→Potential negative effects: rmv pasted in Crutzen quote - source not provided |
William M. Connolley (talk | contribs) →Indirect geoengineering: rm section: nothing to do with strat; gross OR |
||
| Line 114: | Line 114: | ||
*manipulation of natural process to indirectly induce greater abundance of particles |
*manipulation of natural process to indirectly induce greater abundance of particles |
||
====Indirect geoengineering==== |
|||
Enhancing the natural [[sulfur cycle]] in the [[Southern Ocean]] by fertilising a small portion with [[iron]] in order to enhance [[dimethyl sulfide]] production and cloud [[reflectivity]]. The goal is to slow [[Antarctic]] ice from melting and raising [[sea level]]<ref>http://www.climos.com/news/articles/slowingglobal.htm [[Oliver W. Wingenter]]</ref><ref>http://adsabs.harvard.edu/abs/2002AGUFMOS22D..01C</ref> Such techniques also tend to sequester carbon, but in this specific project the enhancement of cloud albedo was both the desired outcome and measured result.<ref>http://www.pmel.noaa.gov/pubs/outstand/bate1229/estimate.shtml</ref> The natural [[sulfur cycle]] has a significant effect on climate.<ref>http://ocw.mit.edu/NR/rdonlyres/Earth--Atmospheric--and-Planetary-Sciences/12-491Fall-2007/8379BCC7-1A25-441F-83FF-99F1F4A3143A/0/clayton.pdf</ref> The [[CLAW hypothesis]] suggests that this effect is not only effective as a [[steady-state]] phenomenon, but that it is also significant contributor to a variable [[feedback]] process which helps to stabilise the climate. It is notable that the processes behind the CLAW hypothesis may be disrupted by climate change, leading to the creation of a new and destructive process (the anti-CLAW hypothesis). |
|||
By altering this cycle, such as through ocean [[iron fertilization]] (as in the SOFEX experiments run by Climos,<ref>http://www.climos.com/news/articles/slowingglobal.htm</ref>) it may be possible to exercise indirect control over stratospheric sulfur aerosols.<ref>http://www.cosis.net/abstracts/EGU2008/12210/EGU2008-A-12210.pdf</ref> However, the main effect envisaged is a change in the reflectivity of clouds due to the sea-level height of gas emissions. However, stratospheric-tropospheric exchange processes<ref>http://www.agu.org/pubs/crossref/1975/RG013i004p00459.shtml</ref> suggest that transfer of particles and precursor gases is possible,<ref>http://www.springerlink.com/content/p0778612j2w88p7h/</ref><ref>http://www.agu.org/pubs/crossref/1994/93JD02179.shtml</ref> although further research is needed. |
|||
====Direct geoengineering==== |
====Direct geoengineering==== |
||
Revision as of 20:19, 31 January 2009
Stratospheric sulfur aerosols are tiny sulfur-rich particles of solid or liquid, or a mixture of the two, which exist in the stratosphere region of the Earth's atmosphere. They produce a significant global cooling effect[1] at these altitudes by reflecting sunlight, and as they fall out of the stratosphere, by modifying clouds.[2]
An aerosol is a suspension of fine solid particles or liquid droplets in a gas. The sulfate particles in the atmosphere are about 0.1 to 1.0 micrometer (a millionth of a meter) in diameter.
Sulfur aerosols are common in the troposphere as a result of pollution with sulfur dioxide from burning coal, and from natural processes. Volcanos are a major source of particles in the stratosphere as the force of the volcanic eruption propels sulfur-containing gases into the stratosphere.
Creating stratospheric sulfur aerosols deliberately is a proposed geoengineering technique which offers a possible solution to some of the problems caused by global warming whilst causing problems of its own.[citation needed]

Origins

Natural sulfur aerosols are formed in vast quantities from the SO2 ejected by volcanoes,[3] which are often injected directly into the stratosphere during eruptions. They can also form in the troposphere from volatile sulfur compounds such as dimethyl sulfide from oceans and from H2S emitted by marshes, then be carried aloft by tropospheric-stratospheric exchange. In addition terrestrial vegetation and the erosion of sulfurous rocks are all significant sources. A comprehensive analysis of sources of of sulfur sources in the atmosphere is provided by Bates et al.[4]
Man made emissions of SO2 from the burning of fossil fuels (notably coal) are also abundant in the atmosphere. Aerosols from anthropogenic sources are more abundant in the troposphere (which is the lower atmospheric region, into which the particles are typically emitted) than in the stratosphere.
Chemistry
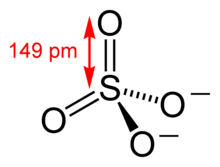
Sulfur aerosols are those containing sulfate salts or sulfuric acid. The sulfate ion is a polyatomic anion with the empirical formula SO42− and a molecular mass of 96.06 daltons; it consists of a central sulfur atom surrounded by four equivalent oxygen atoms in a tetrahedral arrangement. The sulfate ion carries a negative two charge and is the conjugate base of the bisulfate (or hydrogen sulfate) ion, HSO4−, which is the conjugate base of H2SO4, sulfuric acid. Organic sulfates, such as dimethyl sulfate, are covalent compounds and esters of sulfuric acid.
Except in the case of wind-blow rock dust, these sulfate ions are generally produced by the oxidisation of gaseous or aqueous sulfur dioxide to form sulfate ions. The SO2 precursor may be emitted directly or formed from a number of precursor gases. Sulfur dioxide, SO2, is the most abundant anthropogenic and volcanically emitted sulfur-containing gas. Most of the SO2 emissions globally result from fossil fuel burning. For this reason, most of the aerosol produced from the oxidation of SO2 is considered to be anthropogenic sulfate aerosol (ASA). The source strengths for this trace gas are fairly well known compared to other aerosol precursor gases, and recent estimates differ by no more than ~20-30%.[5]
Sulfur dioxide precipitates out of the troposphere as dry deposition, or as acid rain, limiting its contribution to stratospheric aerosol formation. By contrast, other chemicals such as OCS do not deposit in this manner and thus can be, at various times and places, more significant than the more abundant SO2 for stratospheric aerosol formation.[6]
Atmospheric SO2 may be created by the oxidation of precursor gases including:
- Hydrogen sulfide (H2S)
- Carbonyl sulfide (OCS) The reactivity of COS in the troposphere is low, allowing COS to enter the stratosphere by diffusion. During periods lacking volcanic activity (and thus direct injection of SO2 into the stratosphere), oxidation of COS dominates the production of stratospheric sulfate aerosol.
- Methanethiol (CHSH)
- Dimethyl-sulfide (CHSCH or DMS), which is the most abundant of the biologically-emitted gases, and its precursor dimethylsulfoniopropionate (DMSP).
- Dimethyldisulfide (CHSSCH)
- Carbon disulfide (CS2)
Most of the above are biological in origin, with the significant exception of the proportion of H2S from volcanoes and other geological sources, such as geysers.[7] Significant anthropogenic sources of H2S also exist, notably vehicle exhausts treated by catalytic converters.[8] Bates, et. al. discuss the origins and processes behind a wide variety of precursor gases.[9]
"SO2 must be oxidized to SO4-2 (sulfate) before it plays a role in aerosol formation. The oxidation can take place while SO2 is still in the gas phase, or after SO2 becomes dissolved in cloud droplets (aqueous production). In the latter case, the sulfate becomes aerosol after the cloud droplets evaporate. Both pathways produce sulfate aerosols in the submicrometre size range that are efficient light scatterers. Positively charged ions must be present on sulfate aerosols to neutralize the sulfate anions. Thus, the sulfate in aerosol particles is present as sulfuric acid (H2SO4), ammonium sulfate (NH4)2SO4, or intermediate compounds, depending on the availability of gaseous ammonia to neutralize the sulfuric acid formed from SO2."[10]
The chemistry of stratospheric sulfur aerosols varies significantly according to their source. Volcanic emissions vary significantly in composition, and have complex chemistry due to the presence of ash particulates and a wide variety of other elements in the plume.[11]
"Sulfate is known to condense onto larger aerosol particles with lower scattering efficiencies and shorter atmospheric lifetimes. At least two pathways for this interaction are recognized. The first is heterogeneous reactions of SO2 on mineral aerosols. The second is oxidation of SO2 to sulfate in sea salt-containing cloud droplets and deliquesced sea salt aerosols. This process can result in a substantial fraction of non-sea-salt sulfate to be present on large sea salt particles, especially under conditions where the rate of photochemical H2SO4 production is low and the amount of sea salt aerosol surface available is high. The calculated residence times of SO2, defined as the global burden divided by the global emission flux, range between 0.6 and 2.1 days as a result of different deposition parameterizations. Because of losses due to SO2 deposition, only 46-82% of the SO2 emitted undergoes chemical transformations and forms sulfate. The residence time of sulfate is mainly determined by wet removal and is estimated to be between 2.7 and 7.2 days."[12]
The chemical reactions affecting both the formation and elimination of sulfur aerosols are not fully understood. It is difficult to estimate accurately, for example, whether the presence of ash and water vapour is important for aerosol formation from volcanic products, and whether high or low atmospheric concentrations of precursor chemicals (such as SO2 and H2S) are optimal for aerosol formation. This uncertainty makes it difficult to determine a viable approach for geoengineering uses of sulfur aerosol formation.
Transport
Most volatile sulfur compounds are released at or near ground or sea level, into the base of the troposphere. Volcanoes are distinct, as their eruptions of hot gases are able to force volatile sulfur compounds and sulfur-containing particulate matter directly into the stratosphere. The two layers of the atmosphere are not isolated, and are affected by a process of stratospheric-tropospheric exchange.[13]
Scientific study
Understanding of these aerosols comes in large part from the study of volcanic eruptions,[14] notably Mount Pinatubo in the Phillipines[15], which erupted in 1991 when scientific techniques were sufficiently far advanced to study the effects carefully.[16]
The formation of the aerosols and their effects on the atmosphere can also be studied in the lab. Samples of actual particles can be recovered from the stratosphere using balloons or aircraft.[17]
Computer models can be used to understand the behaviour of aerosol particles, and are particularly useful in modelling their effect on global climate.[18]
Biological experiments in the lab, and field/ocean measurements can establish the formation mechanisms of biologically-derived volatile sulfurous gases.[19]
Effects
Effect on climate
It has been established that emission of precursor gases for sulfur aerosols is the principle mechanism by which volcanoes cause global cooling.[20]
The Intergovernmental Panel on Climate Change, IPCC, says:
- "the cooling effect of sulfur emissions may have offset a significant part of the greenhouse warning in the northern hemisphere during the past several decades."[21]
- While the radiative forcing due to greenhouse gases may be determined to a reasonably high degree of accuracy... the uncertainties relating to aerosol radiative forcings remain large, and rely to a large extent on the estimates from global modelling studies that are difficult to verify at the present time [1].
The aerosol particles form a bluish[22] or whitish[23] haze in the sky. This creates a global dimming effect, where less of the sun's radiation is able to reach the surface of the Earth. This leads to a global cooling effect. In essence, they act as the reverse of a greenhouse gas, which tends to allow visible light from the sun through, whilst blocking infra-red emitted from the Earth's surface and its atmosphere. The particles also radiate infra red energy directly, as they lose heat into space.
Effects on light transmission through the atmosphere

All aerosols both absorb and scatter solar and terrestrial radiation. This is quantified in the Single Scattering Albedo (SSA), the ratio of scattering alone to scattering plus absorption (extinction) of radiation by a particle. The SSA tends to unity if scattering dominates, with relatively little absorption, and decreases as absorption increases, becoming zero for infinite absorption. For example, sea-salt aerosol has an SSA of 1, as a sea-salt particle only scatters, whereas soot has an SSA of 0.23, showing that it is a major atmospheric aerosol absorber.
Aerosols, natural and anthropogenic, can affect the climate by changing the way radiation is transmitted through the atmosphere. Direct observations of the effects of aerosols are quite limited so any attempt to estimate their global effect necessarily involves the use of computer models. The Intergovernmental Panel on Climate Change, IPCC, says: While the radiative forcing due to greenhouse gases may be determined to a reasonably high degree of accuracy... the uncertainties relating to aerosol radiative forcings remain large, and rely to a large extent on the estimates from global modelling studies that are difficult to verify at the present time [2].
The main direct effect of sulfates on the climate involves the scattering of light, effectively increasing the Earth's albedo. This effect is moderately well understood and leads to a cooling from the negative radiative forcing of about 0.5 W/m2 relative to pre-industrial values,[24] partially offsetting the larger (about 2.4 W/m2) warming effect of greenhouse gases. The effect is strongly spatially non-uniform, being largest downstream of large industrial areas.
Effect on clouds
Aerosols particles affect the nucleation of water droplets which form clouds.[25][26] However, the stratosphere is very dry; unlike the troposphere, it rarely allows clouds to form.[27]
The nucleation effect of sulfur aerosols in the stratosphere may be significant in the formation of polar stratospheric clouds, which govern the principle mechanisms for ozone depletion.
Although clouds are more abundant in the troposphere than in the stratosphere, the aerosol particles are influential on clouds during their ultimate descent through the troposphere. Depending on the source of the aerosols, they are also potentially significant during their ascent. Sulfate aerosols can act as cloud condensation nuclei (known as the Twomey effect) and this leads to greater numbers of smaller droplets of water. Lots of smaller droplets can diffuse light more efficiently than just a few larger droplets.
Further knock-on effects come from having more cloud condensation nuclei. It is proposed that these include the suppression of drizzle, increased cloud height, [28] to facilitate cloud formation at low humidities and longer cloud lifetime.[29] Sulfate may also result in changes in the particle size distribution, which can affect the clouds radiative properties in ways that are not fully understood. Chemical effects such as the dissolution of soluble gases and slightly soluble substances, surface tension depression by organic substances and accommodation coefficient changes are also included in the second indirect effect.[30]
The indirect effects probably have a cooling effect, perhaps up to 2 W/m2, although the uncertainty is very large. Sulfates are therefore implicated in global dimming, which may have acted to offset some of the effects of global warming. Further research in this area is needed, especially if manipulation of sulfur aerosols for geoengineering is intended.
Effect on ozone
The aerosols have a role in the destruction of ozone[31] due to surface chemistry effects.[32] Destruction of ozone has in recent years created large holes in the ozone layer, initially over the Antarctic and then the Arctic. These holes in the ozone layer have the potential expand to cover inhabited and vegetative regions of the planet, leading to catastrophic environmental damage.
Ozone destruction occurs principally in polar regions,[33] but the formation of ozone occurs principally in the tropics.[34] Ozone is distributed around the planet by the Brewer-Dobson circulation.[35] Therefore, the source and dispersal pattern of aerosols is critical in understanding their effect on the ozone layer.
Effect on sky appearance

Aerosols scatter light, which affects the appearance of the sky and of sunsets. Changing the concentration of aerosols in the atmosphere can dramatically affect the appearance of sunsets. A change in sky appearance during the year without a summer (attributed to the eruption of Tambora) was the inspiration for the paintings of J. M. W. Turner. Further volcanic eruptions and geoengineering projects involving sulfur aerosols are likely to affect the appearance of sunsets significantly,[36] and to create a haze in the sky.
Effect on the biosphere
Aerosol particles are eventually deposited from the stratosphere onto land and ocean. Depending on the volume of particles descending, the effects may be significant to ecosystems, or may not be. Modelling of the quantities of aerosols used in likely geoengineering scenarios suggest that effects on terrestrial ecosystems from deposition is not likely to be significantly harmful.[37][38]
Geoengineering
The ability of stratospheric sulfur aerosols to create this global dimming effect has made them a possible candidate for use in geoengineering projects[39] to limit the effect and impact of climate change due to rising levels of carbon dioxide, methane and other greenhouse gases.[40] The appeal of this technique is partly based on the fact that it is the adaptation of an existing atmospheric process, the behaviour of which is relatively well understood. It is also partly based on the great speed with which an effective solution could be delivered, as existing technology is essentially sufficient to implement a rapid geoengineering 'fix' based on stratospheric sulfur aerosols for many of the problems posed by global warming. However, certain gaps in understanding of these processes exist (e.g. the effect on stratospheric climate), and further research is needed.[41]
Tom Wigley calculates the impact of injecting sulfate particles, or aerosols, every one to four years into the stratosphere in amounts equal to those lofted by the volcanic eruption of Mount Pinatubo in 1991. If found to be environmentally and technologically viable, such injections could provide a "grace period" of up to 20 years before major cutbacks in greenhouse gas emissions would be required, he concludes.[42]
There are two main methods for enhancing aerosol formation:
- direct deposition of particulates or precursors into the stratosphere
- manipulation of natural process to indirectly induce greater abundance of particles
Direct geoengineering

Direct delivery of precursors, proposed by Paul Crutzen.[43] This would typically be achieved using H2S or SO2, delivered using artillery, aircraft (such as the high-flying F15-C) or balloons.[44][45][46][47] H2S is preferable due to its lower molecular weight, despite both molecules containing a single sulfur atom.
Delivery methods
Various techniques have been proposed for delivering the aerosol precursor gases (H2S and SO2).
- Aircraft such as the F15-C variant of the F-15 Eagle have the necessary ceiling, but limited payload. Transport aircraft and bombers are not able to reach the necessary altitude,[48] although some aircraft exist which could be developed or adapted.
- Artillery has the necessary capability. Old-fashioned naval guns have very large calibres ideally suited to large payload lifting. For example, the 460-millimetre (18 in) guns used on the World War 2 Japanese Yamato class battleships fired a 1,460 kilogram (3,219 lb) projectile to a maximum range of 42,000 metres (45,930 yards) (26.1 miles). This far exceeds both the range and payload necessary for aerosol precursor delivery.
- Railguns or coilguns can be used for regular deliveries, as they do not require a polluting and expensive cordite charge to loft the payload. The more gradual acceleration also allows more sophisticated shells to be used, which may incorporate gliders, parachutes, etc. that allow re-use or recovery of projectiles. There are currently no artillery pieces based on this principle, although there has not been a pressing need to develop the technology to date, due to the existence of suitable chemical propellants for military artillery.

A multistage coilgun could launch aerosol shells without the need for chemical propellants. - High-altitude balloons can be used to lift tanks containing precursor gases, or these gases can be contained within the balloon itself an then release, along with the hydrogen, when the balloon reaches a sufficient altitude.
- A daisy chain of moored balloons could be used to lift gas tanks, or a hosepipe which would then be used to pump the precursor gases into the stratosphere. No tethered balloon has yet reached the necessary heights, and the technology requires significant development to be able to deploy, control and use such a long hose from the daisy chain.

Moored balloons could be adapted to carry hoses or gas bladders to the stratosphere.
Efficacy problems
All geoengineering schemes have potential efficacy problems, due to the difficulty of modelling their impact and the inherently complex nature of the global climate system. However, certain efficacy issues are specific to the use of this particular technique.
- Lifespan of aerosols: Tropospheric sulfur aerosols are very short lived.[49]Delivery of particles into the lower stratosphere will only ensure they remain aloft for typically a few weeks or months.[50] To ensure endurance, high-level delivery is needed, ensuring a typical endurance of several years. Further, sizing of particles is crucial to their endurance.[51]
- Aerosol delivery: Even discounting the challenges of lifting, there are still significant challenges in designing a delivery system that is capable of delivering the precursor gases in the right manner to encourage effective aerosol formation. For example, it is unclear whether aerial shells should be designed to leak slowly or burst suddenly. The size of aerosol particles is also crucial, and efforts must be made to ensure optimal delivery.[52]
- Distribution: It is logistically difficult to deliver aerosols evenly around the globe. Challenges therefore exist in creating a network of delivery points sufficient to allow viable geoengineering from a limited number of launching sites.
Potential negative effects
Geoengineering in general is a controversial technique, and carries problems and risks such as weaponisation, but certain problems are specific to, or more pronounced with this technique.[53]
- Drought, particularly monsoon failure in Asia and Africa is a major risk.[54]
- Ozone depletion is a potential side effect of sulfur aerosols.[55][56]
- Whitening of the sky: Aerosols will noticeably affect the appearance of the sky, resulting a potential whitening effect, and altered sunsets.[57]
- Tropopause warming and the humidification of the stratosphere.[58]
- Effect on clouds: Cloud formation may be affected, notably cirrus clouds and polar stratospheric clouds.
- Health effects: Whilst the sulfate particles are natural, they still have potential health effects.[59] Minimising these effects is principally achieved by ensuring the particles stay aloft as long as possible, thus reducing the tonnages returning into the lower atmosphere.
- Effect on ecosystems: The diffusion of sunlight may impact on plant growth.[60][61][62]
- Effect on solar energy: Incident sunlight will be lower,[63] which may affect solar power systems both directly and disproportionately, especially in the case that such systems rely on direct radiation.[64]
- Deposition effects: Although predicted to be insignificant,[65] there is nevertheless a risk of direct environmental damage from falling particles.
- Uneven effects: Aerosols are reflective, making them more effective during the day. Greenhouse gases block outbound radiation 24hrs a day.[66]
Further, the delivery methods may cause significant problems, notably climate change[67] and possible ozone depletion[68] in the case of aircraft, and litter in the case of untethered balloons.
Alternatives
Sulfur-based aerosols are not the only choice for geoengineering projects of this type. Glass-based aerosols have also been proposed,[69] as have aluminium-based powders. It is thought that these will minimise the potential for ozone depletion and acidic or toxic effects on deposition. However, these materials are not believed to be as well understood as the naturally-abundant sulfur aerosols.
In 1975 the US Navy patented a device for producing "a powder contrail having maximum radiation-scattering ability." The powder contained a mixture of 0.3 micrometre-sized titanium dioxide pigment particles coated with 0.007 micrometre hydrophobic colloidal silica and 4.5 micrometre particles of silica gel. The purpose of the apparatus was "to generate contrails or reflective screens for any desired purpose."
The Welsbach Patent[70] proposed using "very fine, talcum-like" powder of 10 to 100 micrometre-sized aluminium oxide to produce a "pure white plume" in the sky.
See also
References
- ^ http://www.geocrisis.com/cp_wigley_cool_earth_injections.htm
- ^ http://findarticles.com/p/articles/mi_m1200/is_/ai_12140641
- ^ http://volcanoes.usgs.gov/hazards/gas/s02aerosols.php
- ^ http://www.pmel.noaa.gov/pubs/outstand/bate1229/estimate.shtml
- ^ http://www.newmediastudio.org/DataDiscovery/Aero_Ed_Center/Category/A_sulfate.html
- ^ http://www.newmediastudio.org/DataDiscovery/Aero_Ed_Center/Category/A_sulfate.html
- ^ http://www.american.edu/TED/geyser.htm
- ^ http://www.wipo.int/pctdb/en/wo.jsp?IA=US2003004573&DISPLAY=DESC
- ^ http://www.pmel.noaa.gov/pubs/outstand/bate1229/estimate.shtml
- ^ http://www.newmediastudio.org/DataDiscovery/Aero_Ed_Center/Category/A_sulfate.html
- ^ http://www.sciencedirect.com/science?_ob=ArticleURL&_udi=B6VH3-4D2FG64-7&_user=10&_rdoc=1&_fmt=&_orig=search&_sort=d&view=c&_acct=C000050221&_version=1&_urlVersion=0&_userid=10&md5=1c4665fbf2e86907a5353ec113b2f610
- ^ http://www.newmediastudio.org/DataDiscovery/Aero_Ed_Center/Category/A_sulfate.html
- ^ http://adsabs.harvard.edu/abs/2002AGUFM.A71D0130S
- ^ http://www.sciencemag.org/cgi/content/abstract/315/5808/84
- ^ http://pubs.usgs.gov/pinatubo/self/index.html
- ^ http://earthobservatory.nasa.gov/Features/Volcano/
- ^ http://dsa.uniparthenope.it/dsa/LinkClick.aspx?link=DUSTER_Summary.pdf&tabid=926&mid=4373&language=it-IT
- ^ http://www.geocrisis.com/cp_wigley_cool_earth_injections.htm
- ^ http://www.pmel.noaa.gov/pubs/outstand/bate1229/estimate.shtml
- ^ Rampino and Self, 1982
- ^ http://findarticles.com/p/articles/mi_m1200/is_/ai_12140641
- ^ http://volcanoes.usgs.gov/hazards/gas/s02aerosols.php
- ^ http://climate.envsci.rutgers.edu/pdf/20Reasons.pdf
- ^ Figure 3: The global mean radiative forcing of the climate system for the year 2000, relative to 1750. Climate Change 2001: Working Group I: The Scientific Basis. IPCC.
- ^ http://oea.larc.nasa.gov/PAIS/Aerosols.html
- ^ http://remus.jpl.nasa.gov/info.htm
- ^ http://www.meteoros.de/psc/psce.htm
- ^ Pincus & Baker 1994
- ^ Albrecht 1989
- ^ Chemical Amplification (or dampening) of the Twomey Effect: Conditions derived from droplet activation theory. T.A. Rissman, A. Nenes, J.H. Seinfeld.
- ^ http://volcanoes.usgs.gov/hazards/gas/s02aerosols.php
- ^ http://www.agu.org/pubs/crossref/2003/2001JD001508.shtml
- ^ http://www.theozonehole.com/ozonedestruction.htm
- ^ http://www.solcomhouse.com/ozone.htm
- ^ http://www.atmosp.physics.utoronto.ca/MAM/jones_Brewer_Dobson.pdf
- ^ D. W. Olsen et al., “When the Sky Ran Red: The Story Behind The Scream,” Sky & Telescope, February 2004, pp. 29–35
- ^ http://adsabs.harvard.edu/abs/2008AGUFM.U43A0041K
- ^ http://climate.envsci.rutgers.edu/pdf/aciddeposition7.pdf
- ^ http://journals.royalsociety.org/content/84j11614488142u8/
- ^ http://www.cogci.dk/news/Crutzen_albedo%20enhancement_sulfur%20injections.pdf
- ^ http://www.met.reading.ac.uk/pg-research/downloads/2009/pgr-charlton.pdf
- ^ http://www.geocrisis.com/cp_wigley_cool_earth_injections.htm
- ^ http://www.geocrisis.com/cp_escape_route_gw.htm
- ^ Five ways to save the world-overview
- ^ http://www.wired.com/science/planetearth/magazine/16-07/ff_geoengineering
- ^ Paul Crutzen, “Albedo enhancement by stratospheric sulfur injections: a contribution to resolve a policy dilemma?” Journal of Climatic Change, vol. 77, no. 3-4, August 2006
- ^ http://www.pmel.noaa.gov/pubs/outstand/bate1229/estimate.shtml
- ^ http://www.aviationtrivia.info/HIGHEST-FLYING-AIRCRAFT.php
- ^ http://findarticles.com/p/articles/mi_m1200/is_/ai_12140641
- ^ http://www.crosswalk.com/news/11572945/
- ^ http://www.sciencedaily.com/releases/2008/02/080217094602.htm
- ^ http://www.agu.org/pubs/crossref/2008/2007GL032179.shtml
- ^ http://climate.envsci.rutgers.edu/pdf/20Reasons.pdf
- ^ http://climate.envsci.rutgers.edu/pdf/2008JD010050small.pdf
- ^ http://www.pnas.org/content/99/5/2609.full.pdf
- ^ http://www.cosis.net/abstracts/EGU2008/10823/EGU2008-A-10823.pdf
- ^ D. W. Olsen et al., “When the Sky Ran Red: The Story Behind The Scream,” Sky & Telescope, February 2004, pp. 29–35
- ^ http://www.cosis.net/abstracts/EGU2008/10823/EGU2008-A-10823.pdf
- ^ http://www.dh.gov.uk/en/Publicationsandstatistics/Publications/PublicationsPolicyAndGuidance/DH_4102961
- ^ L. Gu et al., “Responses of Net Ecosystem Exchanges of Carbon Dioxide to Changes in Cloudiness: Results from Two North American Deciduous Forests,” Journal of Geophysical Research, vol. 104, no. 31, pp. 421–31, 434 (1999)
- ^ L. Gu et al., “Advantages of Diffuse Radiation for Terrestrial Ecosystem Productivity,” Journal of Geophysical Research, vol. 107, (2002)
- ^ L. Gu et al., “Response of a Deciduous Forest to the Mount Pinatubo Eruption: Enhanced Photosynthesis,” Science, vol.299, pp. 2,035–38 (2003)
- ^ Balan Govindasamy and Ken Caldeira, “Geoengineering Earth’s Radiation Balance to Mitigate CO2-Induced Climate Change,” Geophysical Research Letters, vol. 27, pp. 2,141–44 (2000)
- ^ Michael C. MacCracken, “Geoengineering: Worthy of Cautious Evaluation?” Climatic Change, vol. 77, pp. 235–43 (2006)
- ^ http://climate.envsci.rutgers.edu/pdf/aciddeposition7.pdf
- ^ http://www.washington.edu/research/pathbreakers/1969e.html
- ^ http://www.grida.no/publications/other/ipcc_sr/?src=/climate/ipcc/aviation/064.htm
- ^ http://stason.org/TULARC/science-engineering/ozone-depletion-intro/24-Will-commercial-supersonic-aircraft-damage-the-ozone-laye.html
- ^ http://www.crosswalk.com/news/11572945/
- ^ US Patent 5003186
