2CID 126911180.. Maybe in 2019 he only meant content, not the format but if so he didn't make that clear in his article. Michael D. Turnbull (talk) 17:33, 23 July 2020 (UTC)
- Nor does IUPAC prescribe which elements, for example hydrogen and helium, belong in which group. All they have done is recommend collective names for like elements e.g. chalcogens (oxygen, selenium, selenium, tellurium, polonium) and noble gases (helium, neon, argon, krypton, xenon, radon). For example, if helium is placed over beryllium then helium still belongs to the noble gases.
- IUPAC does not recommend or approve any particular format of periodic table or system, nor does it mandate the composition of Groups.
- To this end, Scerri (2020) writes:
- "The fourth possible presentation is one that abstains from specifying which elements belong in group 3 or rather it is a presentation that only contains two elements, scandium and yttrium, as being definitely in group 3. This table is sometimes labelled as the official IUPAC periodic table although a leading IUPAC official has clearly stated that this organization does not currently support any particular version of the periodic table and that has only concerned itself thus far with numbering 18 of the groups of the table."
- Scerri E 2020, "Recent attempts to change the periodic table", Philosophical Transactions A, 378: 20190300, in press.
- --- Sandbh (talk) 23:42, 23 July 2020 (UTC)
- @Sandbh: ;-) I'm not advocating for IUPAC. I just find it amusing that they want to police all the cells in the periodic table (i.e. the list of contents you mention above) but are apparently agnostic about the architecture of the prison in which these cells fit, despite showing one ugly logo-ridden "prison" on their website. Let's stick to discussing the things we can control, which is what Wikipedia uses as its navigation boxes and other templates. Michael D. Turnbull (talk) 09:18, 24 July 2020 (UTC)
- @Michael D. Turnbull: I see nothing amusing about it. Someone has to do it. Who better than the international body for chemists. When you say "Let's stick to…", in the context of so few exchanges of perspectives, I get quite annoyed. I'd prefer that you not presume my support for your view of what "we" should do next. Why would IUPAC want to get involved in the architecture of the prison? Would groupthink in this matter be a good thing? To the extent that anything is under control in Wikipedia, control of the navigation boxes and other templates falls within the purview of WP:ELEMENTS. To that end, you may wish to consider becoming a member of our project. Sandbh (talk) 13:15, 24 July 2020 (UTC)
- @Sandbh: The issue is what the "it" is. If one means "settle the dispute about group 3", then IUPAC has a role. If one means "choose which form of the periodic table people must use" that clearly would be groupthink, as
DePiepyou put very eloquently later in this thread (on bookburnings). On Wikipedia I like to WP:AGF and just get on with the task in hand where the "it" is to build a better encyclopedia. I would be a poor member of the WP:ELEMENTS project because I have expertise only in a tiny proportion of them and none at all on the group 3 question. That still won't prevent me chipping in if I think it worthwhile. Michael D. Turnbull (talk) 14:17, 24 July 2020 (UTC)
- --- Sandbh (talk) 23:42, 23 July 2020 (UTC)
- @Michael D. Turnbull: There is no dispute about group 3 within the chemistry community. A few (relatively speaking) people have suggested Lu looks better under Y (ignoring the background to, or features of, La under Y). Jensen had a red hot go in 1983, and failed to gain traction. Scerri, who is chair of the IUPAC Group 3 project described Jensen's arguments as being too selective. Just because there is such a project does not mean IUPAC will take any notice of its report or recommendations. You have no understanding of the politics, history and culture involved. You are more than welcome to chip in here at any time. Please do not do so after the fashion of a WP:BICS bull in a china shop. People in our project have been discussing the group 3 project for several years. That does not mean they are right. It does mean they know quite a bit about the situation. Sandbh (talk) 06:31, 25 July 2020 (UTC)
2. The IUPAC table is now especially "bad" since they deliberately misaligned the 15-element wide f-block purely so that they could fit the IUPAC logo in.
3. The 18-column *-** form results in the f-block becoming part of the d-block. This is so bad that Jensen (2008) referred to it "chemical nonsense" and that, "IUPAC or not, I can hardly believe that a modern inorganic chemist would advocate such an antiquated interpretation of these elements, unless, as noted above, they have lost all contact between the underlying premises of their periodic table and the facts of chemistry." Disclaimer: While I agree with Jensen in this case, I disagree with his position on Group 3.
- Jensen WB 2008, "The Periodic Table: Facts or Committees?", Journal of Chemical Education, vol. 85, no. 11, pp. 1491−1492
4. Using the *-** form is not consistent with the literature. Thus, Mathias (1969) grumbled about La in group 3 being the most popular form. Myers, Oldham and Tocci (2004, p. 130) found La and Ac to be the most popular form of periodic table, a sentiment echoed by Clarke and White (2008); and Lavelle (2008; 2009).
- Clark RW & White GD 2008, "The flyleaf periodic table", Journal of Chemical Education, vol. 85, no. 4, p. 497
- Lavelle L 2008, "Lanthanum (La) and actinium (Ac) should remain in the d-block", Journal of Chemical Education, vol. 85, no. 11, pp. 1482–1483, doi: 10.1021/ed085p1482
- —— 2009, "Response to misapplying the periodic law", Journal of Chemical Education, vol. 86, no. 10, p. 1187, doi: 10.1021/ed086p1187
- Mathias BT 1969, "Systematics of superconductivity", in PR Wallace (ed.), "Superconductivity: Proceedings of the Advanced Summer Study Institute on Superconductivity", McGill University, Montreal, vol. 1, Gordon and Breach, New York, pp. 225−294
- Myers RT, Oldham KB & Tocci S 2004, Holt chemistry, Holt, Rinehart and Winston, Orlando
5. A survey by the IUPAC Group 3 project team found that the majority of textbook and other periodic tables in the 18-column format show the elements of group 3 as scandium, yttrium, lanthanum and actinium. The survey did not include a Google image search, since such results are plagued by bias arising from the false impression that the IUPAC *-** table is “official”, and unreliable sources.
6. As R8R noted, "consistency with literature is of the uttermost importance for Wikipedia if literature itself shows such a consistency; Wikipedia is meant to be a tertiary source."
--- Sandbh (talk) 01:19, 23 July 2020 (UTC)
- Version 1 and 2 have this omission: the bar below is not representend in the main table, there are no placeholders (usually asterisks). One cannot see where the bar below is supposed to be. Also, there is not extra separator (like whitespace) between the main table and the bar below. This
suggestssays that the bar below is part of the main table.
- Version 3 says that the bar below is in total below Sc, Y. Thus, Sc and Y and group 3 all stretch 15 columns. -DePiep (talk) 06:14, 23 July 2020 (UTC)
Please focus now on improving version 3
- Yes, versions 1 and 2 are now superceded by version 3, which is all we need to discuss beyond this point. I will update the current version 3 in the thread as we appear to reach any new consensus about it. Personally, I'd be happy if the "hover-over" on the cells under Y didn't mention IUPAC as we are creating a new standard for en:Wiki, not trying to do the impossible! Is that the crux of your objections @Sandbh:? If so we'll just delete ", as does the current IUPAC periodic table" from the hover-over: which has the advantage of making it shorter for readers, which in the long run could be millions of people. Alternatively, if your objection is that "there can only be one group 3 element below Y", then we'll make the text in that cell "La or Lu" rather than "La—Lu" and again clarify in the hover-over. We would change the colours of the relevant two cells as well, of course. This "or" solution has the huge advantage that Wikipedia's version will now include La in the Lanthanides strip, which was one of the issues I had with our current
{{periodic table}}, which doesn't. I have been WP:BOLD and made these changes now, so everyone can see their effect. Incidentally, the IUPAC website today says "The question of precisely which elements should be placed in group 3 has been debated from time to time. An IUPAC project has been recently initiated to resolve the question. Will group 3 consist of Sc, Y, Lu, and Lr or, will it consist of Sc, Y, La and Ac? (My emphasis)" Michael D. Turnbull (talk) 14:37, 23 July 2020 (UTC)
- It's not a new standard. Sc-Y-* is exactly what we had up till 2014. Then it got changed to Sc-Y-Lu after some discussion at WT:ELEM back then. And then it got changed to Sc-Y-La after the previous RFC at Template talk:Periodic table. (When I knew less and thought that was scientifically better; now I know more and consider Sc-Y-Lu scientifically better.) So I do not mind if it gets changed to Sc-Y-*. It would just be full circle.
- Regarding Sandbh's and DePiep's points:
- We have no problem using IUPAC-internal stuff as a compromise even if not everyone outside follows it, viz. the spelling guideline WP:ALUM. In fact that is a precedent for doing it when there are two significant sides that simply do not agree. Going for Sc-Y-La picks a side. A 2/3 majority of textbooks as the survey shows is hardly so much that picking such a side can be considered neutral; if it were 90% that point would be stronger. IUPAC however seems something we can compromise on.
- The misalignment is not an issue, the footnote is a footnote regardless of where it is placed. It still means that the same elements go in the same place.
- The fact that version 3 says that all the Ln and An are in group 3 is not a problem, as that is how they are indeed considered IUPAC-internally. See the quote above.
- The problem I have now with referring to the literature to chemically analyse and be against Sc-Y-*, as Sandbh did, is that in general as I understand we are not supposed to analyse it ourselves for WP. That is treading close to original research. In fact there is another problem, which is that starting to analyse this will inevitably lead to starting the eka-yttrium holy war again. In fact, I did, but thankfully Michael D. Turnbull came by below and on my talk page and knocked some sense into my head. So I remove that text. Sense and focus knocked into my head aside, to avoid this problem I prefer following IUPAC with Sc-Y-* to compromise. Double sharp (talk) 14:54, 23 July 2020 (UTC)
- @Double sharp:We are in danger of re-running the Group 3 debate in this thread! Please focus on the issue at hand, which is what do you want an 18-column table to look like? There are only 4 possible parts to alter, as I already pointed out below version 3. They are a) the colours in the cells b) the short text where I've currently got "La or Lu"; c) the Hover-text and d) the correct wikilink-on-click. Surely you can confine your comments to making suggestions specifically for these, without saying much else? Michael D. Turnbull (talk) 15:03, 23 July 2020 (UTC)
- @Michael D. Turnbull: My apologies. Thank you for knocking some sense into my head: I removed the old text. I think you will like what is now there instead.
- Now the answer. I prefer the old form; AFAIK IUPAC still considers the other Ln and An to be in group 3 even after the project started judging from the JWP reports above. So I read the "or" as meaning that they will eventually decide on one or the other, but right now the old situation of Sc-Y-* is still their form in the interim. More to the point, the colouring of the cells below Y with the Ln and An colour is also on the IUPAC table. IUPAC is a strong and powerful argument for compromise, let's keep it strong by sticking close to what they do. As for tooltips, anything that says there's a dispute, this is the IUPAC-internal form, and IUPAC is looking into it will be fine with me. Precise wording is not a problem. Double sharp (talk) 15:13, 23 July 2020 (UTC)
- I have made bold edits, see #Step 1 done below; main OP issue solved. However, other changes mentioned here I object to. Later will have time for arguments. -DePiep (talk) 15:23, 23 July 2020 (UTC)
versions 1 and 2 are now superceded by version 3, which is all we need to discuss beyond this point.
No. "Superceded" has not been established. Thi subthread, with its non-sequitir ignation, is invalid. Dead on arrival. No need to spend time on this. -DePiep (talk) 23:47, 23 July 2020 (UTC)
- Apologies, De Piep, my language was not precise enough. By "supercede" I meant that version 1 and 2 are versions which reflect the past discussions in this thread and should not be changed in any way, so new readers will see where we came from, while version 3 can and will change as we stumble towards a consensus — in that respect it's like the thread's sandbox, so is all we need to discuss (and possibly change) beyond this point. What you have already implemented on the live Wikipedia navigation template in 18 columns largely meets my request for change: and this would be complete when you do the same change on the element infobox version, where the problem of tiny cells in 32-column format is even more acute. Frankly, I despair of reaching a consensus over the group 3 issue and I don't really care: it is irrelevant to navigation. Anyone who wants to fight about this narrow issue is welcome to do so below in this thread but I probably won't engage. My only new contribution has been to point out that the hover-over tooltip gives us a mechanism to point out the controversy without resolving it. Michael D. Turnbull (talk) 09:37, 24 July 2020 (UTC)
I make the following observations: The spread is 4:1:1, where Sc-Y-La-Ac is four times as common as either *-** or Lu-Lr. This is nowhere near any degree of ambiguity sufficient to justify a judgement call or compromise. Going for Sc-Y-La does not pick a side. There is no finely balanced contest of sides.
The misalignment of the IUPAC table is an issue in that it prioritises vanity branding over the alignment of the early Ln and An with their 5d analogies. Yes, the perception of such "stupidity" is circulating within the chemistry community.
Nobody takes account of the strong resistance IUPAC has to making recommendations on group composition or the form of periodic table. For example, if the IUPAC recommends group 3 as La will this render the Janet table as being politically incorrect or wrong-headed thinking? No doubt the IUPAC would have to set up re-education internment camps for Lu supporters. Next to be labelled as deviant will be Group 14 in the Earth Scientist’s periodic table being composed of carbon, silicon, titanium, zirconium, and hafnium rather than the standard set of carbon, silicon, germanium, tin, and lead. Book burnings to follow.
Once the Group 3 project has made its report there is no guarantee the IUPAC will accept it. If it is released it will go to the chemistry community for consultation. Even then there is no guarantee the IUPAC will turn the report into a recommendation.
La or Lu, plus La-Lr breaches the maxim of one element one place.
The place for accommodating the variants of Group 3 is in the main body of our periodic table article, as we do now. Here we write that, "Although scandium and yttrium are always the first two elements in group 3, the identity of the next two elements is not completely settled. They are commonly lanthanum and actinium [by a wide margin], and [significantly] less often lutetium and lawrencium." (my brackets)
Just so. --- Sandbh (talk) 00:24, 24 July 2020 (UTC)
- I do not agree that the important figure is 4:1:1. The IUPAC survey breaks things down by decades, and you can see that the La form is steadily losing its majority since the 1990s:
| Decade | La under Y | Lu under Y | * under Y | Total |
|---|---|---|---|---|
| 1970s | 18 69% |
2 8% |
6 23% |
26 |
| 1980s | 31 78% |
6 15% |
3 8% |
40 |
| 1990s | 27 82% |
2 6% |
4 12% |
33 |
| 2000s | 38 62% |
14 23% |
9 15% |
61 |
| 2010s | 16 48% |
6 18% |
11 33% |
33 |
- As you can see, by the 2010s La has lost its majority. I guess the reason is WebElements becoming popular and supporting Lu, though I have no source for that. For this reason I think * would be more justified to show for neutrality. Double sharp (talk) 02:45, 24 July 2020 (UTC)
@Double sharp: Why are you engaging in WP:FORUMSHOPing? You have posted the same content to our project page. Please confine your argument to one forum. And where is the hyperbolic language coming from? It is like there are two of you: one who is calm; the other one who is foaming at the mouth. Sandbh (talk) 06:48, 24 July 2020 (UTC)
- @Sandbh: It seems relevant to both places since the 4:1:1 dominance has been discussed in both places. If you want, I can delete it from the project page and discuss it only here as it seems more relevant for this decision. I have replaced the old text with text from the WT:ELEM page.
- Let me just say regarding your comment there: I do not think we should drill down into where the increase in * and Lu figures are coming from. You say at the project page that probably * comes from a mistaken view of IUPAC and Lu comes from Jensen (who you then refer to Scerri and Parsons to criticise); that's all fine and well, but I could very well also say that the huge figures in favour of La in the earlier decades come from the debate being poorly publicised and most authors being unaware that a dispute existed at all (for which I can refer to Jensen saying in 1982 that no chemists he talked to were aware that the dispute existed, and indeed that he was not aware of it till shortly before he published his article). That's the problem: both sides can say "that's not so suggestive because X" to counter the other one. I think that's exactly the sort of situation that demands simply looking at what the sources say and not trying to second-guess why they chose as they did, if we want neutrality.
- But all in all, I don't really care that much anymore, as long as it cannot be Lu. I have no strong feelings about whether Sc-Y-La or Sc-Y-* is better than the other. Double sharp (talk) 08:04, 24 July 2020 (UTC)
- @Michael D. Turnbull, Double sharp, Sandbh, and R8R: If I understand this well, this is a proposal to present the PT with a different group 3 constitution (actually an "OR" setup) than we present until now, while not changing other general PT graphs this way. I see two major problems with this:
- Problem 1: This would introduce different presentations in general PT graphs. ("general" means non-specific topics, i.e. the basic presentation). I see no reason to introduce these forms, over our current policy to have one preferred version.
- Problem 2: It redoes the group 3 discussion in a side-place (just a navbox). However difficult to conclude the main talk, we cannot create backdoor-decisions. If we want to present group 3 in an "or"-form in general (or, in the IUPAC-form of 32 elements ouch), that should be an explicit proposal.
- -DePiep (talk) 10:41, 25 July 2020 (UTC)
- Not as far as I'm concerned as the OP. I just wanted to suggest changing the 32-column hard-to-use navigation boxes into 18-column ones and I made a proposal. I inadvertently got dragged into a Group 3 battle which in the context of this sub-thread breaches WP:NOTAFORUM. I'm very pleased with what you've already done as Step 1 and will remove myself entirely from the sub-thread once Step 2, the corresponding change in the navigation box used in the Infobox of each element, is also done. I assume you could do it now, although I appreciate it may be a lot of work for you and requires a consensus that the change is sensible: which actually I think already exists. Michael D. Turnbull (talk) 10:56, 25 July 2020 (UTC)
- Minor problems with #New version 3. (Minor = not involving a group 3 decision; should be solvable by graphical/textual means).
- A. Background color cells "La or Lu" and "Ac or Lr" now is says "Transition metal", I guess Ln, An colors are expected there.
- B. The bottom bar (I do not say 'footnote', as footnote is a textual detail or comment while here the bar is full part of the table), the bottom bar cannot be rendered unambiguously into the main table. There is no clue as to where it should end up. It would leave the reader with a puzzle. As we know, there can not be a difference between a 18-column PT and its equivalent 32-column form.
- C. It reintroduces the category names "Lanthanide" and "Actinide" spelled out inside the table. We have maintained since long that we do not add category names inside the table at all. Keeping these gives the LNs and ANs a special status, adding complexity without reason. (If there were one category to be named, it should be metalloids first as that is a major deviding set, in the metal=metalloid-nonmetal trend).
- D. Currently {{hover title}} is used to describe & explain the "or" issue. However, using tooltip texts this way is bad practice. WP:Accessability says
Do not use techniques that require interaction to provide information, such as tooltips or any other "hover" text. [{{abbr}} is the exception]
See for example issues with title: not available in mobile devices, not working well with screen readers. The information better be provided in a textual footnote.
- Most of this stems from the desire to present two group 3 forms; making a choice in this first would help better. -DePiep (talk) 11:04, 25 July 2020 (UTC)
- The choice is effectively already made, as in the current
{{periodic table}}and new live 18-column navbox. I am happy to agree with your technical views on the implementation for the element Infoboxes and give up on the other ideas as being unworkable, bad practice, or impossible to gain consensus. There is no need to present two group 3 forms and I'm happy to stick with Wikipedia's current implementation: I believe that even ^_^ accepts this for now. Michael D. Turnbull (talk) 11:22, 25 July 2020 (UTC)- @Michael D. Turnbull: Indeed, I do. Double sharp (talk) 11:38, 25 July 2020 (UTC)
- The choice is effectively already made, as in the current
- continued in #Micro PT in infobox below. -DePiep (talk) 22:29, 25 July 2020 (UTC)
Micro PT in infobox
-
- Well, not the status quo where readers of Neon see a difficult-to-use 32-column presentation in the Infobox but a new status quo where they see an 18-column one instead. The detection was difficult owing to the WP:NOTAFORUM stuff and my lack of knowledge that a tooltip is not a place to add information. Michael D. Turnbull (talk) 11:49, 25 July 2020 (UTC)
- I am surprised to read that this seems to be the topic of this thread. So somewhere it is noted that the micro PT {{Periodic table (32 columns, micro)}} would improve in 18-column form? (I don't think so btw). Please describe what the difficulty is, if that's not done before. -DePiep (talk) 12:31, 25 July 2020 (UTC)
- That's precisely what I said in the second sentence of this thread. I went on to use the template that's used at the bottom of all element pages for navigation for the purposes of starting the discussion because a) I thought the Infobox one was based on the bigger one b) I wanted to make a specific proposal so we could start a discussion c) my (limited) skills at editing templates meant it was easier to sandbox my ideas in the bigger one and d) I assumed that whatever we decided for the bigger one would be applied to {{Periodic table (32 columns, micro)}} since that's the one that I and presumably others have most difficulty actually using for navigation: the cells are simply soo small to accurately put a cursor on the correct place when you want to click onto another element's article. I now see that I should have started the thread by making a proposal to replace {{Periodic table (32 columns, micro)}} with {{Periodic table (18 columns, micro)}}, which already exists, in element Infoboxes. That's a much simpler proposition! I didn't at the time of starting this thread realise that 18-micro PT existed.... I'd still like to discuss and implement that change, so indeed a new thread may now be the best way forward now that you have already done Step 1. Michael D. Turnbull (talk) 13:11, 25 July 2020 (UTC)
- It's just that this thread started multiple issues at the same time. About this one: it is called 'micro' for a reason; usage in the infobox (and other places [1] 500 in total) is to illustrate a position in the PT, not chiefly a PT navigation box. As it happens, the 18-col variant has equaly sized cells ;-). 18 columns introduces this drawback: the reader is supposed to mentally reposition the bottom bar (LN, AN), like building an IKEA cupboard. This complication is unneeded, and undoes the "PT overview" aim. -DePiep (talk) 15:55, 25 July 2020 (UTC)
- Yes, I see how the thread muddles several issues. I agree that the primary aim of the infobox version is to locate the element and in some respects it is unfortunate that it actually behaves like a navigation box at all. It might be better that a hover over the tiny cells gave the name of the remote element but couldn't be used to jump to that element's page (reserving this functionality for the larger navigation box at the foot of the article). Anyway, that's not worth a discussion nor a change and as a result I suggest we close this thread: thanks so much for your help in doing the implementation of what we did agree. I won't post here again unless pinged. By the way, I'm surprised that no-one seems to have used {{Periodic table (18 columns, micro)}} anywhere in the main encyclopaedia, as that version is a very compact display of the whole table in a format that will be more familiar to most chemists than the 32-column counterpart. I'm going to put it on my user page! Michael D. Turnbull (talk) 16:10, 25 July 2020 (UTC)
- It's just that this thread started multiple issues at the same time. About this one: it is called 'micro' for a reason; usage in the infobox (and other places [1] 500 in total) is to illustrate a position in the PT, not chiefly a PT navigation box. As it happens, the 18-col variant has equaly sized cells ;-). 18 columns introduces this drawback: the reader is supposed to mentally reposition the bottom bar (LN, AN), like building an IKEA cupboard. This complication is unneeded, and undoes the "PT overview" aim. -DePiep (talk) 15:55, 25 July 2020 (UTC)
- Well, not the status quo where readers of Neon see a difficult-to-use 32-column presentation in the Infobox but a new status quo where they see an 18-column one instead. The detection was difficult owing to the WP:NOTAFORUM stuff and my lack of knowledge that a tooltip is not a place to add information. Michael D. Turnbull (talk) 11:49, 25 July 2020 (UTC)
Step 1 done
(ec) I have made these edits boldly:
- Per original Original Post by User:Michael D. Turnbull first issue, stating that 32-columns is
arguably too wide for easy use
, esp on smaller screens: I have reformed the format into an equivalent 18-column Periodic table.
- Note that this does not change the described constitution of group 3. The background is that the enwiki Periodic tables are explicitly supposed to reflect the same PT consistently, basically the one per Periodic table, except when the topic is a deviation (for example, Periodic table#Placement of hydrogen and helium).
- I have Moved the template to new name {{Periodic table (navbox)}}, so as to reflect its most important feature: being a navbox (with all its quirks). The old name now Redirects to this one (no visible change or break for articles).
- However, the OP also states:
Astute readers will realise that this also has something to do with the debate we have recently been having on this page!
. But exactly the opposite is at hand: the group 3 discussion (both at enwiki and in RL) is not related to the form being 18- or 32-column. For illustration: RFC initator Double sharp has presented, correctly and gently, each of the three options in both their 18- and 32-column form, i.e., six PTs in total.
- Also, other changes introduced in this thread (like labeling "lanthanides, actinides" below) are not performed, as these have no consensus (and to be clear, I oppose; more arguments later). -DePiep (talk) 15:20, 23 July 2020 (UTC)
Reverted
I have reverted this change in {{Periodic table (navbox)}} [2], as some editors pointed to a misunderstanding and confusion in this thread. The navbox is back to 32-column format. This does not state any prejudice. New proposals best be made in a new thread. See also #Propose closing as is below. -DePiep (talk) 09:47, 26 July 2020 (UTC)
Next steps
@Michael D. Turnbull, DePiep, Sandbh, and Double sharp: sorry, I haven't been watching the discussion very closely. But did we arrive to the conclusion we wanted? I thought that if we needed to switch from 32 columns to 18 anywhere, it would be the small table in the infobox rather than the one at the bottom of a page. Do we not want to change it instead? Was there really a problem with the table at the bottom of our element articles?
Also, DePiep, on my screen the renewed table looks not only unpleasant (cells have become waay to wide, I can attach a screenshot if you want to look at it) but also strange because some columns differ by their width. Can this be helped?--R8R (talk) 18:21, 25 July 2020 (UTC)
- Maybe "not unpleasantly" is a mistake writing, meant to say 'unpleasantly'? I do see this, which is unpleasant indeed: the cells span wide to fill the width of the page (in my wide desktop screen). But: this is the OP request, and I am supposed to live with it ;-). Could yuou describe what you expect, re PT width in this? And to be clear: no error/break happens, right? -DePiep (talk) 22:24, 25 July 2020 (UTC)
- @R8R: multiple issues were ignited in this thread. Confusing. The micro table issue (PT overview in element infobox) was one of them, incidentally addressed today above. Later more from me. -DePiep (talk) 22:14, 25 July 2020 (UTC)
- Maybed #Micro PT in infobox clarifies. -DePiep (talk) 22:30, 25 July 2020 (UTC)
@DePiep and R8R: I understood R8R and I were supporting an 18-column table at the top of the info-box, and retaining the 32-column table at the foot of each element article. That way the reader gains a sense of familiarity from the 18-column table, then has the "aha" moment at the foot of the page, which shows where the f-block fits into the 18-column table.
The way this proposal was initiated and managed (in good faith by everyone, no doubt) is a text-book example on how not to do it. Sandbh (talk) 07:58, 26 July 2020 (UTC)
More steps
- I see that the current discussion has been confused and a new discussion would be much more focused if it was "Lets replace
with
| | ||||||||||||||||||
in Element infoboxes, making the cells in the latter a bit larger so as to fill the available width of the infobox." I like that format, despite DePiep calling it an Ikea cabinet. I think most chemists are more familiar with that version than the 32-column one. As you !vote, please mention if you support having this new thread, otherwise I won't bother. Michael D. Turnbull (talk) 13:26, 26 July 2020 (UTC)
- @Michael D. Turnbull: This is a very unhelpful comment, because is it starts a discussion while the question is Yes/No. The problem *is* unstructured discussion flow. As noted here, arguments could/should be entered in a new thread. It is useless to take shots here at any future discussion. -DePiep (talk) 13:52, 26 July 2020 (UTC)
Propose closing as is
@Michael D. Turnbull, Sandbh, Double sharp, and R8R:
- Given that this thread has caused much confusion and misunderstandings, I have #Reverted the change in {{Periodic table (navbox)}}, so as to start from a clean sheet. We are now at the status quo ante.
- I strongly propose to close this thread as is, without any prejudice (i.e., no claims or conclusion be drawn from it), to get rid of all confusion. After that, a new thread can be started preferably from a more simple statement. OK? -DePiep (talk) 09:56, 26 July 2020 (UTC)
- !Votes
I request an uninvolved editor to close this thread #A_navigation_aid_for_Wikipedia,_based_on_the_periodic_table as "no consensus" or "procedural close". (per Wikipedia:Closing discussions: # further contributions are unlikely to be helpful). Contributors, including OP, have supported the conclusion that too many issues were discussed intertwined, thereby confusing the threads of argument. This #Propose closing as is invites to restart (from status quo ante, no prejudice) with a more crisp initial question/proposal. -DePiep (talk) 12:07, 1 August 2020 (UTC)
- Hi DePiep, please create a report at WP:RFCL. Best regards, ~ ToBeFree (talk) 19:32, 1 August 2020 (UTC)
More detailed talk on easy closure
|
|---|
|
New thread
This is not an invitation for new discussion here, but a pointer to a new thread.
There are two essential issues around here: (1) what elements should be put in group 3 on Wikipedia, and (2) whether the navbox periodic table should be shown in 18 or 32 column format.
As new information has come in (a past IUPAC endorsement of the form with Lu under Y), I have started another thread below: Talk:Periodic_table#IUPAC:_an_endorsement_on_group_3_(1988). This is about the first issue (group 3 composition) only. I have endeavoured to keep it policy-based and drama-free.
To avoid confusing various issues, I suggest that the second issue about 32 vs 18 column be started in another new thread if anyone wants. I don't consider that very important myself, and therefore I have not started it. Double sharp (talk) 03:14, 2 August 2020 (UTC)
- Double sharp: is this really an extension to the main thread #A_navigation_aid_for_Wikipedia,_based_on_the_periodic_table? And how would this be a useful, late contribution since there is one conclusion drawn: confusing thread beyond repair? I don't see. -DePiep (talk) 09:40, 2 August 2020 (UTC)
- @DePiep: Maybe I did not make myself clear, so let me explain what this is. I agree that this thread covered too many issues and support closing it, which is why this is just a pointer to a new thread (beginning afresh). I mentioned above that there are two major issues that it seemed to be trying to discuss at once: (1) what elements should be put in group 3 on Wikipedia, and (2) 18 vs 32 column format for the navbox periodic table. Therefore, I started a new thread where (1) alone may be discussed. I did not start one about (2) because I consider it relatively unimportant myself, but with no prejudice against anyone else starting one. Does that help clarify matters? Double sharp (talk) 09:45, 2 August 2020 (UTC)
- Stop it. Stop this thread. By any means. Again stating "GF" arguments is the problem. -DePiep (talk) 23:42, 2 August 2020 (UTC)
- @DePiep: But it is already stopped? I simply meant to say "go to the bottom for the new thread". Double sharp (talk) 02:30, 3 August 2020 (UTC)
- Stop it. Stop this thread. By any means. Again stating "GF" arguments is the problem. -DePiep (talk) 23:42, 2 August 2020 (UTC)
- @DePiep: Maybe I did not make myself clear, so let me explain what this is. I agree that this thread covered too many issues and support closing it, which is why this is just a pointer to a new thread (beginning afresh). I mentioned above that there are two major issues that it seemed to be trying to discuss at once: (1) what elements should be put in group 3 on Wikipedia, and (2) 18 vs 32 column format for the navbox periodic table. Therefore, I started a new thread where (1) alone may be discussed. I did not start one about (2) because I consider it relatively unimportant myself, but with no prejudice against anyone else starting one. Does that help clarify matters? Double sharp (talk) 09:45, 2 August 2020 (UTC)
- Double sharp: is this really an extension to the main thread #A_navigation_aid_for_Wikipedia,_based_on_the_periodic_table? And how would this be a useful, late contribution since there is one conclusion drawn: confusing thread beyond repair? I don't see. -DePiep (talk) 09:40, 2 August 2020 (UTC)
Caption wording
I changed changed a caption, using shorter but nearly synonymous wording to avoid unnecessary line wrapping. DePiep then reverted this, noting correctly that this is a change from "which are in class X?" into "How to classify?". I have now been made aware of an ambiguity. In my WP:ENGVAR, a person asked "How do you classify selenium?" might answer in any of these ways:
- I classify it as a metal
- I classify if as a metalloid
- I classify it by considering its chemical properties
- I classify it by considering its physical properties.
Now (1) and (2) represent the sense I intended while (3) and (4) represent the sense that DePiep refers to as a change. I believe native speakers would probably think of the first sense, and most non-native speakers, given the context, would also understand that this is the sense intended. But perhaps the sense of (1) and (2) is not used in other ENGVAR. In that case I gladly accept the revert. But if I am correct, then I think the ambiguity is very slight and should be accepted in order to improve the line wrapping of the caption. YBG (talk) 15:13, 26 July 2020 (UTC)
- @YBG: do you mean this edit? ;-) Later more. -DePiep (talk) 19:41, 26 July 2020 (UTC)
- @DePiep: Thank you, yes, that was the edit I meant. I have fixed it above. 21:47, 26 July 2020 (UTC)
so you changed [5]
- | Metals, metalloids, nonmetals, and elements with unknown chemical properties in the periodic table. Sources disagree on the classification of some of these elements.
into
- | Metals, metalloids, nonmetals, and elements with unknown chemical properties. Sources disagree on how to classify some elements.
All fine with me, and yes IMO the ambiguity is real ("classify as" vs. "classify by"). Anyway, I'm fine leving it here. -DePiep (talk) 22:04, 26 July 2020 (UTC)
- When you say "all fine by me", I presume that means you're ok with my change, though you don't like it enough to restore it yourself. YBG (talk) 04:58, 27 July 2020 (UTC)
- Yes as you write, I should have included your reply and my reversal.
I wrote I'm fine with both forms because, more precise than my editsummary here I cannot discuss this orthography in classification.I'm not sure it is an ENGVAR thing, I think the wording is explicitly one way or the other (or it should be so). By now, Sandbh has responded with added a line of thought (below), and I hope this leads to fleshing out into a correct, unambiguous wording. -DePiep (talk) 09:25, 27 July 2020 (UTC) - Will pick up this, as there are some things I think I can say. -DePiep (talk) 09:55, 27 July 2020 (UTC)
- Yes as you write, I should have included your reply and my reversal.
It is not so much that they disagree. Rather, classifications may vary depending on the context i.e. criteria of interest. We don't have to worry about that since we go with the majority.
Scerri talks about it, here, when he says, "any system of classification must inevitably remain interest dependent" etc. Thus:
- "Sources may vary in their classifications, according to interest dependence."
Or something like that.
--- Sandbh (talk) 08:05, 27 July 2020 (UTC)
- The first description YBG gives appears correct to me (making a difference between (1),(2) "classify as" versus (3),(4) "classify by"). I don't think it is an ENGVAR issue, but more an imprecision in language or orthography (both are correct answers to "how do you classify ..."). Originally I preferred this ambiguity being resolved in our article, as it is a fundamental to the enwiki presentation of "categories".
- Then, Sandbh leads to an encompassing form (catching all (1)–(4) meanings). In this case, a caption of a small image, this could be OK. In other situations, e.g. describing metalloids, the difference still may be important because there, classification is the topic. Maybe it needs to have a gradual scale of inclusion ("is more a metalloid"), even with a single interest approach. Our Lists of metalloids already does a grading scale, but by a generic, tallying scale not by an "interest" scale. (I note that recently we re-classified At which now follows this Lists' order: At and Po treated the same).
- With this, I prefer Sandbh's proposal. It also prevents making a choice based on line wrapping. -DePiep (talk) 10:19, 27 July 2020 (UTC)
- @YBG: [6]: what is you response to Sandbh's reply? I saw better solution. -DePiep (talk) 18:36, 27 July 2020 (UTC)
- I had planned to incorporate his suggestion, but it proved too difficult to do on my smartphone. Hope to get to it soon. YBG (talk) 02:49, 29 July 2020 (UTC)
- I have changed it to an aggressively abbreviated form of Sandbh's suggestion: Classification may vary depending on author's focus. YBG (talk) 02:28, 1 August 2020 (UTC)
IUPAC: an endorsement on group 3 (1988)
Opening statement by Double sharp
I apologise for raising this again. I beg forgiveness for it because of the title. So, this may be taken as a start of a new and totally policy-based and drama-free discussion.
In rushing to start an RFC above based primarily on the sources debating the scientific arguments (which I now realise isn't really that relevant for Wikipedia), I appear to have overlooked what the most important organisation here has actually said about this in the past. In fact, that appears to have been completely overlooked in all the arguing back and forth over the past few months (mostly between myself and Sandbh). So I remedy this now.
In the comments under a 2019 article by Eric Scerri (for those who do not know him, he is a world authority on the history and philosophy of the periodic table, as our article on him notes), Scerri himself adds that IUPAC already endorsed the Lu under Y form in a 1988 report.
| “ | Thirdly, I should also mention that figure 3 [Sc-Y-Lu, 32 column] that I call an optimal table, was already endorsed in an earlier IUPAC report, E. Fluck, New Notations in the Periodic Table, Pure and Applied Chemistry, 60, 3, 431-436, 1988. | ” |
The periodic table the paper gives in Fig. 3 is only partially drawn, but it clearly shows the La-Yb / Ac-No f block characteristic of the Lu under Y form. And on the last page we find the following text:
| “ | The Elements of the Scandium Group
In the "Red Book" which will appear in 1988 the same arrangement was chosen for the elements of the scandium group as in the periodic table as originally proposed by CNIC and subsequently published by VCH Verlagsgesellschaft, Weinheim. It is a compromise. According to the electron configurations of the elements, the scandium group consists of the elements This was pointed out as early as 1959 by L.D. Landau (ref. 20) and later by other authors (ref. 13, 14, 20 to 25). Most periodic tables in textbooks and classrooms, however, list Sc, Y, La, and Ac as elements of the scandium group and designate the elements Ce to Lu and Th to Lr as lanthanides and actinides, respectively. The historical background for this arrangement is given in a paper by W.B. Jensen (ref. 21). Based upon their electronic configurations and their chemical and physical properties, the elements La to Yb and Ac to No should be inserted between barium and lutetium and between radium and lawrencium or for practical reasons be listed at the bottom of the table. The series La to Yb and Ac to No then, however, cannot be named correctly as lanthanides and actinides since they contain the elements lanthanum and actinium and not only elements similar to lanthanum and actinium as is purported by the ending -ide (or -oid according to an earlier IUPAC recommendation). |
” |
There apparently was no Red Book published in 1988: maybe what is meant is what became the 1990 Red Book, but I cannot get the relevant parts of its periodic tables clearly on Google Books' snippet view. There seem to be multiple ones on pp. ii, 282, and 283. I have asked at WP:REX.
However, granting that does also raise a few questions. If it was decided, then why is IUPAC not using Sc-Y-Lu now even internally, calling all the lanthanides and actinides group 3 elements in the JWP reports? Why does the 2005 Red Book show a Sc-Y-* table? Was the decision rescinded? Why is there a new task force since 2015? (Although I suppose this one is easily answerable: it could be to relook at a previously decided situation even if the earlier decision was not rescinded.)
A reasonable interpretation is that it is something like the spelling of aluminium, in which IUPAC made a decision (it was the only official spelling in 1990–3, so around the same time), and then had to allow "aluminum" as an alternative (not the first one though) when people didn't want to follow. And for Wikipedia that situation is already interesting, as we do in fact follow IUPAC's "aluminium" consistently there to avoid lame edit wars, despite the fact that the 2005 Red Book actually does not endorse "aluminium" anymore (it writes that as the name of the element, but adds a footnote that reads "The alternative spelling ‘aluminum’ is commonly used.") I do not have proof of that, but can find no other explanation that reconciles a past IUPAC endorsement with IUPAC currently not using the Lu under Y form and having a task force ongoing to decide the matter.
Therefore: currently we seem to be in a limbo where IUPAC is relooking at the situation: so, should we follow
- Lu under Y, which has been endorsed by IUPAC, but in an endorsement that doesn't seem to translate to actual use anymore (which parallels "aluminium" where we follow the old IUPAC endorsement from almost the same period anyway)?
- The placeholder * under Y, which IUPAC currently shows, but takes care to say "IUPAC has no recommendation for a specific form of the periodic table", and that we know the new task force is not considering?
- or La under Y, which appears as of the 2010s in a plurality of general chemistry textbooks, but no longer a majority as it used to have (according to the IUPAC survey, statistics listed above)?
How do others see this?
I would, as you all know by now, favour option 1. For here, not because of the science, but because of the precedent of WP:ALUM. In that case, the situation was otherwise rather impossible to resolve, as British sources obviously use "aluminium" and American ones obviously use "aluminum". So we followed an old IUPAC decision, even despite its lack of actual adherence later by IUPAC itself, and in spite of the fact that you'll always find more "aluminum" and "cesium" than "aluminium" and "caesium".
So, that seems to me rather the same situation: the situation is rather impossible to resolve, as comparing textbooks shows a distribution that changes significantly depending on decade, even if La always manages a plurality; comparing periodic tables on Google Images rather favours *; and counting articles arguing about the matter rather favours Lu. There is also an old IUPAC decision, although IUPAC itself does not seem to adhere to it anymore.
And of course, it should be kept in mind that anything we decide now will be provisional. IUPAC has an ongoing task force since 2015 and it should come to another decision, which surely we will follow on Wikipedia whatever it turns out to be.
So: it seems to make the most sense to me to follow what IUPAC actually endorsed in 1988, rather than what they show now but take care not to say they endorse, when what we are waiting for is none other than another IUPAC endorsement.
And it sidesteps any arguments about which sources are the most relevant. We can argue all day about whether the most relevant sources for periodic tables are general chemistry textbooks or the posters you usually see and get off the Internet, about whether the relevant sources are the articles focusing on the matter or what chemists standardly use, etc. etc. etc. But here we have an actual past endorsement from IUPAC, which is surely the most relevant organisation for this.
Now, there is another writing-related reason why I would prefer Lu under Y. It is a sort of WP:IAR. I will now try to express it briefly in a drama-free way. I will rewrite if anyone objects.
The problem is that when it comes to explaining periodicity, which this article should do, showing any form that is not Lu creates a contradiction. The problem is inherent in what reliable sources say when it comes to the science. I will restrict myself to three observations.
- Reliable sources agree that actually the gas-phase ground-state configurations aren't very relevant for chemistry. Most chemical processes do not involve gas-phase atoms. Actually, in chemical environments configurations change significantly. The most famous example is probably that in transition metal complexes, even neutral ones, the configuration likely has no outer s electrons, even though that is not what any transition metal atom but palladium is in the ground state. This understanding is, for example, in Greenwood and Earnshaw 2nd edition, page 960 (table of group 4 complexes; neutral ones are labelled d4). And for example Glenn T. Seaborg noted this difference: "It is important to realize that the electronic structures listed in Table 6 are those of the neutral (unionized) gaseous atoms, whereas it is the electronic structure of the ions and compounds that we are chiefly concerned with in chemistry".
- Moreover, reliable sources usually do not even define blocks, much less differentiating electrons. Neither is actually in the index for Greenwood and Earnshaw (they do mention blocks, but they don't define what they are). Cotton and Wilkinson only refer to differentiating electrons twice, and in neither case do they actually give a definition that sorts out the problematic cases like vanadium d3s2 versus chromium d5s1. Not to mention that reliable sources agree that electrons are indistinguishable: Richard Feynman says it, and it's completely standard chemistry that the electrons in a covalent bond are indistinguishable (an atom can't tell which one belonged to it and which one didn't). That does not sit well with the idea of differentiating electrons.
- And reliable sources generally derive periodicity from the Madelung rule. That demands in the sixth row "6s, 4f, 5d, 6p", as in the graphic to the right. Even the IUPAC document mentioned above says that: "CNIC rejected the proposal [for different group numbers] arguing that 1) the code of elements is rather an illustration of the Aufbau principle from which a periodic system is to be developed" (my italics, which are hopefully considered OK to use, as Sandbh has previously objected to bolding).
Now, let's not argue about whether these are right. The important thing is: they are supported by sources, they should go in. The problem is that since the La table is, as the article notes, based on gas-phase ground-state configurations and differentiating electrons, it looks rather strange to explain periodicity following sources that note that they are not relevant, and then focus on a table that came from there (and only partly, as while Ac anomaly is reflected Th is not). And it is very strange to mention the Madelung rule as the basis for the PT, and then show a table that does not follow it, and then apologise for it by referring to sources for 4f becoming non-hydrogenic at La already.
Therefore, I claim: for the twin reasons of
- the past IUPAC endorsement from 1988 of Lu under Y;
- the writing problem that appears if you don't pick Lu under Y;
Lu under Y is better for WP.
But, of course I will follow whatever consensus results. Double sharp (talk) 13:46, 1 August 2020 (UTC)
- Sounds well reasoned and drama-free to me. A few comments:
- It is not surprising that a body like the IUPAC would make an endorsement and then forget about it. They've got no language police or WP:wikilawyers.
- The parallel with WP:ALUM is instructive and convincing.
- There is bound to be a fair amount of pushback so we should have our consensus firmly in hand before thinking about changes
- It would be wise to delay if there is a significant chance the Group 3 group will deliver its decision in less than a year, especially if there's a chance they'll come up with something different.
- And of course, I'll abide by whatever consensus is reached. YBG (talk) 22:18, 1 August 2020 (UTC)1
- @YBG: Alas, I don't know how the group 3 project is going. They do post progress updates, but quite sporadically. The last one is from December 2019 and the previous one is from March 2018. As both La and Lu supporters are on it, it could honestly go either way.
- As for getting a consensus: that's why I'm raising it here. ^_^ Double sharp (talk) 03:25, 2 August 2020 (UTC)
Observations by Sandbh
I'll address Double sharp's observations in sequential order.
IUPAC: an endorsement on group 3 (1988)
Untrue. No such endorsement was ever made.
Scerri himself adds that IUPAC already endorsed the Lu under Y form in a 1988 report.
Untrue. If it were, there would be no reason for IUPAC to agree to the establishment of the Group 3 project, chaired by Scerri.
The report Scerri refers to is here. A careful reading of the report shows that it is an historical review of the processes that led to the current group numbering system. That is all. It does not have the status of an IUPAC recommendation. Rather, it was published in IUPAC's journal, Pure and Applied Chemistry. That does not mean anything; you can find varying formats of periodic tables in this journal, for example, including the La form.
Towards the end of the article Fluck makes some passing comments about group 3. He notes that the *-** form is a compromise. This is a personal opinion; he does not give a citation. Then again, Philip Ball, former editor of Nature, who is on the IUPAC Group 3 project, opined that, "Chemistry is about compromise."
Fluck correctly says, "Most periodic tables in textbooks and classrooms…list Sc, Y, La, and Ac as elements of the scandium group and designate the elements Ce to Lu and Th to Lr as lanthanides and actinides, respectively.
In support of his personal opinion Fluck notably cites Landua & Lifshitz, and Jensen. L&L are not reliable as I noted here.. Jensen was described by Scerri as too selective in his arguments.
Fluck's article was never "endorsed" by IUPAC. It was an historical review, nothing more.
Textbooks, Google Images, counting articles
Double sharp writes: "…comparing textbooks shows a distribution that changes significantly depending on decade, even if La always manages a plurality; comparing periodic tables on Google Images rather favours *; and counting articles arguing about the matter rather favours Lu.
The 2010's text-book statistics are La 16 (~50%); Lu 6 (~18%); *-** 11 (33%). Google image searches are plagued by bias arising from the false impression that the IUPAC *-** table is “official”, and unreliable sources.
There are some arguments in the literature suggesting the Lu form looks better in some cases. Many of these are one-shots based on a single property. As I noted, Jensen had a red-hot go at corralling these arguments in 1982, in support of Lu in group 3 but his effort failed to gain traction. He was criticized by Scerri, (chair of the IUPAC Group 3 project group) and Parsons (2018, p. 143), for being too selective in his arguments. There was Holden (1985) who looked at seven properties but estimated the argument of Landau & Ligshitz (1958) to be the strongest. As noted, Landau and Lifshitz, are unreliable.
The situation is rather impossible to resolve.
There is nothing needing to be resolved. The La form continues to be the most popular form in the literature. As Scerri (2020) notes: "…the majority of textbook and other periodic tables in the 18-column format show the elements of this group as scandium, yttrium, lanthanum and actinium."
1. Reliable sources agree that actually the gas-phase ground-state configurations aren't very relevant for chemistry.
Reliable sources agree gas-phase ground-state configurations are a highly successful approximation.
2. Reliable sources usually do not even define blocks
Reliable sources usually organise the elements on the basis of blocks.
3. Reliable sources generally derive periodicity from the Madelung rule.
- Correct, noting the MR does not demand anything.
- Reliable sources usually note there are 19-20 deviations from the MR.
- Reliable sources usually note the appearance of an f electron does not occur until Ce.
--- Sandbh (talk) 04:54, 2 August 2020 (UTC)
PS: @Double sharp: IUPAC Red Book 1990 here. Check out the 32-column Lu form, at page 283! Sandbh (talk) 05:12, 2 August 2020 (UTC)
@Sandbh and YBG: I will now address Sandbh's observations dispassionately.
Endorsement. As I see it, Sandbh's interpretation seems somewhat at odds with what reliable sources say.
I quoted the IUPAC 1988 report above: it explicitly says "the scandium group consists of the elements Sc, Y, Lu, Lr", and goes on to justify it by referring to earlier work by Landau and Lifschitz and by Jensen, and by referring to electron configurations as well as chemical and physical properties. And I quoted how Eric Scerri, who is on the current IUPAC project, sees this statement: as an endorsement of the form with Lu under Y. Scerri, being on the current IUPAC project, surely will be well-placed to make a statement about what this document is.
Moreover, the report is only stated to be prepared for publication by E. Fluck. As can be seen from the title page, it rather comes from the IUPAC Inorganic Chemistry Division. And it goes on to state that "this form of the periodic table will therefore be set out in the Appendix to the new edition of the IUPAC Red Book (Nomenclature of Inorganic Chemistry)". That seems like more than Fluck's personal opinion.
And indeed, Sandbh has kindly linked to the 1990 Red Book. On p. ii, it shows a Sc-Y-* form. However, as Sandbh noted above: on pp. 281–3 (the very appendix in question) it shows an 8-column, then an 18-column, then a 32-column periodic table. The 8-column one is Sc-Y-*, as in the cell reading "La*" a footnote is given reading "including lanthanoids (57-71)", and the same for the actinoids. The 18-column one is explicitly Sc-Y-* (with La-Lu* below Y). But: the 32-column one is a Sc-Y-Lu table, exactly in accordance with what the 1988 IUPAC report says. Therefore, one can see that not only was the 1988 report an endorsement of the Lu form, but it translated at least partially to actual usage.
While Fluck does use the word "compromise", he does not refer to the Sc-Y-* form. In fact he goes on to talk about how the f elements fit into the table if the scandium group is considered to be Sc, Y, Lu, Lr. And indeed mentions that many tables show Sc, Y, La, and Ac in the scandium group; however, his language is clearly not endorsing this. And even if Sc-Y-* was in fact the "compromise" mentioned (which is not at all clear from Fluck's wording), it remains that in the 32-column form where no compromise is possible without "stretchy scandium and yttrium", IUPAC shows group 3 as Sc, Y, Lu, and Lr.
There are clearly any number of reasons why IUPAC should now want to relook at its decision. Perhaps it is because new scientific evidence has appeared since 1988, say, and they want to examine that. Or perhaps, it is because the issue got fresh publicity. Or perhaps, it is as YBG said and they simply forgot about their earlier decision. I obviously do not know what the real reason is, but the fact that it is easy to come up with plausible ones rather seems to be at odds with Sandbh's statement that "If it were [true that IUPAC had endorsed the Lu form in this 1988 report], there would be no reason for IUPAC to agree to the establishment of the Group 3 project, chaired by Scerri."
Sources in the literature. Regarding Sandbh's statement that "the La form continues to be the most popular form in the literature", the IUPAC survey shows that 16 out of 33 undergraduate chemistry textbooks from the 2010s showed the La under Y form. That is indeed a plurality of 48%, but it surely cannot be taken as decisive.
Sandbh also argues that the Google Images results are plagued by bias and speculates on why they show what they do. But one can easily raise the very same concerns against textbooks. When a textbook shows the * form, is it because its authors analysed the situation and decided that was the best, or is it because they interpreted IUPAC currently showing this form as an endorsement, when they quite clearly state that it isn't? When a textbook shows the Lu form, is it because its authors really analysed the situation themselves, did they decide they were convinced by Jensen, or did they just do it because it looks neater? When a textbook shows the La form, is it because it authors really analysed the situation, or is it just because they were not aware that a dispute existed in the first place? The last option is in fact quite probable: remember, in Jensen's own 1982 article that the IUPAC report cites (doi:10.1021/ed059p634), he writes:
| “ | Indeed, in talking with his fellow chemists, the author discovered that none of them was aware of the evidence favoring the reassignment of lutetium and lawrencium or indeed that there ever was any question about their placements (a category in which the author must include himself until very recently). | ” |
The bottom line is: we are not here to second-guess why a source made a decision. We can only talk about that if they tell us, and they generally don't. What matters for Wikipedia is what they show. And that differs quite strongly depending on what sources you focus on. That is why I consider the situation undecidable on sources alone: textbooks have a plurality for La (but not a majority), Google Images searches give a majority for *, and articles arguing the issue give a majority for Lu. That's exactly the kind of situation where, as with WP:ALUM, we are willing to use old IUPAC decisions even if they are not actually adhered to completely by IUPAC itself.
Analysis of sources. The idea that periodic tables on the internet are "unreliable" also overlooks the problem that the periodic table is not solely the province of undergraduate chemistry textbooks. It's trivially easy to find tons of them in books and online, and especially the online ones are what students will most likely be looking at.
Finally, Sandbh's characterisation of the Lu arguments as "one-shots" is not something that, as far as I am aware, can be found in the literature. It should also be noted that many of these one-shot arguments in fact appear in Jensen's article. Merz and Ulmer, for instance, was called "another pop gun" by Sandbh in Wikipedia talk:WikiProject Elements/Archive 44. Yet it is one of the arguments that is explicitly mentioned in Jensen's paper: it is mentioned as "d-Block-like structure for conduction band" and cited as reference (13). So not only is Sandbh's characterisation rather at odds with how the argument was treated and reliable sources, it seems to suggest that an argument is not worthy of consideration (Merz and Ulmer) as long as it appears alone, but when it is combined with many other ones (as Jensen did) it becomes fair game. That seems strange.
If Sandbh personally is unconvinced by the Lu arguments, that is his right. Nevertheless, they are referred to in reliable sources, and mentioned as the basis for the IUPAC endorsement. We should be following the reliable sources rather than trying to discredit them by our own analyses. Any discrediting can only be done here if it comes from other reliable sources.
An example of this is where Sandbh refers to his own analysis at Wikipedia talk:WikiProject Elements/Archive 48#Landau & Ligshitz (1958): Redux to justify his contention that Landau and Lifshitz's Course of Theoretical Physics is not a reliable source regarding the matter. That is brought in despite the fact that it is not the important thing here (it's just another source); it is rather just a source that the IUPAC 1988 report cited. And even if it was the important thing, it remains that this is a world-renowned textbook that got its authors the Lenin Prize of 1962. And while I am in personal agreement with him that what Landau and Lifshitz says does not go so far as to say that only Lu belongs in group 3 (because they just say Lu doesn't belong with the rare earths, and place La and Lu in a "platinum group" that is supposed to contain the filling of the 5d shell), and rather seems to be closer to saying La and Lu both belong there, I feel his conclusion is marred by the fact that it's WP:OR, against how the IUPAC report that is under consideration interpreted it, and that he refers to a "Landau and Lifshitz periodic table" that is not what Landau and Lifshitz actually gives.
| What Landau and Lifshitz give | What Sandbh in his own analysis calls a "Landau and Lifshitz periodic table" |
|---|---|
   |
 |
Again, I refer to the precedent of WP:ALUM, and the fact that 48%, however you slice it, is not a majority. It would even not be the first time that we followed a IUPAC endorsement even when the majority of authors do not follow it. Not only is there the precedent of WP:ALUM, there is also the precedent of the systematic element names. IUPAC recommended these ("ununennium" etc.), but they are not used much among the nuclear physicists and chemists involved in synthesising these elements. Despite this, we continue to follow IUPAC.
What reliable sources say. It is rather odd to say that reliable sources agree that gas-phase ground-state electron configurations are a highly successful approximation when I have shown sources that explicitly say that they have to be used with caution and that they are irrelevant. I quote them again below in a collapse box, without the bolding that Sandbh objected to before when I posted them. In fact I add one more very recent one.
Extended content
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||
We also have a big-name inorganic chemistry textbook acknowledging it. Greenwood and Earnshaw, it is true, puts gas-phase ground-state electron configurations in its tables. But they well understand that in chemical environments these configurations change. On page 960 they list a bunch of coordination complexes of the elements of group 4. There they refer to [Ti(bipy)3] and call it a d4 complex. Here, titanium is in the 0 oxidation state: it has not gained or lost any electrons. Yet they agree that here it does not show the d2s2 configuration that it has as a gas-phase atom in the ground state.
Since lanthanum is at issue, I also point to a recent article (doi:10.1039/C3CP50717C) showing that La may use its 4f orbitals for bonding despite them being unoccupied in a gas-phase La atom in its ground state. That is significant as it is one of the main arguments authors have used in support of Lu under Y: that La has low-lying non-hydrogenic f orbitals, whereas Lu does not. It appears in Jensen's 1982 paper.
As I mentioned, this is significant for the La form, as it stems from the ground-state gas-phase anomaly in La and in Ac. We must follow reliable sources, and that implies that we should say all of this about ground-state gas-phase anomalies being irrelevant for transition metals and that they can take different configurations when chemically bound. But that sits very badly with saying that the La form is based on ground-state gas-phase anomalies. It does not help the reader's understanding to say something is irrelevant and then justify a form shown based on what we have just said is irrelevant.
It is also, to me, very surprising to see Sandbh say that "reliable sources usually organise the elements on the basis of blocks", when reliable sources often do not even bother to define what a block is. On the rare occasion that a definition is actually offered, the reliable source in question generally does not follow it. Take this text for example, by A. Ramakrishna. It says "When the differentiating electron enters the d-orbital the elements are called d-block elements". Yet it lists zinc as a d-block element just the previous page, despite the fact that its differentiating electron does not enter the d-orbital (copper, the previous element, is [Ar]3d104s1; and zinc adds an s-electron to become [Ar]3d104s2). I do not see such an inconsistent definition as support for organising the elements on the basis of blocks.
Finally, reliable sources already support the idea that it is not at all important that the first f electron appears only in a gas-phase ground-state atom of cerium, because they agree that gas-phase ground-state electron configuration anomalies in the d and f elements are not relevant for chemistry, and that lanthanum has 4f involvement in the bonding already. I have quoted them above. Again: if Sandbh is unconvinced by them, that is his right, but we are supposed to be following the statements of reliable sources here. They say lanthanum is using its 4f orbitals for chemistry: we follow. In order to override what a source says for Wikipedia, you need to at least have a more recent source that refutes what it is saying.
I do not dispute that the gas-phase ground-state configuration is a useful approximation for the s and p elements, where they typically indeed appear in chemistry courses around the world. What reliable sources question their relevance for is when it comes to the d and f elements: lanthanum is one of them.
Writing problems. I once again note to finish off: periodicity is agreed to be based on the Madelung rule. IUPAC itself said so in the 1988 report being referred to. Now, the Madelung rule states that 4f comes before 5d. Is this really well explained by a table like this?

We would first be saying that 4f comes before 5d, as that is what the Madelung rule says. But then we would have to engage in some verbiage explaining why the table shows one 5d electron coming before 4f. And then we would have to find some way to explain why it doesn't also show two 6d electrons coming before 5d, given the electron configurations in the early part of the f block:
| La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 4f05d16s2 | 4f15d16s2 | 4f35d06s2 | 4f45d06s2 | 4f55d06s2 | 4f65d06s2 | 4f75d06s2 | 4f75d16s2 | 4f95d06s2 | 4f105d06s2 | 4f115d06s2 | 4f125d06s2 | 4f135d06s2 | 4f145d06s2 |
| Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No |
| 5f06d17s2 | 5f06d27s2 | 5f26d17s2 | 5f36d17s2 | 5f46d17s2 | 5f66d07s2 | 5f76d07s2 | 5f76d17s2 | 5f96d07s2 | 5f106d07s2 | 5f116d07s2 | 5f126d07s2 | 5f136d07s2 | 5f146d07s2 |
While it's true that lanthanum adds a d electron rather than an f one, and so does actinium: so does thorium. The La under Y table, although stated to be based on ground-state gas-phase electron configuration anomalies, only really reflects two: La and Ac. Many other elements are simply not reflected, such as Th, Pa, U, and Np. The d block in fact has more such anomalies.
And it would then have to follow reliable sources and explain that actually it doesn't matter because ground-state gas-phase electron configuration anomalies for d and f elements don't mean anything much for real chemistry. And then follow reliable sources and say that the activity of 4f orbitals begins at lanthanum, therefore cancelling out all the explanations previously stated. At every sentence a "but this, but this, but this". Surely it will confuse the lay reader to say all of this and then walk it all back.
Whereas: if we made the change, we could simply use a table like this.
No contradiction. We say the Madelung rule says 4f comes before 5d, and we show a periodic table that shows that. No problem, no self-contradictions, and no need to drill down into all sorts of complicated things that we eventually have to follow reliable sources by declaring irrelevant anyway. Of course, we may discuss in the notes the "shifted" f block characteristic of the Sc-Y-La table, but it can be left in the notes and not to bamboozle a beginner just learning this from our article.
For this reason, I claim that sticking to the Sc-Y-La form would not help to improve Wikipedia. Although reliable sources have not switched en masse to Lu, they generally agree with the observations that those who support Lu in the literature make, and disagree with the observations that those who support La in the literature make. I might guess it's because the reliable sources were unaware that the dispute existed, but again, that's not the point. The point is that writing about chemical periodicity becomes very problematic with the Sc-Y-La form as a default because when justifying it you come to a head-on collision with what reliable sources agree on. Therefore, I plead WP:IAR:
| “ | If a rule prevents you from improving or maintaining Wikipedia, ignore it. | ” |
| — WP:IAR | ||
Combined with the fact that Lu under Y has had actual IUPAC endorsement according to Scerri (who, being on the actual IUPAC project, surely must know quite a bit about what IUPAC has or has not done), which makes this not even in the first place ignoring any rules about reliable sources, I consider the case for changing back to Lu under Y quite strong.
Conclusion. After reading Sandbh's comment I see no reason to change my stand on this, and continue to support the reversion to the 2016 situation of Lu under Y as a default form. That is based firstly on what seems to be an actual IUPAC endorsement, even if the extent of its usage is a bit small, because of the WP:ALUM precedent; and secondly because of the writing problems that result if you want to follow reliable sources but not give Lu under Y. Naturally, other forms will continue to be discussed where appropriate, and the decision should be relooked at if IUPAC's new project comes to a different conclusion.
Double sharp (talk) 07:09, 2 August 2020 (UTC)
Discussion by Michael D. Turnbull, YBG, Double sharp
Scerri has recently (March 2020) published online the preprint of an article [7] that appears to show the way his thinking is going. He advocates for a 32-column table essentially identical to the one immediately above (his Figure 10) and for the same reason: not wanting to split the d block. He suggests that an 18-column table should be less preferred but, if used for example for teaching purposes it should that of his Figure 4, which is a Sc-Y-Lu-Lr version with a pair of 14-element f sets shown below. My personal conclusion from all this is that we should wait as the matter may well be settled by the IUPAC working group within a few more months. Michael D. Turnbull (talk) 18:05, 2 August 2020 (UTC)
- +1 Sandbh (talk) 00:24, 4 August 2020 (UTC)
- Wondering if we are using the phrase "reliable source" here in the sense used here at WP. WP:RS Primarily deals with which sources can be cited on WP. Secondarily it helps us identify things that should be excluded because they don't appear in RS. And thirdly, relative prominence in RS should determine relative prominence in WP, lest we give it WP:UNDUE WP:WEIGHT.
- This last wikilink is most instructive. See especially the last two paragraphs of the section beginning here:
Keep in mind that, in determining proper weight, we consider a viewpoint's prevalence in reliable sources, not its prevalence among Wikipedia editors or the general public.
If you can prove a theory that few or none currently believe, Wikipedia is not the place to present such a proof. Once it has been presented and discussed in reliable sources, it may be appropriately included. See "No original research" and "Verifiability".- ~ YBG (talk) 01:40, 3 August 2020 (UTC).
- @YBG: Lu under Y is not something that "few or none currently believe". It is something that has been believed since 1892 by various chemists and has gotten the endorsement of the relevant organisation itself, IUPAC, in 1988. True, it is not the most common form in chemistry textbooks (21% of textbooks published since the turn of the millennium show it according to the IUPAC survey). But if you look at the statistics above, you will see that since no form has a majority anymore: anything as default produces an undue weight problem: it is unavoidable. That's why I favour saying "look, IUPAC said this in 1988".
- Especially when you take into account the following situation: although reliable sources don't have a majority showing the Lu table, they absolutely do support en masse the principles behind it. It is completely standard in the literature that the bases for the La table, which are gas-phase ground-state differentiating electrons, are irrelevant for the d and f block elements. I quoted four articles and one textbook to support that. And yet, it is completely standard in the literature that the basis for the Lu table, Jensen's concept of valence electrons and vacancies as well as periodic trends, is correct and important. The first is just HOMO/LUMO, and the second is well known to all freshmen. The Madelung rule is completely standard too, but it is only consistent with a Lu table because it says 4f comes before 5d. There are yet more, but I will just stop here. Giving the La table on Wikipedia means that the requirement of following reliable sources will force us to subvert it all the time simply because those reliable sources agree that its bases are incorrect. We are faced with a choice of the following alternatives:
- Stay with La under Y, with a 48% plurality of chemistry textbooks, but only a 1/6 minority of periodic tables from Google Images results, and against IUPAC's 1988 endorsement and against the majority of the sources focusing on the issue. And which will come in direct conflict with even the textbooks showing La under Y itself, because the bases of the La form are themselves often refuted by those textbooks.
- Go back to Lu under Y. As we had in 2016, as IUPAC endorsed in 1988, as they put in 32 column form in the 1990 Red Book. In complete agreement with the literature focusing on the issue. And in complete agreement with the points even textbooks using La make regarding the change of electron configurations of transition elements in chemical environments, which certainly ameliorates the problem of not following their table layout. In terms of Google Images results it is not worse, having about the same prevalence as the La under Y form.
- It seems to me that (2) is clearly better.
- As above: I looked at reliable sources, I quoted what they say directly. It is explicit enough without any need for my own analysis. Sandbh seems to be using his own WP:OR and selective analyses to try to discredit them. One can see that from how:
- Sandbh doesn't accept a statement from IUPAC in 1988 that goes "the scandium group consists of the elements Sc, Y, Lu, Lr", and a written statement from Scerri, the chair of the current IUPAC project on group 3, that says that it was an endorsement. Instead Sandbh says that "no such endorsement was ever made" in direct contradiction with the chair of the current IUPAC project on group 3 (Scerri), who is obviously a reliable source on what IUPAC did or did not endorse in the past.
- And how Sandbh attempts to discredit the Google Images result (with a 2/3 majority for * under Y, and about equal 1/6 shares for La under Y and Lu under Y each) of being "plagued by bias arising from the false impression that the IUPAC *-** table is “official”", but fails to note that a statement in Jensen's 1982 article quoted by me above immediately implies that a survey of textbooks that extends before his article of 1982 and the IUPAC endorsement of 1988 will be plagued by bias arising from chemists not having been very aware of this dispute (even though it was already known among the physicists then). That is indeed why I only refer to the figures from the 2000s and 2010s, as by then Jensen and WebElements would have roused chemists' attention to the fact that the issue exists.
- And how Sandbh dismisses Lu arguments in the journal literature as one-shots, never mind that that characterisation AFAIK appears nowhere in the literature, that Jensen included in his 1982 article exactly some of those one-shots, and that IUPAC referred to Jensen's article among other sources (including Landau and Lifshitz, see point 6 below) to justify their 1988 endorsement.
- And how Sandbh mentions that Scerri and Parsons considered Jensen's arguments selective, while not mentioning that Scerri and Parsons actually support Jensen's conclusions anyway, albeit for different reasons.
- And how Sandbh claims that reliable sources agree that gas-phase ground-state configurations are a highly successful approximation, when I have managed to produce four articles and one textbook substantiating my point that they are not so for the transition elements.
- And how Sandbh attempts to discredit the reliability of a renowned textbook on physics (Landau and Lifshitz) on this matter, on the grounds of his own WP:OR analysis that contradicts how the IUPAC report interprets Landau and Lifshitz's statement, and that involves drawing a "Landau and Lifshitz periodic table" that appears nowhere in Landau and Lifshitz.
- Is this neutral? Is this correctly reflecting the sources?
- We are waiting for IUPAC, indeed. We have no indication of when they will say something, given the sporadic updates of the project, and given that Scerri has been using this argument to advocate Sc-Y-Lu since at least 2009. And indeed, though Scerri is the chair of the current IUPAC task group on group 3, he was complaining about things in the very 2019 article that alerted me to the fact that IUPAC did endorse Sc-Y-Lu in 1988 already:
| “ | Why will IUPAC not see things quite so simply? That’s a big and complicated question which I can only touch upon here. Like many organizations with rules and regulations, when push comes to shove, decisions are made by committees. As a result, the science takes second place while the various committee members vie with each other and ultimately take votes on what periodic table they should publish. Unfortunately, science is not like elections for presidents or prime ministers, where voting is the appropriate channel for picking a winner. In science there is still something called the truth of the matter, which can be arrived at by weighing up all the evidence. The unfortunate situation is that IUPAC cannot yet be relied upon to inform us of the truth of the matter concerning the periodic table. In this respect there is indeed an analogy with the political realm and whether we can rely on what politicians tell us. | ” |
| — Eric Scerri | ||
- That's why I think we cannot say anything about the progress of the project, being outside it. So why not follow in the meantime the last thing that IUPAC endorsed in 1988, and put in their 1990 Red Book: group 3 as Sc, Y, Lu, and Lr? Double sharp (talk) 02:25, 3 August 2020 (UTC)
- @Michael D. Turnbull and Sandbh: Forgot to ping you, sorry. Double sharp (talk) 02:41, 3 August 2020 (UTC)
Contribution #2, Sandbh
Endorsement
- Fluck’s saying that group 3 is Sc-Y-Lu-Lr was a personal opinion, relying on questionable sources.
- Eric seeing Fluck’s statement is a personal opinion, not connected with his role as the Chair of the IUPAC Group 3 project.
- Fluck referring to this form of table being in the new edition of the IUPAC Red Book referred only to the new group numbering scheme.
- The 32 column table appearing in the 1990 Red Book came with a caveat that IUPAC does not advocate any form of table.
- IUPAC is looking at the group 3 question in response to Eric’s request to be able to do so.
Sources in the literature
- The IUPAC survey spanned the period from the 1970s to the 2010s. You have to remember that the chemistry establishment includes chemists who studied in the 1970s or earlier. The project did not therefore confine its survey to the 2010s.
- There is no profit in deciding we are in a position to decide the reliability of close to 200 textbooks.
- The fact that when Jensen spoke to some chemists and found that they were not aware of any controversy around group is because, effectively, there wasn’t any.
- As Scerri has observed, Jensen was too selective in his arguments.
- Landau & Ligshitz, as noted, IMO, cannot be relied on. On the one hand, Double sharp refers to them as not an important thing. On the other hand, he refers to it "as a world-renowned textbook that got its authors the Lenin Prize of 1962." You can now find my commentary about L&L at the Internet Database of Periodic Tables, run by Dr Mark Leach.
Analysis of sources
Yes, Merz and Ulmer, for example, is not worth considering. They said La does not have a typical d-block-like structure for its conduction band. Never mind non-typical conduction band structures can be found elsewhere (Ca-Ba; Group 11; Bi). Never mind x-ray isochromats of Gd to Lu do not support Merz and Ulmer's conclusion that Lu is more favourably placed in group 3. In fact, over half the lanthanides–not just lutetium—have conduction band structures that are more characteristic of transition metals such as hafnium. Double sharp knows this.
What reliable sources say
- The fact that all of five sources say gas phase configurations should be used with caution (I agree!) and that they are irrelevant, is irrelevant compared to the thousands of sources that use them. Even Scerri (pers. comm.) has noted that gas phase configurations are a highly successful model.
- Yes, indeed "reliable sources usually organise the elements on the basis of blocks", and they often do not even bother to define what a block is. That is because it is self-evident.
- The few sources that question the relevance of the delayed appearance of the first electron are noise, compared to the thousands of textbooks that treat this as non-controversial and show La under Y.
Writing problems
- Periodicity is not based on the Madelung Rule. Periodicity is approximate only. The MR is an approximation of the approximate periodicity seen among the elements.
- Worrying about the MR is an example of what Schwerdtfeger, Smits & Pyykkö (2020) refer to as an unnecessary dispute.
- No, Scerri does not necessarily know what IUAPC has or has not done.
The 2019 article by Scerri
You are reading things into this article when you do not have sufficient awareness of the background to it. --- Sandbh (talk) 07:11, 3 August 2020 (UTC)
Response #3, DS
Unfortunately, Sandbh's above response continues to show him engaging in his previous behaviour: using WP:OR to discredit reliable sources, both by misunderstandings of the sources and using them only selectively.
- IUPAC's endorsement
Sandbh claims that I am reading things into Scerri's 2019 article and comment when I supposedly do not have sufficient awareness of the background to it. This is a rather weird statement considering that I simply reported what he said. Which is, again, as follows:
| “ | Thirdly, I should also mention that figure 3 that I call an optimal table, was already endorsed in an earlier IUPAC report, E. Fluck, New Notations in the Periodic Table, Pure and Applied Chemistry, 60, 3, 431-436, 1988. | ” |
| — Eric Scerri | ||
From this, I do not see how my conclusion "Eric Scerri says that the 1988 IUPAC report was an endorsement of Lu under Y" could be considered as "reading things into this article". It is literally right there in the author's own comment to it.
And even though I quoted Scerri complaining about politics vs science, I specifically said after the quote: "That's why I think we cannot say anything about the progress of the project, being outside it". That seems rather the opposite of reading things into the article. It is rather a case of me refusing to do so.
Now, let us examine Sandbh's statement that I do not have sufficient awareness of the background to it. This is correct, of course. I don't know anything about the background as I am not on the current IUPAC project. But that is also, of course, totally irrelevant for Wikipedia. As shown above, I have read nothing into it when it comes to the crux of whether IUPAC endorsed the Lu form in 1988 or not. Scerri writes that it did, and I simply reported that. The 1988 IUPAC report goes so far as to centre "Sc, Y, Lu, Lr" when it says the scandium group should have that composition (it refers to electron configurations as well as physical and chemical properties to justify why). Why should the background be important for Wikipedia? It is not in the reliable sources. Any attempt to figure out what is not written is original research.
And what of Eric Scerri himself? His article is indeed not being written by him in his capacity of the Chair of the IUPAC Group 3 project. But by virtue of the fact that he is on that project, he seems better placed than any of us editors here as to know what IUPAC has or has not said in the past. Why should Sandbh's opinion have more weight than what Scerri writes regarding whether Fluck's article was a IUPAC endorsement or not?
That IUPAC notes in the 1990 Red Book that it isn't advocating any form is not relevant. Scerri referred to the 1988 report, not the 1990 Red Book, as the endorsement. It is only a confirmation as the 1988 report says "This form of the periodic table will therefore be set out in the Appendix to the new edition of the IUPAC Red Book (Nomenclature of Inorganic Chemistry), but will be related to those alternatives used most widely in the literature." And indeed it does appear there in 32-column form.
- Reading things into sources
Indeed, it is Sandbh who continually in this discussion reads things into sources in order to discredit those that don't show a La table. As one example, he says above "Google image searches are plagued by bias arising from the false impression that the IUPAC *-** table is “official”, and unreliable sources." But the periodic tables found on such a Google Image search generally do not tell us why they decided to show group 3 the way they did. Sandbh reads a reason into them for it that is not there, with no reliable source backing him. Whereas, when I pointed out that sources from the 1970s are by definition going to be outdated because Jensen's 1982 article postdated the 1970s, he simply says that "the chemistry establishment includes chemists who studied in the 1970s or earlier". Then what about that old idea that hypervalent molecules like PCl5 have d-orbital involvement in phosphorus? Many old textbooks will tell you that. Of course we know by now it's not true. Does the old and incorrect understanding still count as part of the "chemistry establishment" even so? Are those articles refuting the idea of expanded octets with d orbitals noise compared to the ever-present textbook error that such things exist? How far do we go back? Nascent hydrogen? Phlogiston?
The bottom line is: before Jensen wrote his article in 1982, most chemists did not know about this issue. That is not my own analysis, it is what Jensen says himself. I did not read that into him; it was right there, and I quoted it above. So why refer to chemistry textbooks before 1982? With a few exceptions, they likely won't reflect what chemists thought about the group 3 dispute at all because at the time it was not on the radar of most chemists.
Of course that does not mean that "effectively, there wasn’t any" dispute, as Sandbh would have it. There was a lot. Bury (1921). Shemyakin (1932). Landau and Lifshitz (1958). Seel (1961 and 1969). Hamilton and Jensen (1963). Hamilton (1965). Matthias et al. (1967). Merz and Ulmer (1967). Chistyakov (1968 and 1970). Luder (1967 and 1970). Wittig (1973). And I got those all just from the reference lists of the IUPAC endorsement of 1988 and the Jensen article of 1982. It just did not flash on the radar of most chemists, maybe because many of these were physicists. If we push back even further there are even the tables of Bassett (1892) and Werner (1905) which would qualify as Lu under Y if not for the tiny problem that Lu had not even been discovered(!).
Above and below I showed and will show how Sandbh tries to discredit two of these (Landau and Lifshitz 1958, and Merz and Ulmer 1967), not through referring to other reliable sources to refute them as outdated, but rather by means of original research and misrepresenting the source.
- Selective usage of sources
Sandbh moreover treats sources with great selectivity, quoting an author when he says something that seems to support the La form, but not when he seems to support anything else. For example: he mentions that Scerri and Parsons criticised Jensen's 1982 article as being too selective in his arguments for Lu under Y. But he fails to mention that Scerri and Parsons, in an article in Mendeleev to Oganesson where these criticisms appear, support Jensen's conclusion of Lu under Y through other arguments. And while he is willing to quote Scerri when it comes to discrediting Jensen's arguments, he tries to discredit Scerri's statement that IUPAC endorsed the Lu form in 1988 as Scerri's personal opinion only.
How can we square Sandbh's statement "There is no profit in deciding we are in a position to decide the reliability of close to 200 textbooks" with his deciding that he is in a position to decide the reliability of Landau and Lifshitz, and Merz and Ulmer?
- Landau and Lifshitz
Now, let me address the point about Landau and Lifshitz. What I said about Landau and Lifshitz was:
| “ | An example of this is where Sandbh refers to his own analysis at Wikipedia talk:WikiProject Elements/Archive 48#Landau & Ligshitz (1958): Redux to justify his contention that Landau and Lifshitz's Course of Theoretical Physics is not a reliable source regarding the matter. That is brought in despite the fact that it is not the important thing here (it's just another source); it is rather just a source that the IUPAC 1988 report cited. And even if it was the important thing, it remains that this is a world-renowned textbook that got its authors the Lenin Prize of 1962. | ” |
Okay, rereading it now, indeed this was unclear. So let me clarify its meaning. Here is what I meant, expressed more discursively.
First of all Sandbh's attempt to discredit its reliability is misplaced, because the important source that is really the crux of why I am proposing this is that IUPAC endorsed the Lu form in 1988. Landau and Lifshitz merely gain some peripheral relevance because they are one of the sources that IUPAC cited to justify its endorsement.
And second of all, even if Landau and Lifshitz were the crux of the matter, Sandbh's attempt to discredit its reliability would become rather astonishing. This is not any old book. It is a book called "renowned" in Science and "celebrated" in American Scientist. As I mentioned: it won its authors the Lenin Prize of 1962, the first time that was ever awarded for teaching physics. In short: this is a classic text. IUPAC has interpreted it as supporting group 3 as containing the elements Sc, Y, Lu, and Lr in its endorsement of 1988. What is Sandbh doing trying to discredit its reliability? And, most importantly, what is Sandbh doing by referring to a thread in which he purports to do that, but in the process draws a so-called "Landau and Lifshitz periodic table" when Landau and Lifshitz did not give a periodic table in their discussion? And when that "Landau and Lifshitz periodic table" is extended by Sandbh beyond curium, the last element Landau and Lifshitz actually gave a configuration for, thus necessitating Sandbh to read into Landau and Lifshitz's minds to figure out how they would deal with the unprecedented anomalous electron configuration of lawrencium ([Rn]5f146d07s27p1 rather than [Rn]5f146d17s2)? Not to mention that it would require more than just mind-reading, since this anomalous electron configuration was first predicted in 1971 when Landau had already passed away, and it only got clear experimental support in 2015 when Lifshitz had also already passed away?
And what does Sandbh say above? "Landau & Ligshitz [sic], as noted, IMO, cannot be relied on." In his opinion? Why is his opinion more reliable than the fact that this is a standard text and how IUPAC saw it?
- Merz and Ulmer
Sandbh likewise uses his own WP:OR to dismiss Merz and Ulmer as "not worth considering". Despite the fact, of course, that they got published in an academic journal and are certainly a reliable source on those grounds. And that Jensen cited them and included their argument in his 1982 article. And that the IUPAC report, which despite what Sandbh says is a report and not just Fluck's personal opinion, cited Jensen's article as justification for its endorsement of Lu under Y. But to Sandbh that is all irrelevant because his original research claims it doesn't work. Not to mention that his original research irrelevantly brings in the lanthanides from Gd to Yb inclusive, forgetting that none of them are actual candidates for being placed under yttrium: what is relevant is only La or Lu. And finally he claims I know this. Why? Because when he and I agreed on La in the past we mentioned that in that old 2017 submission to the IUPAC group? Then what about me having explicitly said that I have since learnt more and withdraw my support for it?
- Wikipedia policy
Is any of this behaviour of Sandbh in accordance with the Wikipedia policies of WP:RS, WP:NOR, and WP:NPOV? Sources, if reliable, should not be selectively quoted to support an editor's bias. And no amount of original research will discredit a source for Wikipedia. Noting that a source both supports a statement and its opposite might do the trick, noting that more recent reliable sources say something else would too, but coming up with a new and original counterargument will not do.
I do not propose to do that at all. Indeed, I have mentioned above that even though I would prefer to describe Madelung's rule following the Lu table due to its concordance with it (Lu-Lr), I would add a note or even a full-blown extra paragraph detailing that many authors use a La table instead. And then discuss it appropriately using reliable sources. Rather than sweep inconvenient facts under the rug without a good reason, such as a later, very reliable source saying "actually this view is obsolete".
- Gas-phase ground-state electronic configurations
Sandbh refers to the fact that many sources do still continue to give gas-phase ground-state electronic configurations. Indeed, they do. But what consequences are drawn from this? Nothing without contradiction, so far as I can see.
Let us consider what Greenwood and Earnshaw says about such anomalies. Let us take for example the nickel group, because it has a notable total mismatch in such configurations. Nickel is d8s2, palladium is d10s0, and platinum is d9s1. Greenwood and Earnshaw show these in their table on p. 1148. And what conclusions do they draw from it? Absolutely nothing. They don't even mention it in the main text. Rather on p. 1150 they list complexes, including neutral ones. In which, corroborating my point, [Pt(PPh3)3] is given as an example of a Pt(0) complex and assigned to d10, which is not the configuration of a bare Pt atom.
Now, Greenwood and Earnshaw do consider it for the copper group. On p. 1176 Greenwood and Earnshaw say about the stable members of the Cu group "This is the first triad since Ti, Zr, and Hf in which the ground-state electronic configuration of the free atoms is the same for the outer electrons of all three elements". Except that that's an inaccurate statement because Mn, Tc, and Re are another such triad. It's just that they don't think it is because in their tables Tc is claimed to be d6s1, while Mn and Re are claimed to be d5s2 (p. 1043). But then again their statement is still inaccurate even from the data they used because on p. 979 they claim that V, Nb, and Ta are all d3s2, when Nb is now known to actually have a d4s1 ground state. No one can blame them about that, as the lowest J-level of the d3s2 configuration is literally only 0.14 eV up from the ground state. So their observation is not even true on two levels: it is contradicted by the data they themselves use, and the data they use has been superseded by better data since, but the better data also contradicts their observation. Not much analysis is needed for that, it is an obvious contradiction with publicly accessible data. Unlike what Sandbh has to do to discredit Merz and Ulmer.
Now, do they draw any consequences from this tidbit? They try once, where they claim on p. 1177 that the physical properties of the coinage metals Cu, Ag, and Au can be related to the d10s1 configuration. But that is refuted by the Fernandes et al. article I cited above:
| “ | The elements of the coinage family have an electronic configuration given by (n-1)d10ns1 and are said to have irregular electronic structure (with respect to the electronic structure and orbital ordering based on the hydrogen atom). This means that, for this family, the reference line lies in the s orbital while the d orbital should be lower in energy and narrow since all d components are doubly occupied (as shown in Figure 1). Given that picture, these atoms should present a metallic band similar to that of an alkali metal when forming a solid, i.e., each atom contributes with one electron to the chemical bond, which is constructed with s and p orbitals and thus forms an sp band with relatively low occupancy. The d orbitals of each atom are completely filled and, therefore, should not directly participate in the bonding, only providing certain freedom to the external electron in the s orbital. This would lead to a metal which should have a relatively high electron conductivity, but also very reactive since the binding energy involves essentially one electron per atom.
Obviously, this is not an accurate description of copper, silver and gold solids which have characteristics of noble metals and are among the most conducting metals. Then what went wrong in the analysis we used? Maybe it is too simple to capture the qualitative behavior of the chemical bond or, maybe, it is missing some information. At least, in this case, the analysis is missing a very important piece of information: the transition metal elements are not adequately represented by a single electronic configuration. This happens in atoms and other systems in which energy levels are close to each other or when two different configurations lead to the same energy. This situation arises in transition metal atoms due to relatively close d and s orbital energies. Each electronic state of a given transition metal atom is probably not well represented by a single configuration, but instead, it is a combination of two or more configurations. Let’s take the coinage metals as an example: the copper electronic ground state is not best represented by 3d104s1. A better representation of such an electronic state would be 90%(3d104s1) + 10%(3d94s2) (obtained with the solution of the non-relativistic Schrödinger equation under the MRCI/aug-cc-pwCV5Z approximation). This means that bonds involving copper atoms should present characteristics of both configurations. The first one was described above: it leads to an alkali-like metal with a high mobility electron per atom. We shall now analyze the latter configuration, 3d94s2. This leads to a very different bond scenario: the reference line is at the 3d orbital, i.e. above the 4s and thus the sp band can accommodate more electrons. Besides that, one of the d orbital components participates directly in the bonding, creating a slightly broader d band which would be occupied below the reference line (one unpaired d electron per atom). If the former configuration can be related to the alkali metal, the latter is to some extent related to the alkaline earth metal, since it has two electrons in an s orbital that will populate an sp band. However, the binding electrons from the d band can flow to the sp band and increase the binding energy. In this case, the electrons in both the sp and d bands would enhance the chemical bond and also the number of electrons available for conduction. This means that the solid formed by this configuration would have much more binding energy than an alkaline earth metal (considering the same period of the periodic table) with a comparable or higher conductivity due to its three electrons per atom. Combining the solids that would result from both 3d104s1 and 3d94s2 configurations isolated, one has a more complete picture of the characteristics of copper: a high conducting metal (from 3d104s1) with high binding energy (from 3d94s2). Since the latter contributes only with 10% of the atom behavior, the characteristics of this configuration are attenuate in the resulting solid, making it only regular in its binding energy and mechanical properties when compared to other transition metals, but sufficient to reduce its reactivity that would result from a low binding energy per atom. Another effect that would increase the reactivity is the excess of binding sites unoccupied, but this is not an issue for this system. |
” |
| — G. F. S. Fernandes, L. A. Cunha, F. B. C. Machado, and L. F. A. Ferrão, The Chemical Bond across the Periodic Table (2020) | ||
It's not just my original research saying "let's supersede this source because I have my own argument against it, even though that never appeared in the literature". I refer to a later source that refutes this one. Not to mention that Greenwood and Earnshaw themselves weaken their own statement on p. 1177 itself in the footnotes by themselves referring to the sp conduction band to explain the colours, which rather contradicts the idea that things come from a d10s1 configuration with no p orbitals involved.
And indeed, when explaining the valencies of the coinage metals on p. 1180, they take care to focus on ionisation energies and hydration enthalpies, rather than attempt to derive things from gas-phase ground-state electronic configurations. Indeed, they take care to note that CuII is more stable than CuI in aqueous solution despite the latter's d10 configuration.
So, where is the relevance? When reliable sources just put the configurations in tables, very rarely actually refer to them, and the one time they do refer to them proceed to contradict themselves? And when the instance that is being referred to is exactly an instance refuted by later sources with an explanation?
And is a personal communication from Scerri a relevant source for Wikipedia, considering that no one here can verify it?
- Differentiating electrons
And that's not even getting into the murky notion of differentiating electrons. Which occurs precisely twice in Cotton and Wilkinson and does not even appear in the index of Greenwood and Earnshaw. And for which I have never even seen any reliable source clearly defining them across the whole periodic table. Where does any reliable source define what the differentiating electron between vanadium 3d34s2 and chromium 3d54s1 might be?
If the La form is based on ground-state gas-phase configurations and differentiating electrons, then this evidently shows rather that its bases are totally at odds with what reliable sources say about the elements outside the main groups. Faced with a choice between (1) following La under Y with a plurality of textbooks only that vanishes once you look at periodic tables in other places, whose bases are contradicted by reliable sources, and (2) following Lu under Y which was endorsed by IUPAC in 1988 and whose bases are consistent with reliable sources, (2) is clearly better.
As for the delayed start of La under Y, Sandbh refers to "thousands of textbooks" when the IUPAC survey shows only 130 such. There are actually a few interesting questions about this that it would be good to know the answer to. How many such textbooks give the standard Madelung rule with 4f before 5d (and if so how do they deal with the contradiction?), and how many edit it to claim that one 5d electron hangs up before 4f is filled (yes, that's sometimes done, see the 2006 edition of Zumdahl and Zumdahl's Chemistry)? And how many note the contradiction that such an edit makes with the general statement of the rule: "electrons fill in orbitals with increasing n+l; if two orbitals have the same value of n+l, the one with the lower value of n fills first"?
Why should we be forced to leave a gaping contradiction or go off on a tangent when explaining the Madelung rule on Wikipedia, when we could just solve all these problems by taking the form that IUPAC endorsed back in 1988 as a default? It even makes complete sense since we are doing this as an interim measure before IUPAC's current project makes a new recommendation.
- Blocks
What a block is is not "self-evident", despite what Sandbh says. The fact that this debate has lasted nearly a century and has at stake whether lanthanum is an f-block or d-block element is rather self-evident proof that it is not that obvious. Sandbh has a rule for block start indeed. According to Wikipedia talk:WikiProject Elements/Archive 42, it is "a block starts upon the first appearance of the applicable electron" (must be in the gas-phase ground-state, or else La goes straight to the f-block). I am not aware of this appearing in any reliable source anywhere. (It comes from that submission I made to IUPAC with him when I supported La, but again: I withdrew my support for it when I learnt more.) And I and Droog Andrey have been questioning this definition greatly in that very archive. How can Sandbh still claim, after all that, that it is "self-evident"?
Indeed, reliable sources often refer to blocks. But where is the reliable source defining them that does not contradict its own definition?
- Periodicity and the Madelung rule
Let us see what the IUPAC Commission on the Nomenclature of Inorganic Chemistry had to say on the matter.
| “ | On August 19, 1987 CNIC heard delegates from the Deutsche Zentralausschuß für Chemie, the German National Adhering Organization to IUPAC, and the American Chemical Society on this matter. Prof. Brodersen, University of Erlangen, Germany, recommended the use of the "code of chemical elements" (ref. 14) as the periodic system. CNIC rejected the proposal arguing that 1) the code of elements is rather an illustration of the Aufbau principle from which a periodic system is to be developed... | ” |
| — E. Fluck, New notations in the periodic table (1988) | ||
That's rather in contradiction with Sandbh's statement "Periodicity is not based on the Madelung Rule".
The periodic table comes from the Aufbau principle, because chemical periodicity is based on electronic structure, and the Aufbau principle predicts that. Of course, for d and f elements what is more important is the total number of valence electrons and their valence orbitals, rather than minutiae of exact ground-state gas-phase configurations. That's why you can see that anomalous configurations often make precisely no difference to the chemistry of the d and f elements concerned. That is the understanding of the recent reliable sources focusing on the matter.
- Conclusion
Sandbh's selective usage of and misreading of sources is something that he already did in that long, long four-archive discussion at Wikipedia talk:WikiProject Elements. I mentioned that he was taking sources out of context, Droog Andrey (an actual chemist) also noted that some of his sources did not support what he was using them for. It is, in fact, a significant part of why I eventually lost my patience with Sandbh.
Now, I did go too far above: I apologised, we went back to a gentlemen's disagreement. I would like to keep it as a gentlemen's disagreement, as we have fruitfully collaborated on Wikipedia before. And so far it has been simply discussion. But now the content of the article is at stake regarding which form should be taken as a default. And if Sandbh should continue to in the above way favour his own original research over explicit statements by reliable sources when it comes to the content of the article, then I am sorry to say that I will have no choice but to reluctantly escalate this dispute to WP:ANI. Double sharp (talk) 09:55, 3 August 2020 (UTC)
- @Michael D. Turnbull, Sandbh, and YBG: Forgotten pings again, my apologies. Double sharp (talk) 09:55, 3 August 2020 (UTC)
Contribution #3 by Sandbh
| Round | DS post | Sandbh response |
|---|---|---|
| 1 | 2,050 | 620 |
| 2 | 3,830 | 590 |
| 3 | 4,385 |
It's unfortunate this topic takes up so much wordage. Addressing Double sharp's contributions now takes me so long it's starting to impact my RL obligations. The table shows the cause.
When I can I'll see if I can give some concise observations.
Double sharp and I, as he said, have worked together productively on other matters. No doubt we will continue to do so. And I look forward to it.
His threat to take this particular matter to WP:ANI leaves me speechless. --- Sandbh (talk) 01:00, 4 August 2020 (UTC)
- @Sandbh: As I see absolutely nothing in your posts that addresses your misuse of sources, I will now be forced to act on the threat. Double sharp (talk) 01:24, 4 August 2020 (UTC)
ANI thread
Please see WP:ANI#Misuse of sources by User:Sandbh (started by me). Double sharp (talk) 02:09, 4 August 2020 (UTC)




