Double sharp (talk | contribs) |
→Mass summoning: Unacceptable behaviour |
||
| Line 5,957: | Line 5,957: | ||
{{ping|ComplexRational|DePiep|Droog Andrey|Officer781|R8R|Sandbh|Дрейгорич|AzaToth|Tazerdadog|Maproom|CuriousMind01|Silvio1973|Timtempleton}} Pinging all megathread and previous RFC participants. And also [[User:AnthonyDu0122|AnthonyDu0122]] who raised it above. ^_^ |
{{ping|ComplexRational|DePiep|Droog Andrey|Officer781|R8R|Sandbh|Дрейгорич|AzaToth|Tazerdadog|Maproom|CuriousMind01|Silvio1973|Timtempleton}} Pinging all megathread and previous RFC participants. And also [[User:AnthonyDu0122|AnthonyDu0122]] who raised it above. ^_^ |
||
====Unacceptable behaviour==== |
|||
{{hidden ping|ComplexRational|DePiep|Droog Andrey|Officer781|R8R|Дрейгорич|AzaToth|Tazerdadog|Maproom|CuriousMind01|Silvio1973|Timtempleton|AnthonyDu0122|Michael D. Turnbull}} |
|||
I '''object''', in the strongest possible terms, to the way Double sharp initiated this RFC. |
|||
I allege that Double sharp's background to the RFC is biased; incomplete; out-of-context; contains inappropriate content; and—at 33,000 words—is too long for anyone to gain an informed understanding of the issue at hand. This follows other unacceptable behaviour alluded to on his part, including his hack work on our [[periodic table]] article; removing some of my citation supported content; slandering me; swearing; and effectively demanding I provide a falsifiable hypothesis when I was under no obligation to do so. |
|||
The context for my objection is my long-term participation in the Group 3 debate, including two submissions to IUPAC on the matter, one with Double sharp (which he has now dissociated himself from). |
|||
My other objection is that an article written by me will soon appear in the journal ''[[Foundations of Chemistry]],'' entitled "The location and composition of Group 3 of the periodic table". I have the proofs in hand. The article has been reviewed by three PhDs and accepted for publication by [[Eric Scerri]], a world authority of the periodic table. Scerri is also the chair of the IUPAC Group 3 project. |
|||
Double sharp knows all this, yet chooses to go ahead with the RFC. So much for taking into account "reliable sources that focus on the matter." So much for extending me the courtesy of professional respect. So much for working collegiately within [[WP:ELEMENTS]]. |
|||
Here again is a continuation of his pattern of unacceptable behaviour. |
|||
Most of the rest of the editors who have voiced their opinions for Lu, it seems to me, do so on the basis of a personal preference or a very limited grasp of the full extent of the arguments and literature involved in this case. |
|||
I'm not holier than anyone. I do claim, along with Double sharp, to have maintained a depth of familiarity and interest in this question since at least 2016. In my case I've been arguing the question with Scerri, on and off, since 2008. And along the way, Double sharp and I have changed our minds about Group 3 as we both learnt new things along the way. |
|||
As Jones (2010) has written: |
|||
:"Scientists should not lose sleep over the hard cases. As long as a classification system is beneficial to economy of description, to structuring knowledge and to our understanding, and hard cases constitute a small minority, then keep it. If the system becomes less than useful, then scrap it and replace it with a system based on different shared characteristics." |
|||
::<small>Jones 2010, ''Pluto: Sentinel of the outer solar system,'' Oxford University Press, p. 171</small> |
|||
As Schwerdtfeger, Smits & Pyykkö (2020) wrote: |
|||
:"Fuzzy concepts like chemical similarity often lead to unnecessary disputes concerning the PTE." |
|||
::<small>Schwerdtfeger P, Smits OR & Pyykkö P, 2020, "The periodic table and the physics that drives it". ''Nature Reviews Chemistry,'' vol. 4, pp. 359–380</small> |
|||
That is why we have an IUPAC Group 3 project team, to assess the literature and address these unnecessary disputes. |
|||
And the IUPAC project team [https://iupac.org/projects/project-details/?project_nr=2015-039-2-200 has reported that,] based on a survey of chemistry textbooks and other sources, that the La form dominates by a 4:1:1 margin, where the two 1's are the *-** form and the Lu form. |
|||
As Scerri wrote: |
|||
:"…the majority of textbook and other periodic tables in the 18-column format show the elements of this group as scandium, yttrium, lanthanum and actinium…" |
|||
<small> |
|||
::Scerri ER 2020, "Recent attempts to change the periodic table", ''Philosophical Transactions A,'' [https://www.researchgate.net/publication/339815154_Recent_attempts_to_change_the_periodic_table here] |
|||
</small> |
|||
As [[WP:Elements]] member [[User:R8R|R8R]] wrote: |
|||
:"…I'll say that the 4:1:1 consideration will be very important for the upcoming RfC, especially given that it's something that can be found on [https://iupac.org/projects/project-details/?project_nr=2015-039-2-200 IUPAC's official website]. I agree that consistency with literature is of the uttermost importance ''for Wikipedia'' if literature itself goes shows such a consistency; Wikipedia is [[WP:TERTIARY|meant]] to be a tertiary source. It'll be the thing that will hold me from supporting the motion ''on WP right now''…--[[User:R8R|R8R]] ([[User talk:R8R#top|talk]]) 16:51, 19 July 2020 (UTC)" |
|||
Precisely. |
|||
The way ahead, as I see it, is (1) cancel the RFC until my article appears on line; and (2) Double sharp and I to agree on a balanced introduction to a new RFC, should Double sharp wish to proceed with another RFC. Failing this I will take up the matter on [[WP:AN]]. --- [[User:Sandbh|Sandbh]] ([[User talk:Sandbh|talk]]) 03:27, 21 July 2020 (UTC) |
|||
===Discussion=== |
===Discussion=== |
||
Revision as of 03:27, 21 July 2020
 | Periodic table is a featured article; it (or a previous version of it) has been identified as one of the best articles produced by the Wikipedia community. Even so, if you can update or improve it, please do so. | |||||||||||||||||||||||||||
| This article appeared on Wikipedia's Main Page as Today's featured article on February 28, 2004, and on January 8, 2018. | ||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||
| This article is of interest to the following WikiProjects: | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Index
|
|||||||||||||||||
|
This page has archives. Sections older than 60 days may be automatically archived by Lowercase sigmabot III when more than 4 sections are present. |
Mendeleev Tabli first edition
According to the final chronology of the first publications of the Mendeleev’s Periodic Table (Druzhinin, 2020[1]), the first Medneleev ‘s Table published March 26-27 [O.S. March 14-15] 1869 in the Mendeleev’s The Principles of Chemistry 1st Edition, and around March 17 [O.S. March 29] 1869 Mendeleev printed separate broadsheets with Periodic table - Attempt - to be sent to overseas scientists; April 17/19 for the first time the Mendeleev’ Periodic table was published in Europe in Journal für Praktische Chemie (in German)[2]
References
- ^ Druzhinin, Petr (2020). "The First Publication of Mendeleev's Periodic System of Elements: A New Chronology". Historical Studies in the Natural Sciences. 50: 129–182.
- ^ Mendeleev, Dmitri (1869). "Versuche eines Systems der Elemente nach ihren Atomgewichten und chemischen Functionen" [System of Elements according to their Atomic Weights and Chemical Functions]. Journal für Praktische Chemie. 106: 251.
A relevant argument, unfortunately incomplete in sources
I wrote this paragraph, but then deleted it:
| “ | Because of the almost completely regular variation of lattice and solvation energies across the lanthanide series, the most important factor determining redox stability of lanthanide compounds is the third ionisation energy. This almost always involves the loss of one f electron, for which ligand field stabilisation energies are negligible as the f shells are deeply buried; that is distinct from the behaviour of the d transition elements. In almost all cases this ionisation is M2+ (4fn) → M3+ (4fn−1); although lanthanum (La2+ [Xe]5d1) and gadolinium (Gd2+ [Xe]4f75d1) are exceptions, the regular configurations [Xe]4f1 and [Xe]4f8 are very low-lying excited states (energies 0.8881 eV and 0.295 eV) respectively that negligibly affect the ionisation energy trend on being subtracted (the third ionisation energies of La and Gd respectively are 19.1773 eV and 20.63 eV). Similar processes are observed for example in the condensation of the lanthanides, which generally involves the transition 4fn6s2 → 4fn−15d16s2 and so varies in energy similarly to the above 4fn → 4fn−1 ionisations. In condensed phases the configurations of the Ln2+ may also change due to smaller destabilisation of the 5d orbitals compared to the 4f by ligand field effects. Energy variation of a process across the lanthanide series can be predicted from how the number of 4f electrons changes during its reactions. However, lutetium cannot be considered to be of this type as an f shell cannot hold the 15 electrons that would be required. Similarly, if we consider the processes where it is the relative stability of M3+ and M4+ that is at stake, then lanthanum cannot be considered as getting it to the +4 state would require breaking open the xenon core. (cite Johnson from the article) | ” |
The problem is that this argument, so far as it is taken in the source, does not clearly support either La-Yb or Ce-Lu as the 4f row. (Of course, there are lots of arguments about why La-Yb is the right answer and why the arguments for Ce-Lu are focusing on chemical irrelevancies in the literature. I know it probably doesn't look like the periodic table in your textbook, but (1) that textbook is probably not focusing on group 3 and (2) if you can cite Seaborg, as well as Landau and Lifshitz, on your side, like I have, it's definitely not a fringe view. XD) In order to make it complete, you would have to note that physical properties universally support the +2 to +3 process' double periodicity, but so far I haven't seen that being noted. Which is admittedly a bit puzzling, because it is absolutely obvious from the cited values of melting and boiling points: minima occur at Eu and Yb, just as they do at Mn and Zn. Double sharp (talk) 04:44, 18 May 2020 (UTC)
- Deleting it was a good move.
- Johnson: The relevance of his reference to the third ionisation energy involving the loss of one f electron in the free atom, is puzzling.
- The Ln metals, as opposed to the free atoms, are mostly f1-14d1s2. In their most stable trivalent cationic forms they are f1-14. Here, Ce3+ is f1, Pr3+ is f2, ending with Yb3+ f13 and Lu3+ f14. Thus, the f-block metals and their cations run from Ce-Lu. La is not include in the particular set, since La3+ is [Xe] i.e. it has no f electron.
- As Seaborg wrote, "it is the electronic structure of the ions and compounds that we are chiefly concerned with in chemistry".
- Seaborg again: An examination of Seaborg's article on the development of the actinide hypothesis shows he consistently distinguished between La-Ac, and Ce-Lu, Th-Lr. See figures 2, and 4-6 (pp. 52, 54-56).
- The right answer: There are no arguments about why La-Yb is the "right" answer. Rather, there are some arguments in the literature suggesting La-Yb looks better in some cases. Many of these are one-shots based on a single property. Jensen had a red-hot go at corralling these arguments in 1982, in support of La-Yb, but his effort failed to gain traction. He was criticized by Scerri, (chair of the IUPAC Group 3 project group) and Parsons (2018, p. 143), for being too selective in his arguments. There was Holden (1985) who looked at seven properties but estimated the argument of Landau & Ligshitz (1958) to be the strongest. For a discussion as to the unreliability of Landau and Lifshitz, see here.
- A survey by the IUPAC Group 3 project team found that the majority of textbook and other periodic tables in the 18-column format show the elements of group 3 as scandium, yttrium, lanthanum and actinium. The survey did not include a Google image search, since such results are plagued by bias arising from the false impression that the IUPAC *-** table is “official”, and unreliable sources.
- Mathias (1969) grumbled about La in group 3 being the most popular form. Myers, Oldham and Tocci (2004, p. 130) found La and Ac to be the most popular form of periodic table, a sentiment echoed by Clarke and White (2008); and Lavelle (2008; 2009).
- Clark RW & White GD 2008, "The flyleaf periodic table", Journal of Chemical Education, vol. 85, no. 4, p. 497
- Holden NE 1985, Status of the lanthanides and actinides in the periodic table, accessed 24 November 2016
- Jensen WB 1982, The positions of lanthanum (actinium) and lutetium (lawrencium) in the periodic table, Journal of Chemical Education vol. 59, pp. 634−636
- Lavelle L 2008, "Lanthanum (La) and actinium (Ac) should remain in the d-block", Journal of Chemical Education, vol. 85, no. 11, pp. 1482–1483, doi: 10.1021/ed085p1482
- —— 2009, "Response to misapplying the periodic law", Journal of Chemical Education, vol. 86, no. 10, p. 1187, doi: 10.1021/ed086p1187
- Mathias BT 1969, "Systematics of superconductivity", in PR Wallace (ed.), "Superconductivity: Proceedings of the Advanced Summer Study Institute on Superconductivity", McGill University, Montreal, vol. 1, Gordon and Breach, New York, pp. 225−294
- Myers RT, Oldham KB & Tocci S 2004, Holt chemistry, Holt, Rinehart and Winston, Orlando
- Scerri ER & Parsons W 2018, "What elements belong in Group 3 of the periodic table?". In E Scerri & G Restrepo (eds), Mendeleev to Oganesson: A multidisciplinary perspective on the periodic table, Oxford University Press, New York, pp. 140–151
- Chemical irrelevancies: There are no arguments in the literature, to my knowledge, saying Ce-Lu is focusing on "chemical irrelevancies".
- Double periodicity: As to Eu and Yb, and Mn and Zn, it is more relevant, from a chemistry based view, rather than a physical property-based view, to note double periodicity for the 4f and 3d metals pivots around Gd and Lu, and Mn and Zn.
- Shchukarev (1974, p. 118), a well-regarded Russian chemist, supported -La-Ac on the grounds that the 4f shell does not start filling until cerium and that (effectively) the filling sequence—which runs from cerium to lutetium—is periodic, with two periods. Thus, after the occurrence of a half-full 4f shell at europium and gadolinium, the filling sequence repeats with the occurrence of a full shell at ytterbium and lutetium (Rokhlin 2003, pp. 4–5). A similar, but weaker, periodicity (Wiberg 2001, p. 1643–1645) is seen in the actinoids, with a half full 5f shell at americium and curium, and a full shell at nobelium and lawrencium.
- Placing lutetium and lawrencium under yttrium obscures the start of the filling of the f-block (it would appear to start at lanthanum) and visually truncates its double periodicity (it would be cut off at ytterbium whereas it would actually end in the d-block). In Shchukarev’s words, the "correctness of placing of imitators before gadolinium and curium as well as lutetium and lawrencium, would be lost. The exceptional uniqueness of Gd and Cm, akin to that of Mn and Zn, would also be unclear."
- Here is what a lanthanum table looks like:
+4 +2 | +4 +2
Ce Pr Nd Pm Sm Eu Gd | Tb Dy Ho Er Tm Yb Lu
½f ½f | f f
|
+4 +2 | +4 +2
Th Pa U Np Pu Am Cm | Bk Cf Es Fm Md No Lr
½f ½f f f
- Shchukarev’s imitators are Eu2+ and Yb2+ which like to attain the Gd3+ and Lu3+ cores. Then there is Ce4+, which likes to attain the core of its lanthanoid progenitor namely La3+; and Tb4+ attaining the same configuration as Gd3+.
- Europium, the most reactive of the lanthanoids, is associated with the place of a halogen, and gadolinium is associated with place of a noble gas, although to a much lesser degree. See the Rare-earth metal long term air exposure test (Hamric 2007). Terbium to lutetium are much less reactive, and the Group 17/18 analogy is not seen here.
- The f-block contraction starts with Ce3+ and culminates in Lu3+.
- The lutetium option is less regular:
+4 +2 | +4 +2
La Ce Pr Nd Pm Sm Eu | Gd Tb Dy Ho Er Tm Yb
½f | ½f f
|
+4 +2 | +4 +2
Ac Th Pa U Np Pu Am | Cm Bk Cf Es Fm Md No
½f ½f f
- The ½-filled and filled f- sub-shell regularity is lost; the correspondence of europium and gadolinium to Group 17 and 18 metals is lost; the f-block contraction starts with Ce3+ and awkwardly finishes in the d-block.
- Shchukarev’s approach is consistent with the observation of a progressively delayed start of filling of the f-shell as one goes down the periodic table i.e. at cerium for 4f; at protactinium for 5f although some 5f involvement may be possible for thorium (Edelstein & Kot 1993, p. 86); and at E123−125(?) for 5g (Eugen Schwarz, pers. comm. 8 Dec 2019).
- This double periodicity had been noted by Klemm (1929; 1930) on the basis of atomic structure (Remy 1956, p. 492); and by Sobolev (2000, pp. 44–45).
- Edelstein NM and Kot WK 1993, "Spectroscopic and magnetic studies of tetravalent Pa and trivalent Th compounds", Journal of Alloys and Compounds, vol. 193. pp. 82–87
- Hamric D 2007, "Rare-earth metal long term air exposure test", Metallium, Inc., viewed 7 March 2020
- Horovitz O & Sârbu C 2005, "Characterisation and classification of lanthanides by multivariate-analysis methods", Journal of Chemical Education, vol. 82 no. 3, pp. 473–483, doi: 10.1021/ed082p473
- Klemm W 1929, "Measurements on divalent and quadrivalent rare earth compounds. II. A system of rare earths based on periodic property changes of their ions", Zeitschrift für anorganische und allgemeine Chemie, vol. 184, p. 345
- —— 1930, "Measurements on divalent and quadrivalent rare earth compounds. IV. Supplement to the communication: A system of rare earths", Zeitschrift für anorganische und allgemeine Chemie, vol. 184, p. 29
- Remy H 1956, Treatise on inorganic chemistry, vol 2, Elsevier, Amsterdam
- Rokhlin LL 2003, Magnesium alloys containing rare earth metals: Structure and properties, Taylor & Francis, London
- Sobolev BP 2000, The rare earth trifluorides: The high temperature chemistry of the rare earth trifluorides, vol. 1, Institut d'Estudis Catalans, Barcelona, pp. 44–45
- Ternström T 1976, "Subclassification of lanthanides and actinides", Journal of Chemical Education, vol. 53, no. 10, pp. 629–631
- Wiberg N 2001, Inorganic Chemistry, Academic Press, San Diego
- Physical properties: Concluding with these, lutetium resembles the 5d transition metals more than is the case for lanthanum. That said, lutetium resembles closely erbium and holmium, and while it melts at a slightly higher temperature and is essentially non-magnetic, the details of producing, purifying and fabricating it are almost identical to that for holmium (Spedding et al. 1968).
- Spedding FH & Beadry BJ 1968, "Lutetium", in CA Hampel (ed.), The encyclopedia of the chemical elements, Reinhold Book Corporation, New York, pp. 374–378
Recent attempts to change the periodic table (Scerri 2020)
Here's a link to the latest article by Eric Scerri, a world authority on the periodic table.
Here are some extracts from, and my commentary on, this interesting piece:
Philosophy v pragmatism?
The focus of the paper is philosophy rather than pragmatism. (p.2)
I suggest a need to focus on philosophy and pragmatism.
Focusing on just one results in needless arguments, including of the kind I used to make.
Insisting on one PT
Scerri writes:
- "There is no need to insist on the periodic table having a format that is suited mainly for the purposes of the chemical community and for chemical educators." (p. 6)
As far as I know, no one has made such an insistence. It is rather a case of people insisting on such a table within a particular sub-context or interest dependence, and usually not making this clear enough.
Who owns the PT?
Scerri writes:
- "The periodic table has now become as much the property of physicists, geologists, astronomers and others as it is of its chemical originators." (p. 7)
This is a contentious generalisation.
I suggest the periodic table, in the first instance, remains the organising icon of chemistry. Thus, here is what Scerri said in the 2nd (2020) edition of his Red Book:
- "…it helps to remember that, when all is said and done, the periodic table remains primarily in the domain of chemistry, although the relationship between chemistry and the underlying explanation from physics remains as the underlying theme… (p. ix)"
Rather than becoming a shared commodity, the periodic table concept has been borrowed, adapted, tailored and presented in various different guises—including the 15-element wide f-block version—by the physicists, geologists, astronomers and others.
As Scerri rightly says:
- "It becomes increasingly clear that there may not be any such thing as one optimal table in a purely objective sense. The question seems to depend on what criteria are considered and, most importantly perhaps, on whether one favours chemical or physical criteria or general didactic considerations." (p. 12)
To this commendable end, he goes on:
- "We should accept that a degree of convention must be used in selecting a periodic table that can be presented as perhaps the best possible table that combines objective factors as well as interest dependence." (p. 14)
Quite so, having regard to the priorities of each interest group.
The Madelung Rule
- "In any case, it is interesting to see that Pyykkö admits that it is surprising, in view of the relativistic effects, that the Madelung rule survives so well all the way up to atomic number 172." [!] (p. 8)
As far as the MR anomalies/symmetry breaking we observe in real life are concerned, the analogy is to an aeroplane experiencing turbulence. The flight path always returns to normal, after each turbulence episode.
The MR can also be regarded as the "spine" underlying the pattern of free atom electron configurations. The spine has bumps, dips, and knobbly bits on it, but still runs "true", so to speak.
Group 3
- "While the majority of textbook and other periodic tables in the 18-column format show the elements of this group as scandium, yttrium, lanthanum and actinium, a significant number of more recent tables feature the last two elements as lutetium and lawrencium instead." (p. 10)
Serving the largest audience
Scerri seems to go off-message in his conclusion, with references to:
- "…the format of the periodic table that serves the greatest number of periodic table users including students, instructors and practising chemists alike"; and
- "…how the periodic table is presented to the widest possible audience of users." (p. 15)
Surely the result of these notions will be a periodic table that attempts to cater to everyone but pleases no one?
Going off-message at this point is peculiar, since he earlier wrote:
- "It becomes increasingly clear that there may not be any such thing as one optimal table in a purely objective sense. The question seems to depend on what criteria are considered and, most importantly perhaps, on whether one favours chemical or physical criteria or general didactic considerations." (p. 12)
Accordingly, give me a tailor-made PT anytime, whether that is 14CeTh, 15LaAc, or 14LaAc, Adomah, AAE, Janet or some other variation, as long as the applicable context is set out.
The bugaboo of the split s-block
Scerri notes the periodic table is generally depicted with helium in group 18, and this splits the s-block (p. 11).
This is one of those things—the split s-block—that effectively all chemists (to a first approximation) do not lose any sleep over.
Same goes for the split d-block, which is less visible.
That said, better chemists keep both of these interesting aspects of the PT in mind.
It is like what Jones says:
- "Scientists should not lose sleep over the hard cases. As long as a classification system is beneficial to economy of description, to structuring knowledge and to our understanding, and hard cases constitute a small minority, then keep it. If the system becomes less than useful, then scrap it and replace it with a system based on different shared characteristics." Jones 2010, Pluto: Sentinel of the outer solar system, Oxford University Press, p. 171).
The anomalous first period
- "Another attractive feature of the left-step table is that it restores regularity and perhaps even balance to the otherwise awkwardly shaped traditional periodic table representation. More significantly than such aesthetic considerations, this table provides greater regularity in depicting every single-period length as repeating once as, 2,2,8,8,18,18,32,32. Meanwhile, the traditional table features an anomalous first period that, unlike all subsequent ones, does not repeat in length to give a sequence of 2,8,8,18,18,32,32. (p. 13)
I don't understand why the lack of repetition of the length of the first period is regarded as anomalous. There is no first principles derivation being breached here, as far as I know. It only means that, from a chemistry perspective, it is more meaningful to break the periods after the end of the noble gases. At the same time, the left step table is still good for its particular uses.
Thorium
- "Needless to say, the characterization of these blocks of the periodic table is only approximate, just as the assignment of electronic configurations to atoms represents an approximation. Moreover, one may readily concede that an element such as thorium does not actually possess any f-orbital electrons and yet it is classified as being among the f-block elements even in all four of the periodic table representations shown in figures 10 to 13.
--- Sandbh (talk) 00:44, 19 July 2020 (UTC)
RFC: Should the default form of the periodic table be changed to put Lu and Lr in group 3, rather than La and Ac?
Should the default appearance of the periodic table on Wikipedia be changed to what it was in 2016, i.e. to have lutetium (71Lu) and lawrencium (103Lr) in group 3 rather than lanthanum (57La) and actinium (89Ac)? Or should it simply have markers below yttrium where all the lanthanides and actinides are supposed to go in? Double sharp (talk) 10:08, 20 July 2020 (UTC)
- Note for further clarification: this does not propose to treat any form as the only one, because that would contradict WP:NPOV. On this article we must of course discuss both forms and treat them neutrally but briefly, that goes without saying. I simply mean to discuss the appearance of our templates like
{{Periodic table}},{{Compact periodic table}}, the one in the infoboxes like{{infobox helium}}, etc. These normally appear as navigation aids in articles where the group 3 dispute is simply irrelevant, therefore unless someone has a better solution we seem to need to pick a default form for those. Double sharp (talk) 12:44, 20 July 2020 (UTC)
The three forms
Lu and Lr below Y. The form I propose, and which we had before 2016.
18-column:
| H | He | |||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | |||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | |||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | |
| Cs | Ba | * | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | ** | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| * | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | ||||
| ** | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No |
32-column:
| H | He | ||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
La and Ac below Y. The current form.
18-column:
| H | He | |||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | |||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | |||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | |
| Cs | Ba | La | * | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac | ** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| * | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| ** | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
32-column:
| H | He | ||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
Markers below Y. The compromise form. IUPAC shows it on its periodic table, but they also plan to change it to one of the other forms. Which one has not been decided.
18-column:
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | * | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | ** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| * | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||
| ** | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
32-column (note the stretching of scandium and yttrium necessary):
| H | He | ||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
Background information by Double sharp
I spent some time making this issue understandable for any bystanders who may want to join in, helped by feedback from User:Dreigorich. As such, here is an explanation in the form of a dialogue. I have tried to keep answers very short. (The first version was a huge wall of text. Now it is not.)
The issue had its most recent reignition on Wikipedia in December 2019 and raged on through Wikipedia talk:WikiProject Elements/Archive 42, Wikipedia talk:WikiProject Elements/Archive 44, Wikipedia talk:WikiProject Elements/Archive 46, and until a few days ago Wikipedia talk:WikiProject Elements. I say "until a few days ago" because that's when I gave up the endeavour as fruitless: nobody seems about to have a change in opinion, and the arguments are raging in circles, and civility has been thrown out the window (and I realise now that I'm partly responsible for that, which I'm sorry for).
At that point we had exactly, among eight participants:
- one (1) editor in favour of La (Sandbh);
- five (5) in favour of Lu (myself, Droog Andrey, Officer781, Dreigorich, ComplexRational);
- and two (2) who at least found the arguments being made in favour of La questionably convincing (DePiep, R8R).
Since the issue was previously decided in an RFC back at Template talk:Periodic table, discussion between me and R8R concluded that going to another RFC would be the best way to propose it be changed back to how it was before the first RFC. Given that that RFC was over three years ago, and one of the main La proponents in it was me, and I've learnt enough new information since then that makes me think that the arguments I was propounding were wrong-headed, I think starting a new RFC is also most reasonable.
In theory one should get a neutral editor to write the background information. However, the problem is that this issue has something in common with many of the disputes at Wikipedia:Lamest edit wars. It is impossible to find a neutral editor to write background information because as soon as she looks at the issue enough to understand the background information, she takes a side herself and is no longer neutral. That is quite unusual given that Wikipedia:Lamest edit wars does not have a category for natural science, much less chemistry. Indeed, the main positive difference between the issue here and the ones over there is that so far there has not been any actual edit warring. Some uncivil remarks when people on either side get fed up, yes, and I am sorry that I have made some of them. But, thankfully, no edit warring.
So I wrote it. Even if I obviously have an opinion and am obviously supporting it with arguments here, I'm at least referring to reliable sources focusing on the issue to do it. If that is still not suitable, then this may at least be considered as a long explanation of my !vote rather than background information.
Questions 1 to 5
Q1: What are you arguing about?
Well, it turns out that somewhere near the bottom-left of the periodic table there's a bit of a dispute about which elements should go where.
Q2: Really? But I thought the periodic table was ancient history chemically speaking.
Yes, Mendeleev's first table dates from 1869. We still haven't sorted everything out, though. There are a bunch of elements that are rather difficult to place.
Q3: That sounds interesting. Tell me more.
In the words of Eric Scerri, "there's trouble at the beginning, middle and end of the periodic table". This RFC is about the middle: the rare earth elements.
Q4: So are you going to tell us what it is already?
It comes down to whether lutetium (Lu) or lanthanum (La) is a better fit for the position under yttrium (Y) in the periodic table. There's also a third form which has all fifteen lanthanides under yttrium, which we did have on WP a while ago, but not anymore.
Q5: Why is this so terribly important? If an element fits well in two places, why not duplicate it in both positions?
This is not generally done by reliable sources, except maybe when making a point. Therefore on Wikipedia we should not start doing it.
Questions 6 to 10
Q6: What about the "markers below yttrium" form? Isn't it a reasonable compromise?
It looks like one, but it is also a side in itself.
I do not support it for two reasons. One is technical. We sometimes use 32 column templates because they fit better as footers, e.g. {{compact periodic table}}, and then this approach becomes really problematic: scandium and yttrium have to stretch to cover 15 columns.
The other is chemical. This form claims that the f-block is a degenerate branch of the d-block and thus by extension that the f orbitals are a part of the valence core, which is simply false for almost all the elements involved (all but Lu and Lr, in fact). Every redox-capable f element (which is almost all of them) is obviously using its f electrons, and even those which are not seem to have some f contribution anyway (see Q22). See Jensen for a discussion. Maybe all the lanthanides are similar enough to fit below yttrium, but can anyone really claim that for the actinides?
Q7: Well, shouldn't we follow NPOV and show all three options all the time then?
That makes sense, and is what I support, when the group 3 controversy is the topic at hand. But usually it is not. Then you have to pick one to avoid going off on a tangent which is not relevant. Below I argue for Lu under Y.
For reasons of brevity, I will hence forth shorten the options' names to:
- Lu table: Lu and Lr below Y.
- La table: La and Ac below Y.
- * table: Markers * and ** below Y.
Q8: The things are called lanthanides and actinides. Shouldn't lanthanum and actinium continue to be used as the placeholders?
They're not placeholders. The * table and La table are different. Moreover, the whole point of the categories "lanthanide" and "actinide" is that those elements are similar to La and Ac. It seems quite weird to put La and Ac away from the rest of them. It is less weird for Lu (whose properties among the lanthanides are furthest from those of La) and not weird at all for Lr (which is quite unlike the other late actinides Es-No in properties).
Q9: IUPAC is the relevant source here, surely? What do they have to say?
They show their table as a * table, but they also state:
| “ | While IUPAC has no recommendation for a specific form of the periodic table, i.e. 18-column or 32-column format, the version here presented is in the conventional long form and is yours to use. | ” |
| — IUPAC Periodic Table website | ||
So it is not clear if that is an actual IUPAC recommendation for the * form or not.
IUPAC also organised a project to resolve precisely this issue, as can be seen from number 7 on their periodic table page. That project considers the La and Lu alternatives, but not the * one that they currently show. It started in 2015 and still has not concluded anything. Since it has both strong La and Lu proponents on it I also doubt it will conclude anything soon.
All we can conclude for now is that they seem to intend to deprecate the * form they currently show, thus leaving us without a firm guide in the meantime.
Q10: Why aren't we waiting for them then?
Because (1) I doubt that their work will be finished anytime soon, (2) we don't always follow IUPAC (we follow it for spelling aluminium but not for phosphane), and (3) any way you look at it, we have to show something in the meantime, see Q7.
I argue that we should look at the consensus of reliable sources that focus on the matter. Because an article focusing on group 3 and explaining why the authors think La or Lu should be there, I argue, outweighs in terms of reliability somebody who just draws a periodic table and gives no clue as to why he or she has drawn it that way. I claim that's in favour of Lu.
Questions 11 to 15
Q11: What do most reliable sources say?
Depends really. If you do a Google Image survey on "periodic table", * below Y dominates. It has been claimed during discussion that La below Y dominates in textbooks, but usually those textbooks have * in the same cell as La with a group 3 header above, therefore also implicitly claiming that the other lanthanides go there. So there is, at least, an ambiguity. Also, all these dominances are just 2/3 majorities, it's not a rare thing at all to find a textbook or periodic table poster giving Lu below Y.
I claim it's more relevant to look at what articles specifically focusing on this issue tell us, because it's common for textbooks to repeat standard errors on issues like this (e.g. claims that d orbitals are involved for phosphorus in PCl5, which are false but ubiquitous in textbooks), and most of these textbooks are hardly focusing on these heavy 4f and 5d elements that the dispute is all about. Those have a majority supporting the Lu form. I tallied them with Sandbh in an old submission to IUPAC we made on this matter in 2016; I've updated the list to 2020 below. Back then we agreed that La is better; then I changed my mind in response to new evidence from Droog Andrey that Lu is better, so we started arguing.
| Advocates Lu | Advocates La | Advocates * |
|---|---|---|
| Bury (1921) Shemyakin (1932) Landau and Lifshitz (1958) Hamilton (1965) Merz and Ulmer (1967) Chistyakov (1968) Mathias (1971) Wittig (1973) Jensen (1982 and again 2015) Holden (1985) Fang et al. (2000) Horovitz and Sârbu (2005) Wulfsberg (2006) Ouyang et al. (2008) Scerri (2012) Nelson (2013) Settouti and Aouragi (2014) Alvarez (2020) |
Smith (1927) Trifonov (1970) Shchukarev (1974) Atkins (2006) Lavelle (2008) Restrepo (2017) Cao et al. (2020) |
Xu and Pyykkö (2016) |
Q12: Aren't you essentially claiming that a whole lot of eminent scientists are wrong?
It's a controversy. No matter which side you pick you're going to be claiming that a whole lot of eminent scientists are wrong. La advocates like Sandbh have Sergey Shchukarev on their side, Lu advocates like me have Landau and Lifshitz, so we can see that both options have had respected adherents. That's why I claim we have to look at the articles focusing on this issue and also consider the chemistry involved.
Q13: Many chemists have never heard that there was such a debate. Are you sure you're not just another fringe theory peddler?
Landau and Lifshitz are hardly "fringe". Neither is Clayden et al.'s Organic Chemistry, which has a Lu table. And Jensen, before his 1982 article, had not heard of this debate either.
Q14: What is the status of that interminable thread at Wikipedia talk:WikiProject Elements, stretching from last December to this July?
We have 5-1 for Lu and an extra two who at least think the La arguments proffered are problematic.
- Well, I guess it goes without saying that I support the Lu form, yes?
- "La arguments are totally local, while Lu arguments are pretty regular. That exactly matches Ptolemy vs. Copernicus. The history just repeats itself. Nothing more to say." - Droog Andrey
- "Cool, thanks. This makes perfect sense. Well said. Team Lu for me!" - Dreigorich
- "I'd really just want to get rid of the Scandium/Yttrium overhang in the long periodic table. It is ugly and very artificial IMO." - Officer781
- "I am more convinced by the Lu arguments I've read, and given the support of the scientific community and lack of consensus against it here (no "battleship" as you describe), I'll join the RfC once it's underway." - ComplexRational
- "Even worse, a publication may be biased towards a preference for more simple or elegant or 'by authority' (IUPAC) form, which is a beauty contest not science. I understand Sandbh does argue for such criteria seriously, but that still does not convince me—FWIW." - DePiep
- "I have noted in the very beginning of this discussion, which started with an article you [Sandbh] wrote, that it did not appear to me that pro-La-Ac and pro-Lu-Lr arguments were given the same weight, and I said, perhaps not as explicitly but to the same meaning, that it looked like this was done so deliberately so that one option is favored over the other. I am afraid that what I've read so far reinforces this thinking within me." - R8R
versus a single one:
- "Ahead of the recommendation of the IUPAC project I see no case for change." - Sandbh
However since we carried out the last change in an RFC, I feel (after discussion with R8R) that a proposed reversal should also go through an RFC. It seems only fair.
Q15: Have you addressed Sandbh's arguments there?
Yes, I responded to everything new he raised. Though not always in the most polite way after a few months of arguing, for which I am sorry.
Questions 16 to 20
Q16: Exactly why were you arguing this fiercely with Sandbh?
Well, he was writing an article, to be published in Foundations of Chemistry, that supports La under Y, and brought it to WT:ELEM for peer review last December. I and Droog Andrey disagreed with his arguments on what seemed to me to be logical grounds. Eventually it got to this:
| “ | When you get as old as me, you'll learn there is much more to the world than logic. ...
You keep asking for a falsifiable theory. There isn't one! |
” |
| — Sandbh, 07:49, 12 May 2020 (UTC) | ||
I found this quite surprising and responded in a rather too harsh manner, which I'm sorry for. Nevertheless I still cannot agree with this approach. Unless we use logic as a base, I don't see how we can judge the strength of arguments.
Q17: So why did it last so long?
Arguing with him was very helpful to sharpen my own understanding. But let's be honest: "someone is wrong on the Internet" also had something to do with it on my part, and I should really wean myself off of that.
Q18: But you've not been a complete angel yourself dealing with him.
That is true. I'm sorry. I will undertake to be more civil in future.
Q19: What's the history of this neverending argument anyway?
Mendeleev more or less gave up on placing the lanthanides, mostly leaving it to his Czech colleague Bohuslav Brauner. After some other attempts, eventually a form with all lanthanides under yttrium (basically the * form) was settled on. However even during this time tables that quite clearly don't put La under Y had already appeared (Henry Bassett 1892, Alfred Werner 1905). Actually Werner's form (yes, that's the father of coordination chemistry we're talking about) is incredibly close to the modern 32-column Lu form (with the exception that Be and Mg go over Zn instead of Ca). The only reason I do not call it a Lu table is because Lu had not yet been discovered(!).
In the 1920s and 1930s some chemists assigned Lu under Y because the chemistry of Y is more similar to Lu than La (Sc, Y, and Lu separate in the yttrium group of rare earths, but La and Ac in the cerium group).
The current dominance of the La form seems to stem, according to Jensen, from wrong electron configurations from the 1940s in which the lanthanides were assumed to be fn-1ds2 instead of fns2. These were later corrected, but by that time it seems the La form had stuck. We should not forget that these electron configurations are for the gas-phase ground state and are not really relevant for chemistry anyway (in chemical environments they change).
Meanwhile articles supporting Lu have appeared since 1921 and have been going on until the present; there have not been as many articles supporting La. Some authors have changed their periodic tables, others have not, there is still no consensus. IUPAC started a project in 2015 to look into it, they still have not made a decision.
It is possible that things will change. Be-Mg-Zn was present in Werner's periodic table of 1905, B-Al-Sc was once shown by Pauling(!), and prior to Seaborg it was generally accepted that the actinides known (Ac, Th, Pa, U) form a fourth d block row with uranium being eka-tungsten. Now you don't see any of these.
Q20: Should tradition count for something here?
There's not much of a tradition when chemists still argue about it. I prefer that we take the option that most of them who really analyse the issue support when we're not discussing the issue.
Questions 21 to 25
Q21: Would it not simply work if we just added footnotes like for helium where we explain that the other option is also common?
For the infobox, for the footer, you do not want to emphasise the issue where it is irrelevant, e.g. for the article on helium where group 3 matters not at all. The "overhang" the La table has in the 32 column form draws the eye's attention. The "stretchy scandium and yttrium" of the * table draws even more of the eye's attention. In order to make them look good, you have to use an 18 column instead of a 32 column form, which strikes me as not terribly good considering that those are supposed to be the same thing (just with the footnote glued in or not). So for these purposes I claim the Lu table is better: it preserves the symmetry. Moreover there is even scientific basis for preserving that symmetry, as the periodic table is explained by the valence orbitals and thus quantum mechanics, the Madelung rule having derived in the 1950s by Klechkovsky already.
Of course, whenever it is relevant (i.e. article of lanthanum, article of lutetium, and the period 7 congeners), we will add a note like there is for helium (about helium over beryllium being supported by some chemists). That goes without saying, for WP:NPOV reasons. But I think the reader would be better served with a Lu under Y default for when the situation does not matter.
Q22: OK, so are you going to get on to the scientific basis of this change or not?
If you want a really long exposé of periodicity and why it supports Lu under Y, then please open the collapse box.
However, I presume most readers have no wish to read that. So here is just a short list of eight arguments.
- Per Jensen (1982) Lu under Y matches the trends in the d block better.
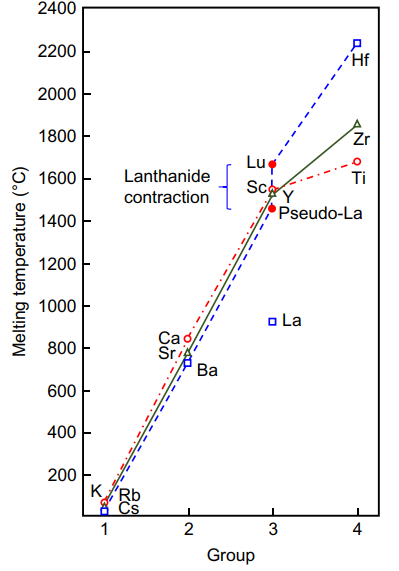
- Per Gschneidner (2016) La has 4f involvement in the metal that impacts its melting point, making it 450 °C lower than expected. The figure to the right from his paper shows "pseudo-La" for the expected melting point of La if it didn't have this 4f hybridisation. Such 4f involvement also ties together lots of properties of lanthanum: it explains melting point, heat of sublimation, high coordination numbers around 12 (difficult to explain without f orbitals), cubic molecular geometries of lanthanum complexes (difficult to explain without f orbitals giving the symmetry), superconductivity (it is superconductive, but Sc, Y, Lu are not), quite possibly also its crystal structure (which matches the early f elements but is different from Sc, Y, and Lu). Putting La under Y misses the point that the first element for which the 4f orbitals are non-hydrogenic and can participate chemically is lanthanum, not cerium.
- Lutetium has no involvement of the f electrons in chemistry. All they do is contribute incomplete shielding effects which is exactly like the effect of the lanthanide contraction on hafnium through mercury. On those grounds it is surely a d element.
- Properties of yttrium are closer to those of lutetium than lanthanum.
- The only precedent for taking an element out of its block is helium, which is obviously much closer in properties to neon than beryllium. Why do that for lanthanum when yttrium is already closer to lutetium in all ways?
- Taking La out of the f block on the grounds of its ground-state gas-phase electron configuration lacking a 4f electron is irrelevant (these configurations change in chemical environments) and inconsistent with thorium. Thorium is in the f block, yet its ground-state gas-phase configuration lacks a 5f electron. Besides the periodic table is not even based on ground-state gas-phase electron configurations in the first place, as evidenced by the fact that no one rips nickel [Ar]3d84s2, palladium [Kr]4d105s0, and platinum [Xe]4f145d96s1 apart from each other. Rather it is based on the number of valence electrons and which orbitals they may enter across all chemical environments.
- Examining properties reveals that La fits well with f elements, but is an outlier in the d elements. Same is true for Ac. Lu is OK either way (mostly), but Lr seems to fit well in the d elements but is an outlier in the f elements (we mostly only have predictions for that one of course).
- When examining properties, Eu and Yb show the properties expected and known from Mn and Zn (half-filled and filled subshell); Gd and Lu do not. That supports La-Yb as the f block. In fact a lot of analogies with the d block strongly support La-Yb as the f block for exactly this reason (standard electrode potentials, electronegativity, ionisation energies, melting and boiling points).
Everything said of the lanthanides above is also true or suspected of the corresponding actinides.
Evidence


| property | La | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg |
|---|---|---|---|---|---|---|---|---|---|---|---|
| m.p. (K) | 1193 | 1925 | 2506 | 3290 | 3695 | 3459 | 3306 | 2719 | 2041.4 | 1337.33 | 234.43 |
| b.p. (K) | 3737 | 3675 | 4876 | 5731 | 5828 | 5869 | 5285 | 4701 | 4098 | 3129 | 629.88 |
| specific heat capacity (J/(g*K)) | .195 | .154 | .144 | .14 | .132 | .137 | .13 | .131 | .133 | .129 | .14 |
| EN (Pauling) | 1.1 | 1.27 | 1.3 | 1.5 | 2.36 | 1.9 | 2.2 | 2.2 | 2.28 | 2.54 | 2.0 |
| EN (Kulsha-Kolevich) | 1.11 | 1.31 | 1.38 | 1.46 | 1.54 | 1.55 | 1.67 | 1.75 | 1.84 | 1.93 | 1.81 |
| Density | 6.145 | 9.84 | 13.31 | 16.654 | 19.25 | 21.02 | 22.61 | 22.56 | 21.46 | 19.282 | 13.5336 |
| Young's modulus | 36.6 | 68.6 | 78 | 186 | 411 | 463 | ??? | 528 | 168 | 78 | ??? |
| Bulk modulus | 27.9 | 47.6 | 110 | 200 | 310 | 370 | 462 | 320 | 230 | 180 | 25 |
| Resistivity (nΩm, close to r.t.) | 615 | 582 | 331 | 131 | 52.8 | 193 | 81 | 47.1 | 105 | 22.14 | 960 |
| Brinell hardness (MPa) | 350-400 | 893-1300 | 1450-2100 | 441-3430 | 2000-4000 | 1320-2500 | 3920-4000 | 1670 | 310-500 | 188-245 | ??? |
| Heat of fusion (kJ/mol) | 6.20 | 22 | 27.2 | 36.57 | 52.31 | 60.43 | 57.85 | 41.12 | 22.17 | 12.55 | 2.29 |
| property | Ac | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn |
|---|---|---|---|---|---|---|---|---|---|---|---|
| m.p. (K) | 1323 | 1900 | 2400 | ??? | ??? | ??? | ??? | ??? | ??? | ??? | 283 |
| EN (Kulsha-Kolevich) | 0.97 | 1.29 | 1.34 | 1.41 | 1.49 | 1.59 | 1.72 | 1.83 | 1.92 | 1.99 | 1.91 |
| Density (g/cm3) | 10.07 | 15.6 | 23.2 | 29.3 | 35.0 | 37.1 | 40.7 | 37.4 | 34.8 | 28.7 | 14.0 |
| 1st IE (kJ/mol) | 499 | 470 | 580 | 665 | 757 | 740 | 730 | 800 | 960 | 1020 | 1155 |
| 3rd IE (kJ/mol) | 1900 | 2228 | 2300 | 2378 | 2484 | 2570 | 2830 | 2900 | 3030 | 3080 | 3160 |
| property | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1st IE (kJ/mol) | 499 | 587 | 568 | 598 | 605 | 585 | 578 | 581 | 601 | 608 | 619 | 627 | 635 | 642 | 470 |
| 3rd IE (kJ/mol) | 1900 | 1978 | 1814 | 1900 | 1997 | 2084 | 2132 | 2026 | 2152 | 2267 | 2334 | 2363 | 2470 | 2643 | 2228 |
| m.p. (K) | 1323 | 2115 | 1841 | 1405 | 917 | 913 | 1449 | 1613 | 1259 | 1173 | 1133 | 1125 | 1100 | 1100 | 1900 |
| EN (Kulsha-Kolevich) | 0.97 | 1.01 | 1.04 | 1.06 | 1.08 | 1.12 | 1.07 | 1.18 | 1.22 | 1.27 | 1.32 | 1.36 | 1.39 | 1.37 | 1.29 |
| Density (g/cm3) | 10.07 | 11.72 | 15.37 | 18.95 | 20.45 | 19.84 | 13.69 | 13.51 | 14.79 | 15.1 | 8.84 | 9.7 | 10.3 | 9.9 | 15.6 |
| M | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| mp (°C) | 842 | 1541 | 1668 | 1910 | 1907 | 1246 | 1538 | 1495 | 1455 | 1085 | 420 | 30 | 938 |
| bp (°C) | 1484 | 2836 | 3287 | 3407 | 2482 | 2061 | 2861 | 2927 | 2730 | 2562 | 907 | 2400 | 2833 |
| M | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| mp (°C) | 727 | 920 | 795 | 935 | 1024 | 1042 | 1072 | 826 | 1312 | 1356 | 1407 | 1461 | 1529 | 1545 | 824 | 1652 | 2233 |
| bp (°C) | 1633 | 3464 | 3443 | 3130 | 3074 | 3000 | 1900 | 1529 | 3000 | 3123 | 2567 | 2600 | 2868 | 1950 | 1430 | 3402 | 4603 |
For these reasons I claim that the La form has no scientifically sound basis. It is not a good chemist's periodic table because yttrium is closer to lutetium than lanthanum, and because lutetium and lawrencium are far more like transition metals than lanthanum and actinium. It is not a good physicist's periodic table, because it goes away from the blocks that underlie the periodic table. And clearly teaching the Madelung rule is going to be pedagogically better than teaching the Madelung rule and then giving an exception for La that doesn't even match reality, given its 4f involvement.
This matches the criteria for putting elements in the periodic table as stated by Jensen, particularly arguments 2 and 3 (4f in La and its complete lack in Lu).
| “ | In actual fact, when assigning an element to a position in the periodic table the above factors are not simply applied as a haphazard empirical blend or combination, rather they are applied in a strictly hierarchical order, consisting of four steps or stages (Jensen 2009):
|
” |
| — William B. Jensen (2015), The positions of lanthanum (actinium) and lutetium (lawrencium) in the periodic table: an update | ||
Very long answer for Q22
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
As you wish! I'm going to start from the beginning, so as to not lose anybody. If you have no time for all of this ground-up explanation of why first principles of basic high-school chemistry already absolutely demand the Lu under Y table, then skip to Part K. But please do at least look at the pictures on your way down. ^_^
Atoms participate in chemical reactions by using their electrons. A proper understanding of electronic structure is therefore extremely important for understanding basic facts about chemistry. The important factor is the atomic number, the number of protons an atom has. This is typically symbolised Z, because German for number is Zahl. As protons are positively charged, and electrons negatively charged, a neutral atom must therefore also have that number of electrons. The electrons are arranged in shells around the nucleus. These may be thought of in a sense as orbits, although they're not really that. The vagaries of quantum mechanics means that, unlike what happens for a planet going around the sun, only certain energies are allowed for the electrons. Therefore, only certain orbits are allowed. The innermost one is called the first shell; then there is a second shell, a third shell, and so on. We can excite electrons by giving them energy, and in so doing promote them to shells as high as we want, or even have them break entirely free of the nucleus' hold. But, in the chemistry of the elements we know, only the first seven shells are involved in the most stable arrangements, known as the ground states. That is why our periodic table has seven rows. (The fact that it looks like there are more comes from the footnote; the sixth and seventh rows are very long indeed, so typically some parts of them are cut out and placed below the table proper to make a better aspect ratio for printing. But the structure's the same, and for clarity we will consider the table with the footnote placed in.) There are particularly stable electronic structures that occur at a set of elements called the noble gases. There are seven of them: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn), and oganesson (Og). The two-letter things in brackets are the symbols of the elements. Because these arrangements of electrons are so stable, these elements typically refuse to engage in chemical reactions, because they are already quite happy how they are. The other elements are not so lucky. They instead try to gain or lose electrons so as to reach this noble gas structure. Once a noble gas configuration is reached, there is no desire to go beyond it. No one has yet succeeded in getting any element to give up even more electrons past a noble gas structure; and no one has succeeded either in getting one to grab even more electrons past one. That is why, in the periodic table, we end each row with a noble gas. They form a natural bookend. This means starting a row with those viciously reactive alkali metals, one electron above the configurations of a noble gas.
Some elements are strong in their desires. The ones near the far left of the periodic table are furiously strong metals which throw away the few electrons they have over a noble gas structure without care. You can see this by observing the explosive results of throwing sodium (Na), one of these metals, into water. And the ones near the far right, such as fluorine (F) and chlorine (Cl), act like a bunch of chemical gangsters ready to forcefully grab electrons away from other atoms. Needless to say, such elements take to each other in a particularly violent form of love at first sight. Sodium would love to give up an electron, chlorine would love to get one, they react with each other and form sodium chloride (NaCl), or common table salt. This forms a huge salt crystal with trillions of trillions of sodium and chlorine atoms, the sodiums all short one electron, and the chlorines are with one extra. The chemical formula NaCl just means that they are in 1:1 ratio. In other ionic compounds, they might not be: calcium (Ca) has two electrons over a noble gas structure, so it forms CaCl2 instead, because every calcium atom can give up two electrons and make two chlorines happy, making a 1:2 ratio. And similarly oxygen (O) is two electrons short of a noble gas structure, so with sodium it forms Na2O, as every oxygen atom can take two electrons, one from one grateful sodium atom, making a 2:1 ratio. This is called ionic bonding, when one atom gives away its electron, and another one gratefully accepts the gift. Others are not quite so strong. Carbon (C), for example, is four electrons above the stable structure of helium. But it is also four electrons short of the stable structure of neon. Totally giving away four electrons, or accepting four, is beyond it: the process would require too much energy, more than can ever be repaid for chemically! But it can engage in an electron-sharing arrangement. A carbon atom can grab hold of four atoms of fluorine, for instance. Fluorine is one of those strong elements, one electron short of the stable neon structure. It turns out that fluorine has seven electrons in its outermost shell, but it would want eight to be happy. Carbon has four, and it would also want eight. And carbon can say, "look, I'm not able to outright give my electrons up to you; however, I can share them with you. Each of you can share one of your electrons with me, and I'll share one of my electrons with each of you, and those shared electrons are going to count for both of us. So you'll all be happy, because you'll have the eight electrons in the outer shell that you want, because each of you share one of mine. And I'll be happy, because my four will be added to the four contributed by all of you, and I'll have eight too. How does that sound?" And fluorine happily agrees! This sort of sharing is called covalent bonding. This is an example of a molecule, carbon tetrafluoride. We write that as CF4, because it has one carbon atom and four fluorine atoms. That's a small arrangement of atoms bonded covalently to each other. You can also have a giant structure of lots of atoms bonded covalently to each other. Diamond is one such: it's a whole bunch of carbon atoms bonding to each other. Some elements can form such structures in many different ways: graphite is another way carbon atoms can bond to each other. These are called different allotropes of the same element. Phosphorus has lots of allotropes: a red one, a black one, a white one! In the above we have also used something called the octet rule. This is because the second shell is full at eight electrons. Therefore, in order to predict how many covalent bonds an element in the second row will form (since those are the elements filling the second shell), we try to make its electron count get up to eight. The first shell is full at two electrons, so instead for those we use a duet rule. For quantum-mechanical reasons, it turns out that the nth shell has room for 2n2 electrons. So the first shell can hold 2 electrons; the second, 8; the third, 18. The pattern continues. And indeed, there's also a slightly less famous 18-electron rule. (Less famous, that is, because most high-school curricula only get to the elements for which this is needed at the very end.) Now, it should be noted that although the electrons are supposedly shared, the sharing is not quite so equitable as you might guess. Fluorine is the stronger element, it grabs hold of electrons more tightly, and it calls the shots. In CF4, the electrons in the bonds are closer to the fluorines than the carbon; the carbon is left with a slight positive charge, the fluorines with a slight negative one, and this difference is the basis for all sorts of interesting chemistry. Electronegativity, symbolised by the Greek letter chi (χ), measures how tightly an atom holds on to electrons that it has gotten in a chemical bond. Unsurprisingly, fluorine is one of the most electronegative elements. Caesium, one of those furiously strong metals at the other end of the periodic table, is one of the least: it couldn't care less about those electrons. Electronegativity is measured on a scale, in which caesium is 0.70, and fluorine is 4.00. Most elements are intermediate between them. As the difference between the electronegativities of two bonding elements grows higher, the bond transitions gradually from covalent to ionic. Consider one of the carbon-carbon bonds in graphite. Both carbons have electronegativity 2.50; neither wants the electron more than the other, so that's more or less a pure covalent bond. In CF4, the carbon has electronegativity 2.50, but the fluorine a significantly greater one of 4.00. This is still covalent, the electron is still shared, but it's definitely skewed more to the fluorine. This is called a polar covalent bond. In LiF, the lithium has electronegativity 1.00, the fluorine 4.00. That's a huge difference, and for all intents and purposes the lithium has essentially totally lost control of its electron to the fluorine: that may be called an ionic bond. Actually, there are no totally ionic bonds in reality. Both atoms always retain a partial hold, and there's only a continuum of more and more polar covalent bonds. But beyond some point, one atom has gotten so much of a lion's share of the custody that it's useful to approximate the bonding as ionic – bearing in mind that this is the first time you're seeing the fundamental principle here, that everything is a continuum. If anyone is telling you that there is a sharp boundary anywhere general on the periodic table, except for the one that comes after each noble gas, she is almost certainly wrong. There are even some special types of covalent bond. There are double and triple bonds, where instead of two electrons being shared among two bonding elements, there are four or six. There are dative bonds or coordinate covalent bonds: in these, one atom contributes both electrons of the bonding pair. And there are electron-deficient bonds like the 3c–4e (3-centre 4-electron) bond, as opposed to the normal 2c–2e (2-centre 2-electron, which is what a normal covalent bond looks like), that help to explain molecules that appear to violate the octet rule like PCl5, phosphorus pentachloride; at first glance the phosphorus seems to have gotten itself ten valence electrons, but actually it has only a stable octet; the average bond has less than two electrons in it! Yes, average, among multiple resonance structures! The molecule isn't in either structure or the other, but a superposition of all of them! One of the marvellous things about quantum mechanics.) I could spend a lot of time talking about this, but I'm just here to explain how the periodic table is built up, so we'll have to leave this fascinating topic. Now I should tell you here that there are two common ways to look at this continuum. One is the electronegativity way, in which you start with a pure covalent bond, and then polarise it because the more electronegative atom wants those electrons more! But the other is the polarisation way. In this you start with the mathematical fiction of a pure ionic bond. Here the electrons, you imagine, have already all jumped ship, even for something like CF4. Well, we have a C4+ cation (a positive ion, having lost four electrons) and four F− anions (a negative ion, each having gained one). Note: an ion is an electrically charged atom that has gained extra electrons or lost some. If it's gained some, it's negatively charged and is called an anion; if it's lost some, it's positively caharged and is called a cation. But just look at that C4+ cation. It's tiny and it has lots of positive charge. Something like that will hugely affect the electron clouds of those fluoride anions! In fact, it'll partly wrest some of the electron density back for itself. And that's how we get back to a more shared arrangement. In the limit you get a pure covalent bond when both atoms are doing the same amount of wresting of electrons from each other. Both ideas are logically sound: we'll use both of them, as sometimes one is better at helping understanding than the other. What you saw above was an application of Fajans' rules, named after the Polish chemist Kazimierz Fajans, who also was a codiscoverer of the element protactinium (Pa, Z = 91). They say that an ion increases in polarising power as its radius shrinks and as its charge grows. That makes perfect sense: that means its charge density goes up! Some of you will have realised that this means that electronegativity is itself a changing thing! Indeed, when an atom has lost more electrons, its electronegativity goes up. Lead (Pb, Z = 82; symbol from Latin plumbum) has an electronegativity that goes almost 0.5 units on this scale when it goes from having lost two electrons to having lost four! But in general, for most elements this effect is small. It only really becomes worrying for some of the heaviest elements, and lead is pretty hefty. But it's good to keep this in mind, in order to master the idea that everything here is a continuum. Surprisingly, fluorine is not actually the most electronegative element. Neon, the noble gas next to it, is, with a whopping great electronegativity of 4.50. This may surprise you because neon is one of those noble gases that is well-known for not bonding: why would it want an extra electron. But that's a key subtlety in the definition of electronegativity. It's not about how much the atom wants an extra electron; it's about how much the atom attracts an electron pair that is already in a bond to it. If you could get neon in a bond, it would attract the electrons in the bond very strongly, and that's what the high figure for it means. Of course that's kind of academic because you can't really get neon in a bond, at least not without some serious effort to force it, but having values for electronegativity for all of the 118 known elements helps us compare them. That's why I have not used Pauling's values here (which have some problems), but rather the values of A. V. Kulsha and T. A. Kolevich, which so far as I know is the only scale that can compare all 118 elements, and corrects some problems with the Pauling scale. Incidentally, A. V. Kulsha is also a Wikipedian with the username Droog Andrey. He is a computational chemist,as already mentioned, and his involvement in this group 3 discussion since 2018 has been very helpful indeed. Prior to his explanations I was erroneously on the La side because I didn't know enough. So, as you can see, we have even had a subject-matter expert opine on this change, and he's for it. That's not to say that a non-expert cannot provide something useful here, it just means that an expert's opinion has some weight. (If you want to know what problems there are: the Pauling scale tends to overestimate the electronegativity of atoms that like bonding to other atoms of the same type. That's why gold (Au), for example, is estimated by Pauling as 2.54, when really it's more of a 1.93. It is not actually as electronegative as carbon! But beginning students are often given the Pauling values because they're quite on the mark for the simplest elements. Because of this, chemists typically don't refer to Pauling values much when it comes to heavier elements, but that's a shame: the idea of electronegativity is still very useful, it's only the scale that's a bit wrong.) It turns out that elements nearer a noble gas configuration are, unsurprisingly, more violent in their desire to get it. It is within reach. Most elements, however, are not so close. That's why the number of electrons in the outermost shells ready for bonding matters a great deal for chemistry. These are called the valence electrons. The number of valence electrons, and valence vacancies (how many electrons are missing from the outermost shells), is what is behind the structure of the periodic table: elements in the same column have the same number of valence electrons and valence vacancies.
Now let us see how the periodic table is built up, following how electrons enter their shells as we build up an atom. One would imagine that electrons would start filling in order of their energy levels: the lowest-energy first shell fills first, and then comes the second one, the third one, and so on. The first row has only two elements: hydrogen (H, Z = 1) and helium (He, Z = 2). They confirms what we expected. Although hydrogen is not really an alkali metal in its chemical properties, it has a similar electronic structure to them, and has more similarities to them than any other elements. Helium completes the row with a stable structure. We may write the electronic structures of these two elements as "1" and "2" respectively, indicating that number of electrons in the first shell, and nothing else in any higher shells. The second row starts with lithium (Li, Z = 3), which begins filling the next shell: its electronic structure is "2, 1". It is a reactive alkali metal, tarnishing readily in air, because it is giving up its extra electron to the oxygen and even the nitrogen in the air. The second row then continues as you might expect: beryllium (Be, Z = 4) has electronic structure "2, 2", boron (B, Z = 5) has "2, 3", carbon (C, Z = 6) has "2, 4", nitrogen (N, Z = 7) has "2, 5", oxygen (O, Z = 8), has "2, 6", fluorine (F, Z = 9) has "2, 7", and neon (Ne, Z = 10) has "2, 8". We've seen many of these elements already. Neon has a stable structure as the first two shells are both completely full. The third row then starts with sodium (Na, Z = 11). (The Na comes from the Latin and German name natrium. Thankfully this sort of obtuseness in symbolism is not common!) As one might gather, it begins to fill the next shell, with electronic structure "2, 8, 1". It is also a reactive alkali metal, in fact more reactive than lithium because its outermost electron is farther away from the nucleus. Typically, the outermost electron would be of course affected by the nucleus, but this time it's further away and the nucleus has less hold over it. To add to that effect, the 8 electrons in the second shell are now also there, and they have a negative charge and partly screen the electrostatic attraction the nucleus would otherwise have on that outermost electron. Thus, sodium gives away its outermost electron more readily than lithium. Similarly, sodium also has a lower melting point than lithium. This is because they are both metals, and the way metals form a solid is to have a sort of "shared custody" arrangement of their electrons. A whole bunch of lithium atoms huddle together and share their electrons. Each lithium atom contributes its outermost electron only into a great "sea" of electrons that flow happily around the whole structure. Since the lithium atoms, having each contributed an electron, are now positively charged, there is a large attraction between the positively charged lithium atoms and the negatively charged electron sea that surrounds them. We call this metallic bonding, and the electrons are said to be delocalised. That's why metals conduct electricity, having mobile charge carriers. You should see the similarity between this and covalent bonding: both involve the sharing of electrons. Indeed, graphite (the other form of carbon, as you may remember) is a nice example of this. Only three of the outer electrons of each carbon are involved in the covalent bonding. The other one is delocalised, and that's why graphite conducts electricity. Now, of course sodium does the same thing. But the nuclei have a weaker hold on the electron, so the bonding is weaker, and so it takes less heat energy to break the bond. That's why sodium melts at a lower temperature than lithium. It is this periodic recurrence of similar chemical and physical properties, as a function of atomic number, that earns the principles building up the periodic table the name of the periodic law. That's an important fact: the periodic law applies to all elements. Something that cannot manage to explain all elements, or at least all but a few for which you can justify that something extremely weird is going on, cannot be accepted as part of the periodic law. As a matter of fact, this also explains what's going on with hydrogen: now there's literally no shielding at all of the outermost and only electron from the nucleus, and so hydrogen is even more reluctant to give up its outermost electron. In fact, it's so weakened that it doesn't even have the strength to form a metal, because the nucleus holds on to that electron too strongly for anything like the shared-custody arrangement lithium and sodium can come up with. Hydrogen instead resigns itself to covalent bonding. It has one valence electron and one valence vacancy. Yet there is not as much difference between hydrogen and the alkali metals as you might think. The entire thing about acids and bases in chemistry starts from the idea of chemicals giving up protons, i.e. H+ cations. These are of course tremendously polarising and are instantly solvated by a lot of water molecules, because water (H2O) is polar covalent: hydrogen has electronegativity 2.20, oxygen has electronegativity 3.50, obviously the negative charge on the oxygen is highly interested in this disturbance. And doesn't that formula H2O remind you of those formulae Li2O and Na2O for the compounds of lithium and sodium with oxygen? It's worth noting that when the Russian chemist Mendeleev made his first periodic table in 1869, he hadn't any idea about atoms, much less electrons. All he had were those periodicities. And yet, he not only managed to exactly get this sequence right, but he also managed to predict some missing elements that would later be discovered. And he even managed to predict their properties extremely accurately. What a genius he was! But let's leave this digression and continue our exploration of the third row: sodium, magnesium, aluminium, silicon, phosphorus, sulfur, chlorine, argon, taking us from Z = 11 to Z = 18. As we might expect, the pattern continues: the third-row elements act generally like the "big sisters" of the second-row ones. And indeed, the third-row elements are in general less electronegative than the second-row ones and more metallic, for that very reason we've just seen comparing hydrogen to lithium to sodium. (They give up their outermost electrons slightly more easily!)
The column names are Roman numerals, explaining how many electrons there are in the outermost subshell. How simple, how beautiful! And we can use the properties of the second-row elements to predict those of the third-row ones too! I've put helium in the second column, because it has two electrons in the outermost shell, like beryllium and magnesium. Normally you will see it placed with its noble gas sisters, neon and argon, however. But we can already see it's the end of the line, as nothing else is there in the first row. As well, it gives a consistent electronegativity trend: notice how helium is more electronegative than beryllium, but it's actually less electronegative than neon. Therefore, for this illustration I have elected not to do this standard move for pedagogical reasons. But something should make you worry. Didn't I just tell you that the third shell could hold eighteen electrons? What on earth is argon playing at then, forming a noble gas as if it were full? There's ten more spaces in the third shell to fill! Why does the row end there? Well, let us proceed anyway to get a feeling for the ground we are standing on. Our disquiet then steps up. The next element is potassium (K, Z = 19). It does not put a ninth electron in the third shell! Instead it goes straight to filling the fourth shell: 2, 8, 8, 1! And calcium (Ca, Z = 20) keeps doing the same thing: 2, 8, 8, 2! What is going on?! What happened to the stability of filled shells?! And when is the third shell going to be finished?!
To answer those questions, I will have to admit something: I simplified at the beginning. Indeed, this took a while to figure out by actual chemists and physicists in the early 20th century, so you're not alone. ^_^ Actually, the shells aren't the simplest structure. Each shell is split into some subshells. These were initially suspected at due to spectroscopy in the 19th century, because that comes from finding the spectra of elements, which comes from the energies it takes to excite electrons into higher shells – and also higher subshells. Of course, the spectroscopists of the time couldn't know what they were seeing, but we still use their names for the subshells: s for sharp, p for principal, d for diffuse, f for fundamental. These are distinguished by their angular momentum, measured by their azimuthal quantum number (symbolised by l, a lowercase L): s has 0, p has 1, d has 2, f has 3. The shell they split from is numbered n: that's the principal quantum number. It doesn't change. After that we just go through the alphabet. g, h, and so on. (Well, actually skipping j, since not all languages distinguish i from j!) But those subshells were not known to the spectroscopists. They are not filled in any elements we actually know yet, which is the reason. So it doesn't really matter that eventually we're going to run into p and s again singing our ABC's, because long before that we're going to reach the point where the nucleus is so bloated and unstable that it doesn't last long enough to do any chemistry. ^_^ It turns out that each subshell is divided into orbitals. Each orbital can fit exactly two electrons: one spin up, one spin down. That's called the Pauli exclusion principle. But the subshells have different numbers of orbitals. The s subshell has just one orbital. The p subshell has three; the d subshell has five; the f subshell has seven. And, yes, the g subshell would have nine! It follows the odd numbers. I'm afraid you're going to have to trust me on this: you need to understand some very difficult mathematics to understand why it does that! The nth shell has n types of subshells. So those of you who recognised that the odd numbers, summed up, produce squares, will now immediately see where the 2n2 came from!
Now, it turns out that orbitals with higher angular momentum are higher in energy. So 2s is lower in energy than 2p. But it's a pretty small effect: the difference between 2s and 2p is quite small. In fact it's small potatoes compared to the difference between 1s and 2s. That's why beryllium (Be: 2, 2) can be a metal in the first place, actually: you'd think that both outer electrons would go to 2s and be happy due to the energy gap, but no, it's well worth it to promote to 2p for bonding. And it's also why a molecule like CF4 is actually a regular tetrahedron. You'd expect the 2s electrons on carbon to behave differently from the 2p ones, but actually the orbitals do some sort of complicated mixing with each other. It's called orbital hybridization. You've got one s orbital and all three p orbitals involving themselves in this hybrid, so we call it an sp3 hybrid. That's needed to form four like bonds. The transition metals, which have d orbitals available, can form hybrids with those d orbitals, and so on. I am well aware that the above is simplified. Indeed, MO theory will not give you such hybrids for CF4. However the idea of 2s and 2p both being close in energy and both contributing to the bonding molecular orbitals is correct. In the 3rd shell this effect is bigger. This gives the 3rd row elements a difference from the 2nd row ones: the 3s-3p energy gap is bigger. So hybridisation is significantly more difficult. And it gets even worse: by the time you get to the 6th row, there's something called an inert pair effect, in which the 6s-6p gap is so big that the 6s electrons may not find it worth bonding at all! This is a general principle called kainosymmetry: the first orbital of each given angular momentum (1s, 2p, 3d, 4f, 5g) is smaller and lower in energy than you'd expect. The nucleus has a whole lot more control over them than you'd think. It's another manifestation of the periodic law, leading to the general first-row anomaly that Mendeleev already noticed. The first element in each vertical column is always the weird one in the group. (In order to not mess up this generalisation, I have not moved helium into the noble gas group above like almost everybody does. That's just for pedagogy, you'll just have to note that almost everybody does that to keep the noble gases together.) In fact, by the time we get to the 3rd and 4th shells, this effect is high enough that the energies start overlapping. In fact 3d is very close in energy to 4s. So close, in fact, that minor differences in chemical environment can impact which one is higher! (The effect of kainosymmetry is sometimes called "primogenic repulsion" or referred to as a consequence of an absence of radial nodes, but it is well-established in chemistry.) Now we understand what's really going on with the building up. The real configurations were:
(You see why the notational shorthand was invented. Sometimes a "1" superscript is left off and treated as understood.) Now we've filled a shell, so we go on to the next one.
We've exhausted 2s. On to 2p! There are three p orbitals, usually called px, py, and pz.
A block is a part of the periodic table in which one type of subshell is being filled. As you might imagine, there are four blocks: the s block, the p block, the d block, and the f block. So we've seen that the first four elements are s block elements (or, for short: s elements), and the next six are p elements. Really, the three p orbitals are identical. The fact that I chose to pledge loyalty to the alphabet and fill in x first doesn't make any difference: you literally can't tell which is which. That's why in general we write, say, nitrogen as 1s2 2s2 2p3, with the understanding that you know how those electrons are arranged. The occupancy, however, is real. What you are seeing is a manifestation of Hund's rule. You see, two electrons can fit in each orbital, but they kind of don't like it. They're too close, they repel each other. It's kind of like strangers going up the bus: they don't want to sit next to each other unless they have to. So the electrons first occupy each orbital singly, and then pair up once they have no choice. You can actually see this in action. The first ionisation energy of an element is the energy it takes to wrest an electron from it to produce a positive ion: X becomes X+. (And of course, the second ionisation energy measures the energy it takes to go from X+ to X2+; the third ionisation energy from X2+ to X3+; and so on.) If you compare first ionisation energies across a block in one row (e.g. boron through neon), you'll see they usually rise from left to right, which makes sense; the shielding effect from the inner electrons is about the same, but the nucleus has more protons, it's more positively charged, the pull is greater. 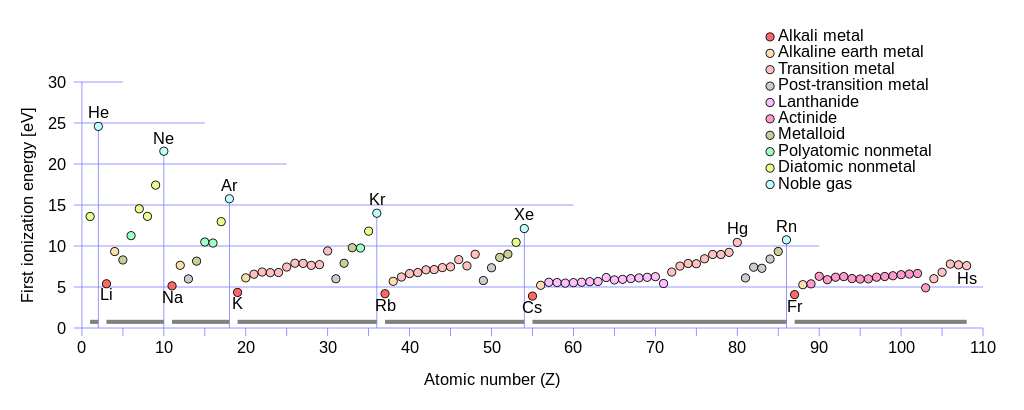 Except that you can see a "blip" from nitrogen to oxygen. That's because the electron being gotten rid of in oxygen has a partner; the one being gotten rid of in nitrogen doesn't. It's easier to remove the oxygen one because it's got repulsion from its partner already. (The blip from beryllium to boron is due to going from removing a 2s electron to a 2p one. The latter's easier, it's a little bit higher in energy.) This effect is called double periodicity. Each block kind of splits into two: a first phase where the electrons go in singly, and a second phase where each electron gets a partner. This means that you can often tell from physical, chemical, and atomic properties where a half-filled and filled subshell occurs. The effect lessens for heavier elements, though: it is strongest for the kainosymmetric orbitals 2p, 3d, and 4f, the first of their kinds. (For the s orbitals this is basically invisible as half of two is just one, so every element is special.) You can see that in how the blip is much smaller from phosphorus to sulfur, and eventually it becomes not enough to actually cause a decrease (though it is still enough to make the expected increase not happen). You can see that for these second row elements, to get the stable octet you have to count both 2s and 2p electrons. Indeed, both are participating; even for lithium and beryllium, 2p acts like an easily accessible subshell for valence vacancies, that bonding atoms can contribute their electrons too. That's why beryllium is a metal, as I've said earlier. Lithium can also use its 2p subshell as an electron sink from surrounding atoms bonded to it, in lithium metal, and also in MeLi. The latter is a very standard phenomenon known to all practitioners of coordination chemistry as back bonding. Such ligand donation also happens into the 4f orbitals of lanthanum. But let's continue relooking at the third row. We'll use the shorthand notation now.
In order to not make everybody's eyes bleed at the notation I have used the common noble-gas shorthand: [Ne] simply means 1s2 2s2 2p6, the electrons forming the noble gas core of neon. Again, since ionisation never goes beyond a noble gas as far as we know, this is totally fine. So [Ar] would mean [Ne] 3s2 3p6 = 1s2 2s2 2p6 3s2 3p6 (you see why the shorthand quickly gets useful). You don't usually see the shorthand for helium; that's because 1s2 is not much longer to write than [He]. Of course, we still know that phosphorus has the three 3p electrons in three separate p orbitals, for instance. So: here we may say that 3s and 3p are where you find the valence orbitals and vacancies. (For sodium 3p involvement is rather weak, I haven't found a good proof of it yet. For magnesium, however, it's well-known.) Now we may go on with fresh eyes!
The Aufbau principle, also known as the Madelung rule, and which was given a theoretical justification by V. M. Klechkovsky in the 1950s, provides a generalisation for how the orbitals fill. We definitely cannot explain to you Klechkovsky's derivation of it (I have not seen it and likely wouldn't understand it either). But I can explain to you the rule. Step 1. Arrange the orbitals in order of increasing n + l: that is, principal quantum number plus azimuthal quantum number. Within each group, arrange them in order of increasing n.
This gets us to element 120, two beyond what we have now. So we stop here. Step 2. Follow this order and put them in a line. 1s | 2s | 2p 3s | 3p 4s | 3d 4p 5s | 4d 5p 6s | 4f 5d 6p 7s | 5f 6d 7p 8s | ... This already gives the order in which the subshells fill. But to get a periodic table from it we must take one more step. Step 3. Now move the dividers to all go just before an s orbital. (Why do we do that? Because we want a period to end at a noble gas and start at an alkali metal! And why do we want that? Because we know that the big energy gap happens between a p shell and the next s shell, or between 1s and 2s in the first period; that's why you don't see ionisation past a noble gas in either direction!) 1s | 2s 2p | 3s 3p | 4s 3d 4p | 5s 4d 5p | 6s 4f 5d 6p | 7s 5f 6d 7p | 8s ... Let's arrange this as a periodic table. 1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s 4f 5d 6p 7s 5f 6d 7p You'll see that this is exactly the standard form, in its version as a Lu table. (Well, except for helium, which is an obvious one-off exception to keep the noble gases that the whole form started from together. That's not much of a precedent for breaking the pattern for La. Helium over beryllium does look odd: helium and neon are very inert gases, beryllium a metal that likes to lose two electrons. Yttrium, lanthanum, and lutetium are all metals that like to lose three electrons each; there's nothing much odd about Y over Lu. In fact, we will soon tabulate properties and find that there is a lot more odd about Y over La.) There is something called Janet's left-step periodic table, that omits the third step entirely, and looks like this: 1s
2s
2p 3s
3p 4s
3d 4p 5s
4d 5p 6s
4f 5d 6p 7s
5f 6d 7p 8s
It has been recommended by some, such as the late Henry Bent of Bent's rule fame. However I think the divide between the noble gas and the next alkali metal is so salient that the typical form is better. But of course, a theory by itself is just a theory. How does this work out in practice? It turns out: very, very well. The f, d, and p blocks correspond exactly to elements for which the highest angular-momentum orbital used is f, d, and p. The break from symmetry in step 3 to accommodate the s block has a real chemical meaning: because of the break happening from the noble gas to the alkali metal, the s orbitals fraternise with the orbitals of the next value of n + l, rather than their own, and stay active throughout the row instead of going silent after their block like the rest do. Let's carry on to the fourth row. We can see that 4s is expected by Aufbau to fill first. And it does!
The symbol of potassium comes from the Latin and German name kalium. As I've said, though, such obtuseness isn't common. However, we should also note that just like there was some 2p involvement in lithium and beryllium, and 3p in magnesium, there's also going to be some mixing in of other orbitals later in the s block. Calcium indeed has some 3d involvement. That's why CaF2, as a gaseous molecule, has a molecular shape you wouldn't expect, because of the repulsion of the extra 3d electrons. You don't have this in MgF2, say. (But for magnesium you do instead have the 3p orbitals working.) So let's now look at what happens next. If Aufbau's right, we should expect ten electrons to file into 3d in a nice and orderly fashion. So let's see what happens. These elements are the first row of the transition metals, that start filling an inner shell.
(The symbols Fe and Cu come from Latin ferrum and cuprum respectively.) That's almost right. Indeed, the electron shells being used for these include 3d and 4s. Also, we can see that the 4p shell is vacant, but it's still part of the valence régime, being in the same row: and indeed there are some compounds (typically when the transition metal hasn't lost a lot of electrons) in which 4p is measurably contributing to the bonding. Only 3d, 4s, and 4p contribute. The Aufbau generalisation is working plenty fine. (Some people will tell you that zinc is not really a transition metal because it supposedly doesn't use its 3d orbitals for chemistry. They are wrong. It does. Zinc fluoride, ZnF2, is an example in which the zinc 3d orbitals significantly overlap with the fluorine 2p orbitals to create a chemical bond. The mere fact that you never see zinc having lost more than two electrons does not disprove this involvement of 3d.) But what's with those exceptions? What do I mean by that funny comment about nickel? Wasn't Aufbau theoretically derived from first principles? Why is it wrong for some elements?
To answer that, I'll have to admit that what I told you at the beginning was a little bit incomplete. That's a common thing, please don't give me a sour face! We always start with something simple for pedagogical reasons. Just think of when you learnt in elementary school or even kindergarten that it doesn't matter what order you add two numbers. We generally illustrate this then just by looking at examples and moving objects around, not by an actual proof from Peano's axioms, and most certainly not the way Carl E. Linderholm does it in his hilarious Mathematics Made Difficult:
Anyway, on to my confession. I told you that electronic structure matters for the chemistry of an element, and I told you that we only usually care about the ground state for chemistry. That's a great approximation for the noble gases, and it's okay-ish for most s and p elements (but you must already be careful about hybridisation). The trouble is that, as I also said, the process of forming chemical bonds also releases energy. If the ground state is quite far away from the next higher excited state in energy, like it usually is for s and p elements, that isn't a problem: you can safely assume the configuration you know that I've told you will be the same in all reasonable chemical environments. But for d and f elements you often have a lot of configurations that are very close in energy to each other. Minor changes in the chemical environment will easily cause one or another to be a more stable. Even in the ground state some quantum effects mean that a single configuration will have many variants close but split in energy (those in the know who need the jargon call it the splitting of J levels). That is why nickel is disputed: the lowest-energy state is an [Ar] 3d8 4s2 one, but the average energy of the [Ar] 3d9 4s1 states is lower. You may begin banging your head against the wall now. (Not too hard though, please. ^_^) Therefore, for the d and f elements especially, in general it's not very good to "sweat the small stuff". It doesn't really matter how many electrons happen to be in which orbital, it changes. What remains unchanged, though, is:
This is a sort of "fuzzy configuration". I will use the notation common for it in solid state physics: bracketing a set of orbitals with a number round them. So scandium may be considered to be [Ar] (3d 4s 4p)3, meaning that it has three valence electrons, and that in chemical environments the 3d and 4s and 4p orbitals will be used for forming chemical bonds. Chemists who seriously work with these d and f elements understand this, of course. They know that the irregularities here and there are not useful things to worry about as they predict nothing useful in most chemistry topics.
And here's Glenn T. Seaborg, discoverer of plutonium (Pu, Z = 94) and codiscoverer of many transuranium elements (my bold):
And W. H. Eugen Schwarz (again, my bold):
You just don't often get told that in school, largely because for the main-group elements that you focus on in school, ground-state gas-phase configurations are also the chemically relevant configurations because the promotion energies are too high, and only the first-row transition metals ever come up often. Sticking to that means staying forever at the high-school level, in which case there's not much point in entering this discussion because most high schools don't cover f elements or heavy transition metals in much detail. For that typically a wrong explanation is given for the irregularities at chromium and copper based on the stability of a half-filled or filled subshell, and the dispute about nickel is swept under the rug. It is probably a relief for those teaching that that the second row rarely gets covered (even though it is some sense more characteristic of transition metal chemistry than the first row), because the second row knocks that explanation out of the water. The gas-phase configurations of the d elements are:
(La and Ac have d1s2 in the gas phase.) Despite an often-encountered emphasis on the irregularities, these irregularities have just about nothing to do with which elements are actually more similar to each other. Well, Lr has a weirdly different electron configuration from Lu. Somehow that makes zilch difference for chemistry: it's an excellent heavier homologue of Lu, and in chemical compounds happily makes use of the 6d shell that you'd think it should even though it's empty in the ground state. We can also take a look at the fourth column above. As we can see, Cr and Mo have the same d5s1 configuration, whereas W is d4s2. We might therefore, believing the importance of gas-phase irregularities, expect Cr and Mo to be similar to each other, and W to be distinctly different. Nope. Actually Mo and W are the similar two. Cr is the different one. We'll understand the real reason for that soon enough. Also, we might expect Rg to be distinctly different from its lighter homologues Cu, Ag, and Au, which should be more similar thanks to the electron configurations. Nope. Actually Rg is an excellent heavier homologue of Au. Cu, Ag, and Au however behave really quite differently from each other. And indeed, the fact that nobody has ever used this argument to move chromium and copper out of their columns shows that to some extent this is understood. Only the total number of valence electrons counts for group assignment, which comes after we assign blocks based on number of valence electrons and vacancies. As you can see, the approach I am advocating here is exactly what Jensen has stated is the basis for periodic table placement:
I just go one better than Jensen to really make the assignment of Lu and Lr to group 3 absolutely clear: I claim, following many, many statements in the literature, that actually even for lanthanum we only need to get to criterion 1 because lanthanum already has low-lying non-hydrogenic valence 4f orbitals that contribute to the bonding. Lutetium does not have any such thing. We will come to that in due course. There's only one useful thing we get out of this, which is a sense of the relative energy levels of the (n−1)d and ns subshells. The energy gap is rather small for 3d–4s; for 4d–5s it is rather big, with 4d quite a bit lower in energy than 5s. It closes back again for 5d–6s, and 6d is even higher in energy than 7s. In fact it is so high that 7p gets even more significant involvement than you'd think (viz. Lr and Rf, for which 6d17s27p1 is a low-lying excited state). That's somewhat useful to know, although even that is just first order: as you ionise lawrencium 6d falls back under 7p like you'd expect. (Same for lanthanum 4f, actinium and thorium 5f.) But, again, the specifics are silly. A compound of chromium (Cr) with no electrons lost is probably not d5s1. In fact it may very well not even be d4s2. Most likely it is d6s0. Chemical environment matters far too much, and it drowns out the small energy differences between all these configurations. Why worry about them? So let's go back to the previous elements and write their configurations in the fuzzy way.
We've gotten back to what looks like the old "2, 8" style of configuration, but what a difference in understanding we now have!
You may recall the issue about the s block. It's joined the "wrong" n + l value party, but it also doesn't seem to be quite sure about it. That's why sodium 3p, and potassium 4s and 3d, and calcium 4p, are rather iffy. But in general, you will find some of this gatecrashing in every period, and you'll notice that unlike every other block's orbital, the s orbitals don't go down into the core once they've been filled, but stick around for the whole rest of the row.
You can incidentally see that manganese is the half-filled element, and zinc is the filled element, when it comes to filling up the 3d shell. When ionising electrons, suddenly we're back to an approximately alone element, and we can use gas-phase configurations again. By going to the 3rd ionisation energy, we get rid of the 4s electrons above, and probe how filled the d shell is. The answer writes itself.  And we see the same trend in the melting and boiling points:
Well, a half-filled or filled subshell element will have the electrons in that subshell not really so keen to leave their atom, because those arrangements are stable. Therefore you expect them to have lower melting points because they have electrons less willing to be delocalised and contribute to a metallic bond. Lo and behold, that is what manganese and zinc show up as doing. This is absolutely standard stuff. You may find it in Greenwood and Earnshaw, 2nd edition, p. 1075.
And as we can see, they hit the nail on the head regarding this effect always being strongest in the first row and weakening thereafter. Same for 2p, same for 3d, same for 4f. We'll go back to this criterion for determining half-filled- and filled-subshell elements later, when it weighs in on the question of the f block. As a preview, however, I will note that it is always Eu and Yb acting like Mn and Zn do, never Gd and Lu. That's exactly what strongly supports that Lu belongs under Y and that La-Yb are the real f elements. The rest of the fourth row returns to main-group style.
Only in gallium does 3d fall silent! And we've made it to the next noble gas. I have used a pipe to separate the "core" from the valence electrons. This is not standard notation, but it is helpful for the pedagogy. 3d has been filled, it is now drowned inside the core, you'll never get it involved in chemical reactions. It is still part of the third shell, after all: it's not that far from the nucleus, and in fact its distance from the nucleus is about as much as 3p. So now 3d falls away into the core, whereas 4s and 4p are still going strong above. Notice how depending on the element we had, the energy-level order of 3d and 4s has changed. That shows how close they are, which is precisely why worrying about the precise occupancy of 3d and 4s in a neutral, gas-phase atom sitting all by itself is something like counting how many angels can dance on the head of a pin when the pin is in fact about to be recycled in the next few seconds. But things are not quite the same. Aluminium through argon have really been basically the big sisters of boron through neon: everything is the same, one more period up, outside the noble gas core. But for gallium through krypton the core is not that of a noble gas anymore. It has ten extra electrons in a d orbital that shield the nuclear charge rather badly. (s shields the best, followed by p, followed by d, followed by f.) As a result, the nucleus now has ten more positive charges than you'd expect. And the outer electrons feel it! That's why initially the electronegativity goes back up. A setback in metallicity is observed because the nucleus has now gotten stronger. (Along the 3d row, as well as if you look from the 3p element to the corresponding 4p element.) The cations are now smaller (the nucleus tugs at the outer electrons stronger now!), they are more polarising! When dissolved in water the cations are more acidic: gallium is more strongly acidic than aluminium. So, they are kind of like "big-little sisters". ^_^
(Those Roman numeral group numbers are my idea, albeit inspired by Gary Wulfsberg's numbering of the f group in his Principles of Descriptive Inorganic Chemistry. They count the number of valence electrons and the block. The Arabic-numeral IUPAC group numbers are more common, in the next row, even though they signify nothing but "finger-count" labels. Here I use those Roman numeral group numbers because I am basically "teaching" here; of course, I do not propose to use them for WP, because they are OR at its finest.) This is our first encounter with a contraction. As the 3d electrons go in, the outermost electrons that determine the radius of the atom suffer more and more charge from the nucleus because the 3d electrons are not fully screening this charge away. That happens actually absolutely everywhere in the periodic table: here's a table of single-bond covalent radii in picometres. (The values are from Pekka Pyykkö, corrected by Kulsha and Kolevich for cerium, which Pyykkö gave as 163 pm, which is too small! O, F, and Ne are also not quite right, but when I asked Droog Andrey about the corrected values he couldn't remember them.)
The end result is that the expected increase in size from going to the next shell is wiped out for the elements that immediately follow a kainosymmetric contraction. Since size impacts electronegativity (i.e. polarising power) by Fajans' rules (charge is also a factor, remember!), it is not surprising that everywhere outside the s block, going down a group (vertical column) results in a "kinked" trend that alternates between going linearly and staying about constant, because at every even period a new block is inserted and a new contraction asserts itself. That's exactly why arguments along the lines of "but basicity should increase down the periodic table" or "but ionic radii should increase down the periodic table" do not work. (I used to believe them. Now you see I know more.) Just see if they do after a contraction. That's in fact absolutely famously a side effect of the lanthanide contraction, which almost exactly cancels out the expected expansion going from the fifth to the sixth row when you'd think the electrons should go from the fifth to the sixth shell and get farther away. Oops, I am getting ahead of myself, but you can see that this situation is not standing in a vacuum. ^_^ Only by the later elements, selenium through krypton, have things recovered somewhat. Even then, notice that the electronegativity dip is nowhere near as big as what it was from 2p to 3p. That's exactly following the general first-row anomaly principle: the first element is the most distinct, because contractions wash out the expected later changes. And just observe the difference from Na-K and Mg-Ca at the very beginning with no such insertion and a completely straightforward fall in electronegativity. In case you noticed that the half-filled 3d elements Mn and Zn displayed a dip in electronegativity: congratulations. The reason for that is that these elements find it harder to go beyond losing two electrons (corresponding to getting rid of 4s), because a half-filled or filled 3d shell is more stable and it's harder to pluck electrons out of there and get them in chemical bonding. Remember what I said: an element becomes more electronegative as it loses more electrons, although generally the effect's pretty small (but it's still enough to cause a dip here). These Kulsha-Kolevich electronegativities consider lots of chemical environments, so this stabilisation of +2 helps to make the element less electronegative. We will come back to that. By the way, it doesn't happen for the s and p blocks, because the shell filling is then the outermost one. So there's a difference between the so-called "main group" s and p blocks, filling outer shells, and the "transition" d and f blocks, filling inner shells. We may now completely mechanically fill in the fifth row correctly: it exactly follows the fourth.
The 4d subshell stops being a valence subshell for indium through xenon. However, compare: gallium [Ar] 3d10 | (4s 4p)3 vs indium [Kr] 4d10 | (5s 5p)3. Indium is really the big sister of gallium in a way gallium isn't quite the big sister of aluminium. This, combined with the kainosymmetric effect that the first subshell of any kind is smaller than expected and closer to the nucleus, explains the even row vs odd row difference that is a completely general thing in the periodic table with only the s block as an exception. Because an even row element is either kainosymmetric, or it has suffered a kainosymmetric contraction; so it is smaller, its valence electrons are less keen to be chemically involved, more polarising, more acidic, more electronegative. So it favours losing less electrons as well (i.e. lower oxidation states; higher oxidation states either do not exist or are strong oxidants). An odd row element behaves in just the opposite way. One can see this in 1s vs 2s (the only s block row with this situation); 2p vs 3p or 4p vs 5p; 3d vs 4d; 4f vs 5f. Things are not exactly so clear cut from 5d onwards because then the electrons are going fast enough that relativity must be considered, but until then it works well as a generalisation. I remind you of the periodic chart: 1s row 1 2s 2p row 2 3s 3p row 3 4s 3d 4p row 4 5s 4d 5p row 5 6s 4f 5d 6p row 6 7s 5f 6d 7p row 7 It's blatantly obvious from the "two rows at a time" shape what's going on. The first row of the p, d, and f blocks always happens in an even row. That will be kainosymmetric, the elements in it will get the effects from the valence electrons being too close to the nucleus. The next row will form normal "big sisters"; it's an odd row. But the next row after that is another even row and has a new block inserted before it, causing a contraction; that's a new even row. You can see the s block stands out in two ways immediately. Firstly, its first row happens in an odd row. That's unique. Secondly, after the initial situation of 1s vs 2p, the previous (shielding) column stays as a p orbital after that. Therefore you don't have any effects from the incomplete shielding causing a contraction as we're always starting a whole new shell. The only small effect you get comes from 1s shielding even better than 2p; after that, everything remains constant. That's why it's not a problem for the theory that the s block often behaves weirdly. s electrons not going into the core after their block, higher orbitals intruding, total lack of even-odd periodicity. The "original sin" has already happened in the big energy gap being between p and the next s instead of between n+l values: the situation really is categorically different. That's exactly why to stick with alkali metal to noble gas and not Janet's table; it shows this anomaly and 1s as a simply spectacular situation (the only period length with no "big sister"; see the length pattern 2-8-8-18-18-32-32 with only 2 not repeated; and it suffers kainosymmetry galore). That's why I favour helium over beryllium incidentally: it shows how exceptional 1s is without hiding it, but I understand if you prefer helium over neon for chemistry. It won't cause any other forced changes as helium's weirdness over beryllium is simply unique. Why one would ever want to drag this situation into the d block, as the La table does, is beyond me. But we'll get to that in good time.
The results of the electronegativity drop that is now allowed are spectacular. In the 2nd row, beryllium was the last metallic element; in the 3rd, it's aluminium, one step later. In the 4th row it stays at gallium due to the 3d contraction. But in the 5th row it jumps two spaces to the right: not only tin, but also antimony, skews more to the metallic than the nonmetallic side! (Alloys of antimony with most elements conduct electricity like metals. That's true for tin as well, but false for germanium and arsenic.) Notice how the late 4d elements seem to be more electronegative than the late 3d ones, but the opposite's true for the early ones? Well, that's because of the 4d to 5s energy gap, which widens significantly as the row is traversed. So it starts smaller than the more constant 3d-to-4s one, but it ends larger. The reason I mention this now is that we will see a total analogy to the f transition block: the 5f-to-6d gap starts smaller than the more constant 4f-to-5d gap, but it ends larger too. Notice how the electronegativity dips at Tc and Cd are still there. For technetium, the drop's smaller than for manganese. For cadmium the 4d electrons are more core-like than the 3d ones of zinc, so basically it becomes like the electronegativity-dropping trend of the 5p elements immediately afterwards; still, we can expect some minor usage, looking at ionisation energies, they're not totally core yet. That comes at indium. One fascinating side effect of the electrons getting farther from the nucleus is that the noble gases stop being completely inert! Elements like fluorine with their huge electron-grabbing powers can attack krypton and xenon and force them into compounds. (Not that they're quite happy to be in such compounds. XeO3, xenon trioxide, is quite ready to explode at the slightest provocation back into xenon and oxygen gas.) It seems that helium and argon may not be barren either, but so far a neutral compound of neon seems elusive. This weird order of reactivity He > Ne < Ar < Kr < Xe < Rn < Og is another reason why, for pedagogical reasons, I have chosen to display helium above beryllium throughout. The main reason of course being that this is an electron-based explanation of the periodic table. ^_^
The reader may at this point complain: "I have sat through reading six parts of this. And while it's mighty interesting to see you teach basic high-school chemistry and generalise it to all elements rather than just the ones for which the naïve approach works, I came here to learn about the dispute about where lanthanum and lutetium go in the periodic table. Are you going to get to it or not?" Well, fear not. We've done so much work already in setting the scene. Now I can get the problem sorted out extremely quickly by using the powerful tools we have explained and used the first five rows to illustrate. Or rather we could, if not for the need to refute misconceptions. (As a little test, by the way: looking at how the first five rows were filled, would you expect the f block to go between the s and d blocks as in the Lu form, or to be jammed between the first and second columns of the d block as in the La form? I daresay no one would guess the latter looking at this obvious pattern.) The first two elements of the sixth row are s block elements, filling the 6s shell as Madelung demands.
Now we have the next element. Lanthanum turns out to be the first element to have significant usage of its 4f shell for chemistry. It's thus called an inner transition metal, because it's filling not its second-outermost shell, but its third-outermost shell. Allow me to quote our article on the periodic table itself (text which I wrote there, of course) to back this up with a fistful of citations.
As you can see, the evidence for 4f involvement in lanthanum is simply overwhelming, and has been known for a while. People knew of some of this evidence in the 1960s. It is not news. Neither is it historical, as can be seen from Karl Albert Gschneidner Jr. ("Mr. Rare Earth") writing this in 2016: 
(For a poignant note: this was his last article.)
But really, the idea that the start of 4f is somehow "delayed" from where Madelung says it should start at lanthanum is just not true. So is the idea that 4f is in the core. We know so much more than that old idea, thanks to Gschneidner among many others. One can easily see that from excitation energies. Well, chemical bonds go in energy up to 10 eV. The energy needed to excite a bare lanthanum atom to the 4f16s2 state is nowhere near that. Just 1.88 eV in a matter of fact. Of course the chemical environment may very easily provide that. And a La2+ ion minding its own business, while being [Xe]5d1 in the ground state, has [Xe]4f1 as an excited state with an energy of just 0.89 eV. That's general, when these atoms are positively ionised, the inner subshells become more accessible: just compare thorium [Rn]6d27s2 in the gas-phase ground state with no 5f, with Th3+ [Rn]5f1 managing to be the ground state. Though of course with such a small energy gap that in chemical environments you usually see [Rn]6d1 anyway. That's exactly why the looking at the ground state alone loses way too much information. Maybe unless the element is a noble gas. Just hear it from A. V. Kulsha:
A. V. Kulsha even was so kind as to contribute some computations. He did some work on the complexes [M(CO)8]− (M = Y, La, Lu). Here are the highest occupied a2u orbitals of each: For the La complex, the 4f atomic orbital is bonding. For the analogous Y and Lu ones they are not. You can see from the overlaps. No overlap: no bonding. But lest we be accused of OR, just see the following two recent articles. The first (doi:10.1039/C3CP50717C) was already cited above. It notes:
LaF3 is shown to have the highest Mayer 4f–2p bond order of any lanthanide trifluoride (0.057), with cerium (0.055) as the runner-up. Lutetium is at 0.030, less than even iridium (well past 4f drowning into the core) at 0.037. And also doi:10.1021/acs.jctc.6b00238, where you can see that the 4f involvement of La in LaF3 is similar to that of Ce in CeCl2−
They investigate Lu as well, where "there is no evidence of 4f delocalization in the population analysis or in the localized orbital representation of the electronic structure". In both you can see proof of La 4f involvement that is not in any sense different from that of cerium, which everyone agrees is a 4f element. So let's go forth and fill in the 4f elements:
(Yes, seriously. Terbium, erbium, ytterbium. Unfortunately good naming sense was not applied to those elements.) This lack of a delayed collapse is something which tends to make La supporters unhappy. See, one of their major arguments is the ground-state gas-phase electron configuration: the logic goes, a gaseous lanthanum atom has configuration [Xe] 4f0 5d1 6s2, with no f electron yet, so it cannot go into the f block. We have, of course, devastatingly revealed the insignificance of such things for chemistry above, so you now know better than to take that seriously. What is also not often remarked is that the whole idea is also logically inconsistent. Because, here are the electron configurations of the f elements in the ground-state gas-phase:
You will notice that while it's true that cerium (Ce, Z = 58) is the first element with a 4f electron in the gas-phase ground state, it's also true that protactinium (Pa, Z = 91) is the first element with a 5f electron in the gas-phase ground state. The argument for throwing lanthanum out of the f block suddenly also manages to throw out thorium. (Of course, you know the real reason, that is the changing 5f–6d energy gap. For the early actinides 5f is high in energy, they act like d transition metals; whereas Cm through No act more like La through Eu.) Well, Jensen has made a far better riposte to this than I can.
Sure, the 4f collapse happens in an odd-looking way. In caesium and barium 4f is high up in energy, it almost doesn't feel the nucleus, and it's not chemically active. (Unless you pressurise the metals.) Then in lanthanum it's still not that close, but it's already low enough to feel the nucleus. Same with cerium, in which it is a bit lower still.
Then after that it tucks itself radially near to the xenon core (but still is high enough in energy to hybridise with 5d and 6s and 6p, go figure). But why on earth should that be reflected? We already saw above that the 4d-5s energy gap changes from yttrium to cadmium. Is that a reason to cut off the earlier elements from the d block? No, better to treat it just as a general thing among groupings of subshells that are close in energy. Moreover, this odd form of collapse seems to be normal for heavy elements. It happens for 4f in La-Ce, in 5f in Ac-Th-Pa, in 6d in Lr-Rf-Db, even in 5g in elements 121 through 126 (predicted). Reflecting just the 4f one, which wrongly displays the even more protracted 5f one, and totally ignoring the 6d one, seems very much like a double standard to me. There is a wonderful paradox about the use of this 4f subshell. On the one hand it's very localised near the nucleus, of a similar radius to the 4d shell in the core; on the other hand it's of a similar energy to the outer 5d, 6s, and 6p shells and can readily take its electrons out to do chemical bonding. And since the collapse of 4f down from its hydrogen-like heights far from the nucleus (H to Ba) to something kind of stuck with the core's radius (Pr to Yb) is pretty swift, with only La and Ce going in the middle of it, the hilarious end result is that actually La and Ce with the smallest f occupancy have the biggest involvement of the f orbitals among all the lanthanides (new paper). This positing of 4f involvement of lanthanum also explains a great many things about it and its compounds at one stroke. We have:
Basically, one s orbital, three p orbitals, five d orbitals get you to 9 bonds to adjacent atoms by hybridisation. Lanthanum happily goes around with 11 and 12 and numbers like that. f orbitals are a necessary invocation. Unless you want to posit that the bonds have order less than 1 on average.
A case study is the complex [Ln(2,2′-bipyridine-1,1′-dioxide)4](ClO4)3. It's amazing to watch what happens to the homologues of this compound with the other lanthanides. As you traverse the 4f series the cube slowly distorts into a square antiprism which doesn't need the f orbitals! What an amazing demonstration of the slow drowning of the 4f orbitals!
All this is standard stuff:
La is a superconductor in group 3 but Sc, Y, Lu are all not. That breaks a correspondence between the transition metal rows. Bernd T. Mathias explained this in 1969:
Hamilton noticed it too in his 1965 article arguing for the change to Sc-Y-Lu, even before Jensen (1982):
So let's look at what Wittig wrote later (it's the same source in that article extract above):
An acknowledgement that 4f character is still needed to explain the results, plus an explanation of why it is hard to actually see, and then evidence from LaAl2 in the next few paragraphs not quoted. What's not to like?
This is ambiguous, which is why I left it late. Wittig did think this was related to the f electrons.
Logical. In fact that idea even was how people found out that thorium has some 5f involvement, as explained above. Sandbh has referred to a 1977 source by D. G. Pettifor purporting to rebut this (10.1016/0364-5916(77)90009-8), explaining the crystal structures of the lanthanides in terms of how much d occupancies they have. I don't know who's right, but either way Lu under Y seems better. Yttrium and lutetium, according to Pettifor, have about 1.5 d electron per atom in the solid state. Lanthanum has about 2.5. And if Wittig's right, then Lu under Y is also immediate.
A "normal" trivalent metal at this spot in the periodic table (right under Y in the d block) should have a melting point around that of Lu (~1600°C) per Mathias: you can just run linear interpolation. As you can see, the 4f elements emphatically do not; La is particularly bad. That's already a pretty strong argument from physical properties. But it's easy to explain everything with 4f: the metals La though Yb (excl. divalent Eu and Yb) are not "normal" trivalent metals, some part of the electron density is hybridised into the 4f shell and is unavailable for delocalisation, therefore weakening the metallic bond. (Remember that from when I explained Li and Na above?) The energy gap widens as 4f gets drowned into the core slowly but surely, so this is highest for La and Ce, and then weakens: you see the melting points rise slowly until they get back to where they ought to be at Lu (Mathias, Gschneidner). Now just look at where the divalency crash happens: Eu and Yb. Clearly, those are the half-filled and filled 4f elements, in complete analogy to Mn and Zn which we explained above. A general principle is revealed itself in the periodic table, but only with Lu in the d block under Y. The double periodicity rides again. Third ionisation energies and electronegativites confirm this, as do (most clearly) standard electrode potentials. For 3d:
and 4f/5f:
In no case does the next element (Ga, Lu, or Lr) fit with the trend, as it strongly prefers trivalence. Obviously, they are doing something else. And I didn't make up these analogies. Jorgensen said the same thing.
Only with Lu under Y do all these relationships and generalisations holding fast throughout the periodic table work.
For lutetium, the 4f shell is totally full, none of this happens anymore, the block's over. It's instead a full-fledged member of the next series.
These 5d elements have all suffered the world-famous lanthanide contraction, or 4f contraction. They stand in relation to their 4d congeners more or less as the 4p elements stand in relation to their 3p congeners. Just compare electronegativities in the table that will come a little bit below. Lutetium is a bit more acidic than yttrium, hafnium is similar to zirconium, only tantalum is a tiny bit more basic than niobium (and it's still so similar that many spurious elements were discovered in the 19th century that were really mixtures of them). However, not all the even-odd effect is working anymore. These elements still prefer to lose more electrons, and the electronegativity rise isn't that much. Indeed, near the end, the 5d–6s gap is still pretty weak. (That's why gold is golden. For copper, silver, and gold, the colour comes from the energy it takes to promote an electron from the s orbital to the d orbital. For copper it gives a reddish colour, as the gap's small. For silver the gap is much bigger, the energy is in the ultraviolet, you can't see any colour. But for gold the gap narrows, so it's yellowish.) You see, what I didn't say very much near the beginning was the importance of relativistic effects. When you have a very very heavy nucleus, the electrons basically have to move very quickly, close to the speed of light, and so special relativity kicks in. This has some funny effects, such as "tearing" the p orbitals further into two subtypes (p1/2 with lower energy, p3/2 with higher), lowering the energy of s and p1/2 orbitals, and raising that of p3/2 and everything else. As a result what you can see here is that 5d rises in energy and partly counteracts the lanthanide contraction effect. This is most true near the end of the series: platinum, gold, mercury. This is where relativistic effects are maximised. In fact the 6s shell is so stabilised that the metallic bonds suddenly become quite weak, the melting points drop – and now you know why mercury is a liquid! ^_^ What La is typically accused of (not having valence 4f electrons, supposedly having weak 4f involvement when actually it's one of the most direct 4f users in the lanthanides) is actually quite true of Lu: it has no valence 4f electrons and no 4f involvement at all in its compounds. I do not understand why La advocates propose to only throw La out of the f block but not also Lu. At the most you might get the idea that Lu is analogous to Zn, which I used to believe, and which looks quite convincing until you learn (see above) that actually zinc is using its 3d orbitals for chemistry after all in about the same way lutetium fails to use its 4f ones. I also don't see why only the start of the block is important. (More so, I don't see why La having no 4f electron is important but Th having no 5f electron is apparently unimportant.) We may compare La and Lu with the uncontroversial 5d elements Hf through Hg in physical properties.
Sometimes La looks okay-ish, but it never has a very significant advantage over Lu. And we'll soon find out how La is wrong even in those forms by examining double periodicity in the f block. Alvarez published a paper this year even giving more examples, some from chemical properties (e.g. oxygen affinity). To which I can add acid-base properties (because Lu3+ is much smaller, more like a 5d cation in size and thus polarising power). The fact that Lu is more acidic than Y is exactly a parallel with how Ga is more acidic than Al: Ga has just suffered the 3d contraction, Lu the 4f one. Wittig also looked at superconductive properties for Lu, and arrived at the very same conclusion.
It's been argued before that increase of basicity should be a general principle going down the periodic table by Trifonov. But, Ga being less basic than Al, Hf not being more basic than Zr, makes this somewhat questionable. Of course, we mentioned this earlier, so you understand it too. ^_^ Trifonov supported La under Y on that basis. He did note that La has 4f involvement, but he considered it insufficient to put La in the f block. (But, really, Lu in the f block looks even worse with less 4f involvement than even the much-maligned supposedly weak f involvement of La.) And look at all the properties Jensen followed Chistyakov in collecting:  I admit that there is some selectivity here. Indeed, as you saw above, there are some properties where La looks not worse. But you will not find any where it looks significantly better. As you can see, Lu is far more like a d element than La is; many chemists realised this too in the 1920s. When separating the rare earth elements, you have more basic "cerium group" elements and less basic "yttrium group elements". Sc and Y are in the yttrium group, and so's Lu. La, however, is in the cerium group. Whereas La looks totally normal in the f block:
Eu and Yb showing double periodicity supports La in the f block too. That works actually quite well to discredit the La table in the cases where La and Lu have about equal values. And especially look at the IE trends when Lu just falls clean off. With these four considerations – the clear beginning of f involvement at lanthanum, the clear lack of f involvement at lutetium, plus the way in which europium and ytterbium clearly emerge as having the half and fully filled f shells in an analogy to the d block, and the way lutetium is in every way a much better fit for the d block than lanthanum – the placement of Lu under Y falls out immediately.
So we continue to the end of the row with a triumphant return to the main-group elements.
Note that the metallicity line goes even further to the right. Not only bismuth, but also polonium and astatine, are pretty metallic. Even radon can form Rn2+ cations in halogen fluoride solution, and radon fluoride (RnF2) is actually an ionic compound! These elements have suffered both a 4f and a 5d contraction; why this? Relativity again: the 6p3/2 orbitals (two of them, being filled from bismuth to radon) are strongly destabilised and metallic bonding is favoured. (This isn't terribly well-known. In fact the usual treatment of astatine in most textbooks, as an exercise for people to practice extending periodicity down the halogens on, is another of those textbook errors like the treatment of hypervalence and, I claim, the whole group 3 argument. It has been long known since even its discovery in 1940 that astatine has distinct similarities to metals that to some extent outweigh its similarities to the halogens! It is just that astatine has a half-life of about eight hours and therefore nobody cares.) It is not surprising that the seventh row falls out in the same way...
Well, not quite. Relativity is causing some havoc, for Fr and Ra 6p3/2 may have some sort of bonding contribution. (Remember, 7s is stabilised and lowered in energy, 6p3/2 is destabilised and raised.) On the other hand, we can understand this whole thing as a trend. It should get more serious in the eighth row. Now, actinium and thorium are known to exhibit the same kind of 5f involvement in its compounds that lanthanum does. As usual, the weird gas-phase configurations don't matter.
At the very beginning, the 5f–6d difference is quite small. Not only that, but the collapse is quite a bit slower: it takes till Am or Cm to get the lanthanide-like situation. But then the collapse immediately becomes so fast that 5f for the latest actinides is more deeply buried than 4f for the latest lanthanides. This gives another analogy: 5f stands in relation to 4f more or less as 4d stands in relation to 3d. (The later shell has no radial node, for 5f it is also relativistically destabilised at the start, but at the same time when it does collapse it does so to a larger extent.) In trying to reflect the delayed 4f collapse, the La table fails to reflect the 5f one. Collapses don't happen at the same rate throughout the table! I argue it's more useful to just reflect groups of orbitals with about the same energy. Notice the beautiful honour Mendeleev was given, of becoming the namesake of the element with Z = 101. We may then proceed to a normal transition series:
Notice incidentally that if you do the same comparison chart for Ac vs Lr compared to what little we know or have predicted about the 6d metals, the difference becomes even starker.
And this time lawrencium's fit at the end of the 5f row is bad, but as seen above its fit at the start of the 6d row is actually quite good:
(Re melting points. The beginning dip is due to f electron hybridisation, as Mathias explained in what I quoted him above. After that you get a lanthanide-like map, which then drops down as the 5f-6d gap shrinks so much that the metals become divalent instead of trivalent, each contributing only two rather than three electrons to the metallic bonding. Same issue with density. Except for lawrencium which is trivalent again. The often-encountered value of 1800 K for fermium is wrong: see Silva's chapter on Fm through Lr in The Chemistry of the Actinide and Transactinide Elements.) Lawrencium is in fact a very odd late actinide, but it's a totally normal transition metal. Fm, Md, and No are easily reduced the lower oxidation state +2, Lr does no such thing. Instead Lr has complete stability of the group oxidation state and anything lower surely very easily oxidised, that's exactly like Rf, Db, Sg, probably also Bh. Es through No are not very dense thanks to it, Lr is normal density starting a trend up to Rf through Hs. Needless to say, relativity causes some havoc near the end of the row. Copernicium has the mercury effect on steroids, it becomes almost as inert as a noble gas, and isn't even metallically bonded! (On the other hand, once you force it to open up the 6d orbitals start going readily at chemistry; 6d is now higher in energy than 7s!) And the double periodicity in the 7p block because rather 2+4 rather than 3+3 elements. But worst of all, the 7p–8s gap lowers, and again the noble gas configuration is inviolable may not be true anymore. So actually the last six elements seem to go like this instead:
7s gets replaced by 8s at the end, and 7p1/2 becomes yet another inert pair. This is the first significant failure of the model due to relativistic effects (not quite the second-order failure of Fr and Ra, in which at least oxidation states past +1 and +2 seem inaccessible for all that the model wrongly predicts 6p3/2 won't be involved) – which is quite impressive considering that you have to wait till element 114 for it to happen. It's not as if the idea is happening for the first time, though. Already the 6s-6p gap is quite big as you may remember I said early in this, the 7s-7p gap is even bigger. Why can't 7p1/2-7p3/2 provide yet another gap, then? So let's just complete the row following the idea. It's impressive that we've gotten all the way to Z = 114 before finding our first exception. (NhF4− has 7s involvement in the bonding, as communicated to me by Droog Andrey.) The trend towards metallicity finally has proceeded to such an extent that even oganesson ought to be a metal. Its predicted chemistry is pretty close to that of tin. So, just like period 1 started out with no metals at all, period 7 ends up with everything a metal. Let's give the complete electronegativity table below, now with a pretty colour gradient, and pat ourselves on the back for making it all the way through.
The anomalously low figures for Mc through Og come from the preemptive shell closure at flerovium thanks to the relativistic splitting of the 7p subshell. And we'll complete this with a chart of chemically active aka valence subshells for each element.
s++ means "including at least some of the higher-l orbitals in the same row". Notice how I handle the Fl through Og problem. It's not perfect: I just treat the relativistic deviation as second order and ignore it for placements, largely because I am aware thanks to predictions that for period 8 there will be a surprising cancellation of them. The rightmost column shows which orbitals are being used. How simple, how logical, how global up to Z = 113. The last five elements go in the usual way for pragmatic reasons; they also continue the "inert pair" trend of Tl-Rn down into an "inert quartet" anyway, so nothing's that new. All the stuff I told you at the very basics back in part A has come back to weigh in on the question with a result of golden certainty: lutetium must go under yttrium, not lanthanum. If you want (and most chemists do) you can then move helium over to the noble gases to keep them together. After all, we did start from the noble gases way back in part A, they may as well stick with each other. Anyone can see however that that's a really exceptional one-off and won't demand any unwanted consequences. Unless you want to claim that any other vertically adjacent pair in the above table looks as odd as helium over beryllium, which is just plain at variance with reality.
Actually: maybe not quite all the way through. The heaviest elements past plutonium are too short-lived to occur in nature, we have to make them ourselves in laboratories. Although we should note that "too short-lived" is not what you might expect: a half-life of one second(!) is already enough to do chemistry, which gets us all the way up to element 115! And it's quite likely there are more long-lived forms of the heaviest elements that we haven't discovered yet. Not surprisingly, scientists are beginning with working on making element 119, and then plan to keep going up. It's hoped that we might reach element 126 by 2030, eight away from what we have now. Well, let's see how well the La argument of ground-state gas-phase configurations is going to deal with what our best theoreticians have predicted thus far for these eight new elements.
What a disaster. But maybe out of morbid curiosity we should take a look at when each of these blocks actually seem to finish: 5g at Z = 145, 6f at Z = 157, 7d at Z = 164, but 8p at Z = 172, long after 9s has already started at Z = 159. I do not want to contemplate what such a table is going to look like. That's why it's more pragmatic to just use valence subshells:
That works all the way till the predicted end of this eighth period at Z = 172.
The relativistic effects are readily handled by the idea that slowly 8s is drowned into the core over that huge 5g+6f series, and replaced by 9s for hybridisation. Same with 9p1/2 replacing 8p1/2. Meanwhile the idea of counting the number of valence electrons results in a recognition of the expected situation for the eighth-row elements 157 through 172 being excellent homologues of yttrium through xenon way back in the fifth row. The periodic law still manages to hold, keeping all the elements in order of increasing atomic number. With the next element past that, Z = 173, the "first electron" approach is going to have an aneurysm (if it didn't already) as a configuration of [172] 6g1 is expected. The chemistry is of course that of one of those furious alkali metals we started with. Horrific problem for the La approach. Simple smooth sailing for the Lu one, noting that (6g 7f 8d 10s 10p1/2) should be a set of orbitals of similar energy, and therefore plonking element 173 into the s-block. Which is just as well, as it would probably make caesium look like gold in its reactivity. It is probably just as well that we stop here. We have gotten to an element that is not likely to be synthesised for quite some time, and the calculations have stopped here. (Why did they even go up here in the first place? Because just like Z = 114 is a "magic number" of protons that closes a proton shell and gives more stability you'd think for these oversized nuclei, so is Z = 164. Yes, protons and neutrons arrange themselves in shells too, just like the electrons! But dealing with that would take us too far afield.)
Lanthanum and actinium can use their f orbitals for chemistry, just like the uncontroversial f block elements; lutetium and lawrencium cannot. Moreover, in every way lutetium and lawrencium act like normal early d block elements; lanthanum and actinium are outliers in the d block, but totally fit the trend of the early f block. It's really that simple. The same analogies are used everywhere. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Q23: Are there any other La arguments that you haven't refuted above?
Yes, but I guess going through all of them is not something most people reading this want to do. So here they are in a collapse box.
Those arguments
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
I have covered the biggest misunderstandings of the periodic law involved: the meaning of electrons configurations and double periodicity. So what's left to refute are the other ones.
Let's say you think that La should go under Y, in violation of the blocks that the modern periodic table is based on. The other precedent for such a thing is...helium going over neon instead of beryllium. Does anyone seriously think that this difference is of the same order? So, let's look for other differences. In fact chemically speaking aluminium is somewhat more similar to scandium than gallium. Again, that's because scandium doesn't suffer the 3d contraction that gallium does.
Does anyone move it there now? Basically no one. And in fact chemically speaking beryllium and magnesium are somewhat more similar to zinc than calcium. This used to be the standard placement, in fact. Again, these similarities are because beryllium and magnesium are small, zinc has suffered the 3d contraction that calcium hasn't, therefore of course size makes Be and Mg more similar to Zn.
Does anyone move them there now? Basically no one. So let's ask: are the increased resemblances of lanthanum to yttrium, over lutetium to yttrium, bigger than the Al and Mg issues? But there is no such increased resemblance! As shown above, lutetium is far more like yttrium than lanthanum is. Lutetium is perfectly fine with the d elements; lanthanum in every single way is an outlier for the d elements. But lanthanum fits perfectly fine in the f elements. For actinium this is even more obvious. And for lawrencium, too, because lawrencium is a terrible outlier for the late f elements. So where is the consistency of doing this for La, and not doing it for the far more worthy case of Al over Sc?
Sandbh seems to believe in the importance of the "differentiating electron" of an element, which is the electron that differentiates it in its ground-state gas-phase configuration for the previous element. For example, since titanium is [Ar]3d24s2 (ground-state gas-phase), and vanadium is [Ar]3d34s2 (same), vanadium has a 3d differentiating electron. I do not see why this should be important, given that we are not generally in the business of building elements up by adding one proton and one electron from the previous one. Also differentiating electrons are for the gas phase, and we've already discussed above that this is not relevant for chemistry because atoms usually have a different configuration when chemically bound from what they have sitting alone by themselves. Neither is it that clear all the time what exactly the differentiating electron ought to be. Passing from vanadium [Ar]3d34s2 to chromium [Ar]3d54s1 we have added two 3d electrons and subtracted one 4s one. But all right, suppose we grant that. A usual argument Sandbh gives for why this should supposedly be important is looking at silver. It has a 5s differentiating electron as opposed to the 5d one of its heavier homologue gold, and this supposedly explains why the chemistry of silver is more main-group-like than gold:
On the other hand, technetium also has a 5s differentiating electron as opposed to the 5d one of its heavier homologue rhenium. Judging by how happy technetium is to interconvert between its many oxidation states like the transition metal it really is, this does not seem to do anything at all to make it more main-group-like at all.
If you look at KCl, CaCl2, ScCl3, and TiCl4, the first three are ionic solids and the last one is a volatile liquid. So group 3 has to be cleft from group 4. I used to believe this, but it doesn't really work. We may equally well observe the melting points of Cs2O, BaO, La2O3 or Lu2O3, HfO2, Ta2O5, WO3, Re2O7, OsO4. These are all high melting-point compounds until WO3, and the melting points keep increasing (showing ionic character) until HfO2. And we can multiply other funny examples like Na2O, MgO, Al2O3, SiO2 (melting points increase till MgO). Any way you look at it, this doesn't support any natural group divide at all. Mostly because there is no such thing outside the fundamental one (noble gas | alkali metal).
According to this argument, group 3 is more similar to group 2 than group 4 because group 3 acts like a trivalent version of the main group metals. Whereas for group 4 you cannot find aqueous cations. The problem with that is that for group 4 you can find aqueous cations. Simply make the pH low enough. −1 will do, you'll easily find things like Zr4+ and Hf4+, maybe even Ti4+, then. Well, look at Fajans' rules again. Cation polarising power, hence acidity, depends on charge and radius. Following Gary Wulfsberg's Principles of Descriptive Inorganic Chemistry, the usual dependence is on charge squared divided by ionic radius. We can then draw his table:
And indeed, we find that while the group 4 cations are indeed strongly acidic, they are not so acidic that they react irreversibly with water: the pKa is not quite low enough. Sure, you'll soon get a precipitate if their cations are introduced to aqueous solution at all but pretty low pH. But you can redissolve that precipitate in concentrated hydrochloric acid, at least if it hasn't aged too much. It's cations like Nb5+ and Ta5+ that are "very strongly acidic": too acidic to exist in water at all. So, apparently the group divide is really between groups 4 and 5 instead. But probably the clearest way to refute this whole idea is to look at what happens in the p block. Group 13 metals Ga, In, Tl can form aqueous cations in their group oxidation state. Group 14 metals Ge, Sn, Pb cannot, they are too acidic. (Remember what I said before? Elements in higher oxidation states are more electronegative and hence more acidic, whence how Pd2+ is more acidic than Be2+.) So this seems to also support B-Al-Sc if you apply it consistently. I think that is not a good sign.
If you graph the trends for Sc-Y-La-Ac, they usually follow the approximately linear trend of Ca-Sr-Ba-Ra. Whereas Sc-Y-Lu-Lr usually follows the "kinked" trend of Ti-Zr-Hf-Rf and the following d block groups. But the above has already explained why the s block trend in the table is special. It never suffers any incomplete shielding effects because it comes straight after the previous noble gas. So, all the odd-even periodicity is completely absent from it. There's no reason to expect any other group to do the same thing. Group 3 ought to follow its fellow d block groups 4 through 12. Also, the chemistry of group 3 is not more similar to that of group 2 to group 4, so that justification is also out. Observe the aqueous chemistry of group 4 and the organometallic chemistry of group 3. Not to mention that the standard "transition metal" property everyone knows from high school is forming coloured paramagnetic compounds with incomplete d orbitals. If you think scandium should be disqualified, because you will have a tough time finding stable lower-oxidation state compounds of Zr-Hf-Rf in group 4 and Nb-Ta-Db in group 5, those suddenly are also in danger of being disqualified. And as for the other s-block property of forming hard, class-A cations – the elements up to group 6 show that. Everything is a continuum here, and transition properties are no exception: they go in slowly, they come out slowly. All peripheral groups in blocks have properties similar to their block neighbours, there's no reason why to emphasise the property this much for group 3. The idea of keeping "s-like" elements together by chasing group 3 away from group 4 overlooks the also rather s-like group 4 elements (which form aqueous cations and are not that different from thorium; just look how hydrolysed Th4+ really will be in aqueous solution), and seems to forget that there exist f-block elements, which unite in themselves the hardness of the s elements with the multiple oxidation states of the d elements.
According to this argument, lanthanum cannot start the f block because La3+ fails to have an f electron, and there is complete regularity down Ce3+ to Lu3+ in showing f1 to f14. But such a common stable oxidation state for everybody only exists in the f block, and actually only the 4f elements, not the 5f ones. So this argument is about as local as you can get and doesn't work as part of the periodic law. I also note that by this logic La cannot be in the d block either, since La3+ is d0. In fact it cannot be anywhere in the periodic table either, since La3+ is also s0 and p0. And there are no s block elements either except helium (the least s-block-like of them), because in their common oxidation states (+1 for the column H-Li-Na-K-Rb-Cs-Fr, +2 for Be-Mg-Ca-Sr-Ba-Ra) they are all s0. Why should we use criteria that only apply to one block? Finally, La had been [Xe]4f16s2, all this argument would surely never have started: a La-Yb f block (i.e. a Lu table) would be absolutely standard. But it would still lose the f electron in the +3 state anyway, so this argument would still argue for Ce-Lu anyway. That is hard to accept.
Sandbh has a rule for block start which involves when the first electron for that block appears in the gas-phase (i.e. cerium for f). But thorium is a problem with that as shown above.
According to this argument, advanced by Lavelle, La and Ac cannot be put in the f block because they would represent the only case of two elements going into a block with no outer electrons in common with their block. Of course, this is predicated on the gas-phase ground-state configurations. We have already demonstrated why this is not that relevant. Along with copious quotes demonstrating that chemists understand that. How does this deal with Lu and Lr? Neither of them have any outer f electrons, they also form a pair out of place. So the argument is as much for the Lu table as it is for the La table. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Q24: Weren't you on the other side of this last time?
Indeed. I learnt more, I changed my mind. All thanks to Droog Andrey (= A. V. Kulsha). You may enjoy the periodic table poster he and one of his colleagues created too (link is to version of 2019). ^_^
Q25: So what's wrong with the old arguments that you were using then?
I've addressed them all above in the collapse boxes.
Questions 26 to 30
Q26: So how do I know you won't be fervently arguing to change it back again in four years?
I don't, that's because I believe in the scientific method. If I find out something falsifying my stand, I change my mind. You can argue it if you want... ;)
Q27: Why now? Why not wait for IUPAC?
It's not possible to be neutral. Sc-Y-* is also a side. I also think Sc-Y-* is chemically just weird (those f electrons are not core electrons, and while asking La-Lu to share one flat makes some sense, it's really weird for Ac-Lr), and Jensen agrees.
Q28: Why start it now and not earlier?
Because my ideas about this were not clear enough till this round of argument with Sandbh started. Now they are.
Q29: Do you have nothing better to do than write long screeds here about this?
Well, at R8R's suggestion it is now behind a collapse box, and most of the long screeds have been replaced by very short answers.
Q30: Do you have anything against Sandbh personally?
No, I just find myself in disagreement with his stand on logic and falsifiability.
Questions 31 and 32
These can be skipped if you don't read the long answer to Q22.
Extra questions regarding my Q22 approach
|
|---|
|
Q31: You seem to think helium should go over beryllium rather than neon, and put it there in your explanation above. Is that a requirement for your bottom-up approach to the periodic table? No, because no other element looks that badly placed from the chemistry angle. Therefore one can very readily elect to shift helium over to the noble gases as is standardly done, without forcing any unwanted consequences as the Sc-Y-La table does (because resemblances Al-Sc over Al-Ga are actually better, so if you claim Y-La over Y-Lu is better, which it is not by chemical and physical properties, you have a bit of a consistency problem). It is only that if I am trying to teach it from the very beginning I would rather not deal with this exception at the very start. Q32: Granting that you don't propose it yet, is it inconsistent to use electron configurations to support He over Be on the grounds of s2, and oppose La under Y even though both are ds2? My sympathy to helium over beryllium is not the main issue here. I don't propose to change any defaults about that. But if you're asking: no, the situation is a bit different. I focus on valence orbitals, not actual configurations. Y has valence orbitals 4d5s5p, Lu has 5d6s6p, they fit together; La has 4f5d6s6p, which doesn't fit. Helium 1s vs neon 2s2p is more of an issue. Having said that, I think we can clearly see that helium is so weird over beryllium that no consistency problems result from moving it. |
Mass summoning
@ComplexRational, DePiep, Droog Andrey, Officer781, R8R, Sandbh, Дрейгорич, AzaToth, Tazerdadog, Maproom, CuriousMind01, Silvio1973, and Timtempleton: Pinging all megathread and previous RFC participants. And also AnthonyDu0122 who raised it above. ^_^
Unacceptable behaviour
I object, in the strongest possible terms, to the way Double sharp initiated this RFC.
I allege that Double sharp's background to the RFC is biased; incomplete; out-of-context; contains inappropriate content; and—at 33,000 words—is too long for anyone to gain an informed understanding of the issue at hand. This follows other unacceptable behaviour alluded to on his part, including his hack work on our periodic table article; removing some of my citation supported content; slandering me; swearing; and effectively demanding I provide a falsifiable hypothesis when I was under no obligation to do so.
The context for my objection is my long-term participation in the Group 3 debate, including two submissions to IUPAC on the matter, one with Double sharp (which he has now dissociated himself from).
My other objection is that an article written by me will soon appear in the journal Foundations of Chemistry, entitled "The location and composition of Group 3 of the periodic table". I have the proofs in hand. The article has been reviewed by three PhDs and accepted for publication by Eric Scerri, a world authority of the periodic table. Scerri is also the chair of the IUPAC Group 3 project.
Double sharp knows all this, yet chooses to go ahead with the RFC. So much for taking into account "reliable sources that focus on the matter." So much for extending me the courtesy of professional respect. So much for working collegiately within WP:ELEMENTS.
Here again is a continuation of his pattern of unacceptable behaviour.
Most of the rest of the editors who have voiced their opinions for Lu, it seems to me, do so on the basis of a personal preference or a very limited grasp of the full extent of the arguments and literature involved in this case.
I'm not holier than anyone. I do claim, along with Double sharp, to have maintained a depth of familiarity and interest in this question since at least 2016. In my case I've been arguing the question with Scerri, on and off, since 2008. And along the way, Double sharp and I have changed our minds about Group 3 as we both learnt new things along the way.
As Jones (2010) has written:
- "Scientists should not lose sleep over the hard cases. As long as a classification system is beneficial to economy of description, to structuring knowledge and to our understanding, and hard cases constitute a small minority, then keep it. If the system becomes less than useful, then scrap it and replace it with a system based on different shared characteristics."
- Jones 2010, Pluto: Sentinel of the outer solar system, Oxford University Press, p. 171
As Schwerdtfeger, Smits & Pyykkö (2020) wrote:
- "Fuzzy concepts like chemical similarity often lead to unnecessary disputes concerning the PTE."
- Schwerdtfeger P, Smits OR & Pyykkö P, 2020, "The periodic table and the physics that drives it". Nature Reviews Chemistry, vol. 4, pp. 359–380
That is why we have an IUPAC Group 3 project team, to assess the literature and address these unnecessary disputes.
And the IUPAC project team has reported that, based on a survey of chemistry textbooks and other sources, that the La form dominates by a 4:1:1 margin, where the two 1's are the *-** form and the Lu form.
As Scerri wrote:
- "…the majority of textbook and other periodic tables in the 18-column format show the elements of this group as scandium, yttrium, lanthanum and actinium…"
- Scerri ER 2020, "Recent attempts to change the periodic table", Philosophical Transactions A, here
As WP:Elements member R8R wrote:
- "…I'll say that the 4:1:1 consideration will be very important for the upcoming RfC, especially given that it's something that can be found on IUPAC's official website. I agree that consistency with literature is of the uttermost importance for Wikipedia if literature itself goes shows such a consistency; Wikipedia is meant to be a tertiary source. It'll be the thing that will hold me from supporting the motion on WP right now…--R8R (talk) 16:51, 19 July 2020 (UTC)"
Precisely.
The way ahead, as I see it, is (1) cancel the RFC until my article appears on line; and (2) Double sharp and I to agree on a balanced introduction to a new RFC, should Double sharp wish to proceed with another RFC. Failing this I will take up the matter on WP:AN. --- Sandbh (talk) 03:27, 21 July 2020 (UTC)
Discussion
- Strong Support for change to Lu form, per what I wrote above. Double sharp (talk) 10:08, 20 July 2020 (UTC)
- Also, per Michael D. Turnbull, below: I strongly support that periodic table article should show both forms and briefly discuss the difference. When I suggest changing to the Lu form, it is in the context where we simply show the periodic table as a template, e.g. for navigation like the infobox on neon or other elements. Double sharp (talk) 12:13, 20 July 2020 (UTC)
- Comment I think the reader would be aided massively if there were a one-line response to each question and the rest of the answer were hidden in a collapsed (but uncollapsible) box.--R8R (talk) 10:30, 20 July 2020 (UTC)
- @R8R: You raise a good point. I'll do it soon. Double sharp (talk) 11:22, 20 July 2020 (UTC)
- @R8R:
 Done Double sharp (talk) 12:40, 20 July 2020 (UTC)
Done Double sharp (talk) 12:40, 20 July 2020 (UTC)
- @R8R:
- @R8R: You raise a good point. I'll do it soon. Double sharp (talk) 11:22, 20 July 2020 (UTC)
- I will assume that I do not need to read this giant thing before giving my input? < Atom (Anomalies) 11:33, 20 July 2020 (UTC)
- @AnomalousAtom: I am working on adding one-line responses and collapsing the big ones per R8R's suggestion. In the meantime there is a summary. Double sharp (talk) 12:13, 20 July 2020 (UTC)
- @AnomalousAtom: It is now significantly less giant since all the detail is now in collapse boxes. Double sharp (talk) 12:40, 20 July 2020 (UTC)
- @AnomalousAtom: I am working on adding one-line responses and collapsing the big ones per R8R's suggestion. In the meantime there is a summary. Double sharp (talk) 12:13, 20 July 2020 (UTC)
- I will assume that I do not need to read this giant thing before giving my input? < Atom (Anomalies) 11:33, 20 July 2020 (UTC)
- Neutral on the change but Strong support that the periodic table article should show both forms and give some (brief!) discussion of why some reliable sources use La and others Lu. There is no such thing as the periodic table, it has been presented in numerous forms over the years and Wikipedia should reflect that. However there is only one periodic table article and we need to put a summary there reflecting WP:NPOV on the various forms. Michael D. Turnbull (talk) 11:37, 20 July 2020 (UTC)
- Now Against the change after the useful clarification that we are really only talking about the appearance of Wikipedia templates like
{{Periodic table}}(See top of this RFC thread). We should stick to the same version as IUPAC currently endorse, i.e. IUPAC (2018-12-01). "PERIODIC TABLE OF ELEMENTS". since that's what most readers would expect. (Even British chemists don't use sulphur now, only sulfur.) Michael D. Turnbull (talk) 13:49, 20 July 2020 (UTC)- Actually it is not quite true that IUPAC endorses the form. They write on their website "While IUPAC has no recommendation for a specific form of the periodic table, i.e. 18-column or 32-column format, the version here presented is in the conventional long form and is yours to use."
- Now I can see the argument for using * under Y as that's what IUPAC shows on their webpage, even if they seem to be planning to change it away from * under Y eventually. But it does make things awkward for the 32-column forms encountered in
{{Compact periodic table}}etc. Will scandium and yttrium have to be stretched over 15 columns? The only way it becomes less awkward is if we change those to the 18-column form too. Which is a bit odd as the 18 and 32 column forms are supposed to be saying the same thing in a different layout. Double sharp (talk) 13:56, 20 July 2020 (UTC)
- Strong support on changing to Lu as per arguments above. I was involved in the discussion itself as a minor participant. ― Дрейгорич / Dreigorich Talk 12:29, 20 July 2020 (UTC)
- Support change to Lu per arguments above; I also was a minor participant in the group 3 megathread. I would also like to comment that the IUPAC standard is intended to be deprecated (
that is why this RfC does not consider Sc-Y-*-*;(not originally considered here but later added) it is possibly even more incorrect to place all 30 inner transition metals below Sc and Y, and it still doesn't address how to structure the 32-column table), and the remaining arguments strongly suggest that Lu is the better choice. ComplexRational (talk) 14:11, 20 July 2020 (UTC)- @ComplexRational: BTW, I just added Sc-Y-*-** as an option since Michael D. Turnbull is in favour of it. So now it is not accurate anymore that the RFC does not consider the option. It is however correct that I am against that option. ^_^ Double sharp (talk) 14:18, 20 July 2020 (UTC)