ce |
Michael7604 (talk | contribs) recategorized from Chlorobenzenes to Chlorobenzene derivatives |
||
| (40 intermediate revisions by 27 users not shown) | |||
| Line 10: | Line 10: | ||
| width = 150px |
| width = 150px |
||
| alt = |
| alt = |
||
| image2 = Pyrimethamine-3D- |
| image2 = Pyrimethamine-from-xtal-3D-bs-17.png |
||
| width2 = |
| width2 = |
||
| alt2 = |
| alt2 = |
||
| Line 25: | Line 25: | ||
| licence_US = Pyrimethamine |
| licence_US = Pyrimethamine |
||
| pregnancy_AU = B3 |
| pregnancy_AU = B3 |
||
| pregnancy_AU_comment = <ref name="Drugs.com pregnancy">{{cite web | title=Pyrimethamine (Daraprim) Use During Pregnancy | website=Drugs.com | date=31 July 2019 | url=https://www.drugs.com/pregnancy/pyrimethamine.html | access-date=28 February 2020}}</ref> |
| pregnancy_AU_comment = <ref name="Drugs.com pregnancy">{{cite web | title=Pyrimethamine (Daraprim) Use During Pregnancy | website=Drugs.com | date=31 July 2019 | url=https://www.drugs.com/pregnancy/pyrimethamine.html | access-date=28 February 2020 | archive-date=3 December 2016 | archive-url=https://web.archive.org/web/20161203130908/https://www.drugs.com/pregnancy/pyrimethamine.html | url-status=live }}</ref> |
||
| pregnancy_US = C |
|||
| pregnancy_US_comment = <ref name="Drugs.com pregnancy" /> |
|||
| pregnancy_category= |
| pregnancy_category= |
||
| ⚫ | |||
| dependency_liability = |
|||
| addiction_liability = |
|||
| ⚫ | |||
| class = |
| class = |
||
| ATCvet = |
| ATCvet = |
||
| Line 50: | Line 46: | ||
| legal_NZ_comment = |
| legal_NZ_comment = |
||
| legal_UK = POM |
| legal_UK = POM |
||
| legal_UK_comment = <ref>{{cite web | title=Daraprim Tablets |
| legal_UK_comment = <ref>{{cite web | title=Daraprim Tablets – Summary of Product Characteristics (SmPC) | website=(emc) | date=19 February 2020 | url=https://www.medicines.org.uk/emc/product/938/smpc | access-date=28 February 2020 | archive-date=29 February 2020 | archive-url=https://web.archive.org/web/20200229073618/https://www.medicines.org.uk/emc/product/938/smpc | url-status=live }}</ref> |
||
| legal_US = Rx-only |
| legal_US = Rx-only |
||
| legal_US_comment = |
| legal_US_comment = |
||
| Line 113: | Line 109: | ||
<!-- Definition and symptoms --> |
<!-- Definition and symptoms --> |
||
'''Pyrimethamine''', sold under the brand name '''Daraprim''' among others, is a medication used with [[leucovorin]] to treat the |
'''Pyrimethamine''', sold under the brand name '''Daraprim''' among others, is a medication used with [[leucovorin]] (leucovorin is used to decrease side effects of pyrimethamine; it does not have intrinsic anti-parasitic activity) to treat the parasitic diseases [[toxoplasmosis]] and [[cystoisosporiasis]].<ref name=AHFS2016/><ref name=Ric2015>{{cite book| vauthors = Hamilton R |title=Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition|date=2015|publisher=Jones & Bartlett Learning|isbn=9781284057560|page=54}}</ref> It is also used with [[dapsone]] as a second-line option to prevent [[Pneumocystis pneumonia|''Pneumocystis jiroveci'' pneumonia]] in people with [[HIV/AIDS]].<ref name=AHFS2016/> It was previously used for [[malaria]] but is no longer recommended due to resistance.<ref name=AHFS2016/> Pyrimethamine is [[oral administration|taken by mouth]].<ref name=AHFS2016>{{cite web|title=Pyrimethamine|url=https://www.drugs.com/monograph/pyrimethamine.html|publisher=The American Society of Health-System Pharmacists|access-date=2 December 2016|url-status=live|archive-url=https://web.archive.org/web/20161202172816/https://www.drugs.com/monograph/pyrimethamine.html|archive-date=2 December 2016}}</ref> |
||
<!-- Side effects and mechanism --> |
<!-- Side effects and mechanism --> |
||
| Line 119: | Line 115: | ||
<!-- History and culture --> |
<!-- History and culture --> |
||
Pyrimethamine was discovered in 1952 and came into medical use in 1953.<ref name=AHFS2016/><ref>{{cite book| |
Pyrimethamine was discovered in 1952 and came into medical use in 1953.<ref name=AHFS2016/><ref>{{cite book|vauthors=Sylvie M, Pierre C, Jean M|title=Biodiversity of Malaria in the world|date=2008|publisher=John Libbey Eurotext|isbn=9782742009633|page=6|url=https://books.google.com/books?id=Sk0JBAAAQBAJ&pg=PA6|language=en|access-date=10 September 2017|archive-date=14 January 2023|archive-url=https://web.archive.org/web/20230114034306/https://books.google.com/books?id=Sk0JBAAAQBAJ&pg=PA6|url-status=live}}</ref> It is on the [[WHO Model List of Essential Medicines|World Health Organization's List of Essential Medicines]].<ref name="WHO21st">{{cite book | vauthors = ((World Health Organization)) | title = World Health Organization model list of essential medicines: 21st list 2019 | year = 2019 | hdl = 10665/325771 | author-link = World Health Organization | publisher = World Health Organization | location = Geneva | id = WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO }}</ref> It was approved as a [[generic drug|generic]] in the United States in February 2020.<ref name="FDA PR 20200228" /> |
||
==Medical uses== |
==Medical uses== |
||
Pyrimethamine is typically given with a [[sulfonamide (medicine)|sulfonamide]] and [[folinic acid]].<ref name="AMH">{{cite book|title=Australian Medicines Handbook|publisher=The Australian Medicines Handbook Unit Trust|year=2013|isbn=978-0-9805790-9-3|edition=2013| |
Pyrimethamine is typically given with a [[sulfonamide (medicine)|sulfonamide]] and [[folinic acid]].<ref name="AMH">{{cite book|title=Australian Medicines Handbook|publisher=The Australian Medicines Handbook Unit Trust|year=2013|isbn=978-0-9805790-9-3|edition=2013| veditors = Rossi S |place=Adelaide}}</ref> |
||
It is used for the treatment of toxoplasmosis, [[actinomycosis]], and [[isosporiasis]], and for the treatment and prevention of ''[[Pneumocystis jirovecii]]'' pneumonia.<ref name=AHFS2016/> |
It is used for the treatment of toxoplasmosis, [[actinomycosis]], and [[isosporiasis]], and for the treatment and prevention of ''[[Pneumocystis jirovecii]]'' pneumonia.<ref name=AHFS2016/> |
||
| Line 129: | Line 125: | ||
Pyrimethamine is also used in combination with [[sulfadiazine]] to treat active [[toxoplasmosis]]. The two drugs bind the same enzymatic targets as the drugs [[trimethoprim]] and [[sulfamethoxazole]] - [[dihydrofolate reductase]] and dihydropteroate synthase, respectively.{{fact|date=October 2020}} |
Pyrimethamine is also used in combination with [[sulfadiazine]] to treat active [[toxoplasmosis]]. The two drugs bind the same enzymatic targets as the drugs [[trimethoprim]] and [[sulfamethoxazole]] - [[dihydrofolate reductase]] and dihydropteroate synthase, respectively.{{fact|date=October 2020}} |
||
Pyrimethamine has also been used in several trials to treat [[toxoplasmic chorioretinitis|retinochoroiditis]].<ref name="Pradhan">{{cite journal |vauthors=Pradhan E, Bhandari S, Gilbert RE, Stanford M |title= Antibiotics versus no treatment for toxoplasma retinochoroiditis |journal=Cochrane Database |
Pyrimethamine has also been used in several trials to treat [[toxoplasmic chorioretinitis|retinochoroiditis]].<ref name="Pradhan">{{cite journal | vauthors = Pradhan E, Bhandari S, Gilbert RE, Stanford M | title = Antibiotics versus no treatment for toxoplasma retinochoroiditis | journal = The Cochrane Database of Systematic Reviews | volume = 2016 | issue = 5 | pages = CD002218 | date = May 2016 | pmid = 27198629 | pmc = 7100541 | doi = 10.1002/14651858.CD002218.pub2 }}</ref> |
||
=== Pregnancy consideration === |
=== Pregnancy consideration === |
||
Pyrimethamine is labeled as pregnancy category C in the United States.<ref name="Drugs.com pregnancy" /><ref name="Daraprim label">{{cite web | title=Daraprim- pyrimethamine tablet | website=DailyMed | date=31 August 2017 | url=https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=5368616d-6520-6f6e-2053-686b72656c69 | access-date=28 February 2020}}</ref> To date, not enough evidence on its risks in pregnancy or its effects on the fetus is available.<ref name="Daraprim label" /><ref name=" |
Pyrimethamine is labeled as pregnancy category C in the United States.<ref name="Drugs.com pregnancy" /><ref name="Daraprim label">{{cite web | title=Daraprim- pyrimethamine tablet | website=DailyMed | date=31 August 2017 | url=https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=5368616d-6520-6f6e-2053-686b72656c69 | access-date=28 February 2020 | archive-date=3 August 2020 | archive-url=https://web.archive.org/web/20200803180112/https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=5368616d-6520-6f6e-2053-686b72656c69 | url-status=live }}</ref> To date, not enough evidence on its risks in pregnancy or its effects on the fetus is available.<ref name="Daraprim label" /><ref name="AIDSinfo">{{Cite web |url= https://aidsinfo.nih.gov/drugs/445/pyrimethamine/0/professional#LINK_cdcdcf89-7c83-4eec-8b1c-a99ee17b39ea |title=Pyrimethamine {{!}} FDA Label - Tablet |website=AIDSinfo|access-date=2016-11-07|url-status=dead|archive-url= https://web.archive.org/web/20170118044050/https://aidsinfo.nih.gov/drugs/445/pyrimethamine/0/professional#LINK_cdcdcf89-7c83-4eec-8b1c-a99ee17b39ea |archive-date=2017-01-18}}</ref> |
||
===Malaria=== |
===Malaria=== |
||
It is primarily active against ''[[Plasmodium falciparum]]'', but also against ''[[Plasmodium vivax]]''.<ref name = MD>{{cite web|title=Pyrimethamine|work=Martindale: The Complete Drug Reference|publisher=Pharmaceutical Press| |
It is primarily active against ''[[Plasmodium falciparum]]'', but also against ''[[Plasmodium vivax]]''.<ref name = MD>{{cite web|title=Pyrimethamine|work=Martindale: The Complete Drug Reference|publisher=Pharmaceutical Press|access-date=12 April 2014|date=13 December 2013|url=http://www.medicinescomplete.com/mc/martindale/current/1389-y.htm|veditors=Brayfield A|archive-date=29 August 2021|archive-url=https://web.archive.org/web/20210829163814/https://about.medicinescomplete.com/wp-content/plugins/revslider/public/assets/js/extensions/revolution.extension.layeranimation.min.js?version=5.4.5|url-status=live}}</ref> Due to the emergence of pyrimethamine-resistant strains of ''P. falciparum'', pyrimethamine alone is seldom used now. In combination with a long-acting sulfonamide such as [[sulfadiazine]], it was widely used, such as in [[Fansidar]], though resistance to this combination is increasing.<ref name = MD/> |
||
==Contraindications== |
==Contraindications== |
||
| Line 141: | Line 137: | ||
==Side effects== |
==Side effects== |
||
When higher doses are used, as in the treatment of toxoplasmosis, pyrimethamine can cause gastrointestinal symptoms such as nausea, vomiting, [[glossitis]], anorexia, and diarrhea.<ref name=" |
When higher doses are used, as in the treatment of toxoplasmosis, pyrimethamine can cause gastrointestinal symptoms such as nausea, vomiting, [[glossitis]], anorexia, and diarrhea.<ref name="AIDSinfo" /><ref name="www.drugs.com">{{Cite web|url=https://www.drugs.com/sfx/daraprim-side-effects.html|title=Daraprim Side Effects in Detail |website= www.drugs.com |access-date=2016-11-07 |url-status=live |archive-url= https://web.archive.org/web/20161108052945/https://www.drugs.com/sfx/daraprim-side-effects.html |archive-date=2016-11-08}}</ref> A rash, which can be indicative of a hypersensitivity reaction, is also seen, particularly in combination with sulfonamides.<ref name="AIDSinfo" /> Central nervous system effects include ataxia, tremors, and seizures.<ref name="www.drugs.com" /> Hematologic side effects such as [[thrombocytopenia]], [[leukopenia]], and anemia can also occur.<ref name="www.drugs.com" /> |
||
| ⚫ | |||
| ⚫ | |||
Other antifolate agents such as [[methotrexate]] and [[trimethoprim]] may potentiate the antifolate actions of pyrimethamine, leading to potential folate deficiency, anaemia, and other [[blood dyscrasia]]s.<ref name = AMH/> |
Other antifolate agents such as [[methotrexate]] and [[trimethoprim]] may potentiate the antifolate actions of pyrimethamine, leading to potential folate deficiency, anaemia, and other [[blood dyscrasia]]s.<ref name = AMH/> |
||
==Mechanism of action== |
==Mechanism of action== |
||
Pyrimethamine interferes with the regeneration of [[tetrahydrofolic acid]] from [[dihydrofolate]] by competitively inhibiting the enzyme [[dihydrofolate reductase]].<ref name=TGA>{{cite web|title= |
Pyrimethamine interferes with the regeneration of [[tetrahydrofolic acid]] from [[dihydrofolate]] by competitively inhibiting the enzyme [[dihydrofolate reductase]].<ref name=TGA>{{cite web|title=Product Information Daraprim Tablets |work=TGA eBusiness Services|publisher=Aspen Pharmacare Australia Pty Ltd|date=5 December 2011|access-date=12 April 2014|url=https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent&id=CP-2013-PI-01016-1|page=1|url-status=live|archive-url=https://web.archive.org/web/20161005203017/https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent&id=CP-2013-PI-01016-1|archive-date=5 October 2016}}</ref> Tetrahydrofolic acid is essential for [[DNA]] and [[RNA]] synthesis in many species, including protozoa.<ref name = TGA/> It has also been found to reduce the expression of [[SOD1]], a key protein involved in amyotrophic lateral sclerosis.<ref>{{cite journal | vauthors = Limpert AS, Mattmann ME, Cosford ND | title = Recent progress in the discovery of small molecules for the treatment of amyotrophic lateral sclerosis (ALS) | journal = Beilstein Journal of Organic Chemistry | volume = 9 | pages = 717–732 | date = 2013 | pmid = 23766784 | pmc = 3678841 | doi = 10.3762/bjoc.9.82 | url = http://www.beilstein-journals.org/bjoc/content/pdf/1860-5397-9-82.pdf | url-status = live | archive-url = https://web.archive.org/web/20140413140738/http://www.beilstein-journals.org/bjoc/content/pdf/1860-5397-9-82.pdf | archive-date = 2014-04-13 }}</ref><ref>{{cite journal | vauthors = Lange DJ, Andersen PM, Remanan R, Marklund S, Benjamin D | title = Pyrimethamine decreases levels of SOD1 in leukocytes and cerebrospinal fluid of ALS patients: a phase I pilot study | journal = Amyotrophic Lateral Sclerosis & Frontotemporal Degeneration | volume = 14 | issue = 3 | pages = 199–204 | date = April 2013 | pmid = 22985433 | doi = 10.3109/17482968.2012.724074 | s2cid = 39846211 }}</ref> |
||
===Other medications=== |
===Other medications=== |
||
| Line 155: | Line 152: | ||
===Mechanism of resistance=== |
===Mechanism of resistance=== |
||
Resistance to pyrimethamine is widespread. Mutations in the malarial gene for dihydrofolate reductase may reduce its effectiveness.<ref name="Gatton2004">{{ |
Resistance to pyrimethamine is widespread. Mutations in the malarial gene for dihydrofolate reductase may reduce its effectiveness.<ref name="Gatton2004">{{cite journal | vauthors = Gatton ML, Martin LB, Cheng Q | title = Evolution of resistance to sulfadoxine-pyrimethamine in Plasmodium falciparum | journal = Antimicrobial Agents and Chemotherapy | volume = 48 | issue = 6 | pages = 2116–2123 | date = June 2004 | pmid = 15155209 | pmc = 415611 | doi = 10.1128/AAC.48.6.2116-2123.2004 }}</ref> These mutations decrease the binding affinity between pyrimethamine and dihydrofolate reductase via loss of [[hydrogen bonds]] and [[steric]] interactions.<ref name="Sirichaiwat2004">{{cite journal | vauthors = Sirichaiwat C, Intaraudom C, Kamchonwongpaisan S, Vanichtanankul J, Thebtaranonth Y, Yuthavong Y | title = Target guided synthesis of 5-benzyl-2,4-diamonopyrimidines: their antimalarial activities and binding affinities to wild type and mutant dihydrofolate reductases from Plasmodium falciparum | journal = Journal of Medicinal Chemistry | volume = 47 | issue = 2 | pages = 345–354 | date = January 2004 | pmid = 14711307 | doi = 10.1021/jm0303352 }}</ref> |
||
==History== |
==History== |
||
[[File:Pyrimethamine traditional synthesis.png|thumb|Synthesis of pyrimethamine typically begins with ''p''-chlorophenylacetonitrile, which undergoes a [[condensation reaction]] with [[ethyl propionate]] ester; the product of this then reacts with [[diazomethane]] to form an [[enol ether]], which reacts with free [[guanidine]] in a second condensation reaction.]] |
[[File:Pyrimethamine traditional synthesis.png|thumb|Synthesis of pyrimethamine typically begins with ''p''-chlorophenylacetonitrile, which undergoes a [[condensation reaction]] with [[ethyl propionate]] ester; the product of this then reacts with [[diazomethane]] to form an [[enol ether]], which reacts with free [[guanidine]] in a second condensation reaction.]] |
||
[[Nobel Prize]]-winning American scientist [[Gertrude Elion]] developed the drug at Burroughs-Wellcome (now part of GlaxoSmithKline) to combat malaria.<ref |
[[Nobel Prize]]-winning American scientist [[Gertrude Elion]] developed the drug at Burroughs-Wellcome (now part of GlaxoSmithKline) to combat malaria.<ref>{{cite book | vauthors = Vasudevan DM, Sreekumari S, Vaidyanathan K |title=Textbook of Biochemistry for Medical Students |date=2013 |publisher=JP Medical Ltd |isbn=9789350905302 |oclc=843532694 |page=491 |url=https://books.google.com/books?id=nQz8AAAAQBAJ&pg=PA491 |access-date=15 January 2016 |url-status=live |archive-url=https://web.archive.org/web/20170908221240/https://books.google.com/books?id=nQz8AAAAQBAJ&pg=PA491 |archive-date=8 September 2017 }}</ref> Pyrimethamine has been available since 1953.<ref>{{cite news|newspaper=[[The Washington Post]]|title=CEO who raised price of old pill more than $700 calls journalist a 'moron' for asking why| vauthors = Cha AE |date=2015-09-22|url=https://www.washingtonpost.com/news/to-your-health/wp/2015/09/21/ceo-of-company-that-raised-the-price-of-old-pill-hundreds-of-dollars-overnight-calls-journalist-a-moron-for-asking-why/|url-status=live|archive-url=https://web.archive.org/web/20150923060804/https://www.washingtonpost.com/news/to-your-health/wp/2015/09/21/ceo-of-company-that-raised-the-price-of-old-pill-hundreds-of-dollars-overnight-calls-journalist-a-moron-for-asking-why/|archive-date=2015-09-23}}</ref> In 2010, [[GlaxoSmithKline]] sold the marketing rights for Daraprim to CorePharma. [[Impax Laboratories]] sought to buy CorePharma in 2014, and completed the acquisition, including Daraprim, in March 2015.<ref name=NYTPollack20Sep15>{{cite news| vauthors = Pollack A |title=Drug Goes From $13.50 a Tablet to $750, Overnight|url=https://www.nytimes.com/2015/09/21/business/a-huge-overnight-increase-in-a-drugs-price-raises-protests.html|access-date=17 December 2015|work=[[The New York Times]]|date=20 September 2015|url-status=live|archive-url=https://web.archive.org/web/20151216002021/http://www.nytimes.com/2015/09/21/business/a-huge-overnight-increase-in-a-drugs-price-raises-protests.html|archive-date=16 December 2015}}</ref> In August 2015, the rights were bought by [[Turing Pharmaceuticals]].<ref>{{cite news|work=[[Forbes]]|title=Here's A Way For Pharma To Prevent Outrageous Generic Price Increases – And Help Its Reputation| vauthors = LaMattina J |date=2015-09-21|url=https://www.forbes.com/sites/johnlamattina/2015/09/21/heres-a-way-for-pharma-to-prevent-outrageous-generic-price-increases-and-help-its-reputation/|url-status=live|archive-url=https://web.archive.org/web/20150923090053/http://www.forbes.com/sites/johnlamattina/2015/09/21/heres-a-way-for-pharma-to-prevent-outrageous-generic-price-increases-and-help-its-reputation/|archive-date=2015-09-23}}</ref> Turing subsequently became infamous for a price hike controversy when it raised the price of a dose of the drug in the U.S. market from US$13.50 to US$750, a 5,500% increase.<ref name=CorrectPercent>{{cite web | vauthors = Kliff S | date = 22 September 2015 | title = Vox Explainers: A Drug Company Raised a Pill's Price 5,500 Percent Because, in America, It Can | url = https://www.vox.com/2015/9/22/9366721/daraprim-price-shkreli-turing | website = Vox | access-date = 9 December 2015 | url-status = live | archive-url = https://web.archive.org/web/20151210022145/http://www.vox.com/2015/9/22/9366721/daraprim-price-shkreli-turing | archive-date = 10 December 2015 }}</ref> |
||
== Society and culture == |
|||
=== Economics === |
|||
In the United States in 2015, [[Turing Pharmaceuticals]] was criticized for increasing the price 50-fold, from US$13.50 to $750 a tablet,<ref>{{cite news |vauthors=Pollack A |title=Drug Goes From $13.50 a Tablet to $750, Overnight |url=https://www.nytimes.com/2015/09/21/business/a-huge-overnight-increase-in-a-drugs-price-raises-protests.html |access-date=31 March 2020 |work=The New York Times |date=20 September 2015 |quote=Turing immediately raised the price to $750 a tablet from $13.50, bringing the annual cost of treatment for some patients to hundreds of thousands of dollars. |archive-date=16 December 2015 |archive-url=https://web.archive.org/web/20151216002021/http://www.nytimes.com/2015/09/21/business/a-huge-overnight-increase-in-a-drugs-price-raises-protests.html |url-status=live }}</ref> leading to a cost of $75,000 for a course of treatment reported at one hospital.<ref>{{cite journal | vauthors = Alpern JD, Song J, Stauffer WM | title = Essential Medicines in the United States--Why Access Is Diminishing | journal = The New England Journal of Medicine | volume = 374 | issue = 20 | pages = 1904–1907 | date = May 2016 | pmid = 27192669 | doi = 10.1056/nejmp1601559 | quote = Heavy scrutiny followed, and although Turing agreed to reduce the price, the drug remains prohibitively expensive for many patients. Recently, at our hospital, an immigrant patient with a new diagnosis of HIV– AIDS and toxoplasmosis couldn’t receive first-line therapy because of cost: the price for 100 pills was $75,000. }}</ref> |
|||
| ⚫ | |||
==Availability and price== |
|||
| ⚫ | In the United States, in 2015, with Turing Pharmaceuticals' acquisition of the US marketing rights for Daraprim tablets,<ref>{{cite web | work = Turing Pharmaceuticals AG | url = http://www.prnewswire.com/news-releases/turing-pharmaceuticals-ag-acquires-us-marketing-rights-to-daraprim-pyrimethamine-300125853.html | title = Turing Pharmaceuticals AG Acquires U.S. Marketing Rights to Daraprim (pyrimethamine) | archive-url = https://web.archive.org/web/20150922031456/http://www.prnewswire.com/news-releases/turing-pharmaceuticals-ag-acquires-us-marketing-rights-to-daraprim-pyrimethamine-300125853.html | archive-date = 22 September 2015| date = 10 August 2015 | publisher = PR Newswire Association LLC }}</ref> Daraprim became a single-source and specialty pharmacy item, and the price was increased.<ref name=pt>{{cite web | vauthors = Mahoney MV | url = https://www.pharmacytimes.com/view/new-pyrimethamine-dispensing-program-what-pharmacists-should-know | title = New Pyrimethamine Dispensing Program: What Pharmacists Should Know | archive-url = https://web.archive.org/web/20150906065039/http://www.pharmacytimes.com/contributor/monica-v-golik-mahoney-pharmd-bcps-aq-id/2015/07/new-pyrimethamine-dispensing-program-what-pharmacists-should-know | archive-date = 6 September 2015| work = PharmacyTimes | date = 17 July 2015 }}</ref> The cost of a monthly course for a person on 75 mg dose rose to about $75,000/month at one hospital, or $750 per tablet while it was previously priced at $13.50.<ref name=nyt>{{cite news| vauthors = Pollack A |title=Drug Goes From $13.50 a Tablet to $750, Overnight|work=The New York Times |url=https://www.nytimes.com/2015/09/21/business/a-huge-overnight-increase-in-a-drugs-price-raises-protests.html?ref=health&_r=0|access-date=21 September 2015|date=20 September 2015|url-status=live|archive-url=https://web.archive.org/web/20150925195708/http://www.nytimes.com/2015/09/21/business/a-huge-overnight-increase-in-a-drugs-price-raises-protests.html?ref=health&_r=0|archive-date=25 September 2015}}</ref> |
||
| ⚫ | |||
| ⚫ | In the United States, in 2015, with Turing Pharmaceuticals' acquisition of the US marketing rights for Daraprim tablets,<ref>Turing Pharmaceuticals AG |
||
Outpatients could no longer obtain the medication from a community pharmacy, but only through a single dispensing pharmacy, Walgreens Specialty Pharmacy, and institutions could no longer order from their general wholesaler, but had to set up an account with the Daraprim Direct program.<ref name=pt/><ref name=Lowe> |
Outpatients could no longer obtain the medication from a community pharmacy, but only through a single dispensing pharmacy, Walgreens Specialty Pharmacy, and institutions could no longer order from their general wholesaler, but had to set up an account with the Daraprim Direct program.<ref name=pt/><ref name=Lowe>{{cite web | vauthors = Lowe D | date = 11 September 2014 | work = In the Pipeline | url = https://www.science.org/content/blog-post/most-unconscionable-drug-price-hike-i-have-yet-seen | title = The Most Unconscionable Drug Price Hike I Have Yet Seen | archive-url = https://web.archive.org/web/20160607212017/http://blogs.sciencemag.org/pipeline/archives/2014/09/11/the_most_unconscionable_drug_price_hike_i_have_yet_seen | archive-date = 7 June 2016 }}</ref> Presentations from [[Martin Shkreli#Retrophin|Retrophin]], a company formerly headed by [[Martin Shkreli]], CEO of Turing, from which Turing acquired the rights to Daraprim, suggested that a closed distribution system could prevent generic competitors from legally obtaining the drugs for the bioequivalence studies required for FDA approval of a generic drug.<ref name=Lowe/> |
||
Shkreli defended the price hike by saying, "If there was a company that was selling an Aston Martin at the price of a bicycle, and we buy that company and we ask to charge Toyota prices, I don't think that that should be a crime."<ref name="Ramsey_Aston Martin">{{cite news| vauthors = Ramsey L |title=A pharma CEO tried to defend his decision to jack up the price of a critical drug by 5,000% – and it backfired|url=http://www.businessinsider.com/martin-shkreli-defends-daraprim-price-2015-9|publisher=Business Insider|date=22 Sep 2015|url-status=live|archive-url=https://web.archive.org/web/20151010190344/http://www.businessinsider.com/martin-shkreli-defends-daraprim-price-2015-9|archive-date=2015-10-10}}</ref><ref name="Aston Martin_Reuters">{{cite news |title=Company hikes price of popular drug|url=http://uk.reuters.com/video/2015/09/22/company-hikes-price-of-popular-drug?videoId=365682609|work=Reuters|date=22 Sep 2015|url-status=dead|archive-url=https://web.archive.org/web/20151002142824/http://uk.reuters.com/video/2015/09/22/company-hikes-price-of-popular-drug?videoId=365682609|archive-date=2015-10-02}}</ref> As a result of the backlash, Shkreli hired a crisis public relations firm to help explain his fund's move.<ref name="Martin Shkreli-Ripp">{{cite news| vauthors = Tannahill J |title=PR Man Allan Ripp Representing The "Most Hated Man in America"|url=http://everything-pr.com/allan-ripp-represents-martin-shkreli/69517/|publisher=EverythingPR|date=9 Oct 2015|url-status=live|archive-url=https://web.archive.org/web/20151014235241/http://everything-pr.com/allan-ripp-represents-martin-shkreli/69517/|archive-date=2015-10-14}}</ref> Turing Pharmaceuticals announced on 24 November 2015, "that it would not reduce the list price of that drug after all", but they would offer patient assistance programs.<ref name="NYT_2015_11_25">{{cite web | url=https://www.nytimes.com/2015/11/25/business/turing-refuses-to-lower-list-price-of-toxoplasmosis-drug.html?smid=tw-nytimes&smtyp=cur | title=Turing Refuses to Lower List Price of Toxoplasmosis Drug | work=New York Times | date=24 November 2015 | access-date=25 November 2015 | vauthors = Pollack A | url-status=live | archive-url=https://web.archive.org/web/20170908221240/https://www.nytimes.com/2015/11/25/business/turing-refuses-to-lower-list-price-of-toxoplasmosis-drug.html?smid=tw-nytimes&smtyp=cur | archive-date=8 September 2017 }}</ref> ''[[New York Times]]'' journalist Andrew Pollack noted that these programs "are standard for companies selling extremely high-priced drugs. They enable the patients to get the drug while pushing most of the costs onto insurance companies and taxpayers."<ref name="NYT_2015_11_25" /> |
|||
The price increase was criticized by physician groups such as [[HIV Medicine Associates]] and [[Infectious Diseases Society of America]].<ref>{{cite web |url=http://www.hivma.org/uploadedFiles/HIVMA/HomePageContent/PyrimethamineLetterFINAL.pdf |title= |
The price increase was criticized by physician groups such as [[HIV Medicine Associates]] and [[Infectious Diseases Society of America]].<ref>{{cite web |url=http://www.hivma.org/uploadedFiles/HIVMA/HomePageContent/PyrimethamineLetterFINAL.pdf |title=Letter to Tom Evegan and Kevin Burnier |access-date=2015-09-23 |url-status=dead |archive-url=https://web.archive.org/web/20150923004521/http://www.hivma.org/uploadedFiles/HIVMA/HomePageContent/PyrimethamineLetterFINAL.pdf |archive-date=2015-09-23 }}</ref> |
||
In 2016, a group of high school students from [[Sydney Grammar School|Sydney Grammar]] supported by |
In 2016, a group of high school students from [[Sydney Grammar School|Sydney Grammar]] supported by the [[University of Sydney]] prepared pyrimethamine as an illustration that the synthesis is comparatively easy and the price-hike unjustifiable. His team produced 3.7 g for US$20, which would have been worth between US$35,000 and US$110,000 in the [[United States]] at the time.<ref name = SGS-ABC>{{cite news|vauthors = Hunjan R|title = Daraprim drug's key ingredient recreated by high school students in Sydney for just $20|url = https://www.abc.net.au/news/2016-11-30/daraprim-nsw-students-create-drug-martin-shkreli-sold/8078892|date = 30 November 2016|access-date = 23 April 2019|work = [[ABC News (Australia)|ABC News]]|archive-date = 4 April 2019|archive-url = https://web.archive.org/web/20190404095355/https://www.abc.net.au/news/2016-11-30/daraprim-nsw-students-create-drug-martin-shkreli-sold/8078892|url-status = live}}</ref> Shkreli said the schoolboys were not competition, likely because the necessary bioequivalence studies require a sample of the existing medication provided directly by the company, and not simply purchased from a pharmacy, which Turing could decline to provide.<ref name = SGS-Guardian>{{cite news|title = Australian students recreate Martin Shkreli price-hike drug in school lab| vauthors = Davey M |newspaper = [[The Guardian]]|date = 1 December 2016|access-date = 1 December 2016|archive-date = 1 December 2016|url-status = live|url = https://www.theguardian.com/science/2016/dec/01/australian-students-recreate-martin-shkreli-price-hike-drug-in-school-lab|archive-url = https://web.archive.org/web/20161201054640/https://www.theguardian.com/science/2016/dec/01/australian-students-recreate-martin-shkreli-price-hike-drug-in-school-lab}}</ref><ref name="BBC01122016">{{cite news| vauthors = Dunlop G |url = https://www.bbc.com/news/world-australia-38153254|title = Australian boys recreate life-saving drug|date = 1 December 2016|work = [[BBC News]]|access-date = 1 December 2016|url-status = live|archive-url = https://web.archive.org/web/20161201104648/http://www.bbc.com/news/world-australia-38153254|archive-date = 1 December 2016}}</ref> Nonetheless, the students' work was featured in ''[[The Guardian]]''<ref name = SGS-Guardian /> and ''[[Time (magazine)|Time]]'' magazine,<ref>{{cite magazine|title = Watch Martin Shkreli Respond to the School Kids Who Recreated His Drug for $2 a Dose|vauthors = Lui K|date = 2 December 2016|url = http://time.com/4588458/martin-shkreli-response-sydney-students-daraprim/|magazine = [[Time (magazine)|Time]]|access-date = 23 April 2019|archive-date = 3 November 2020|archive-url = https://web.archive.org/web/20201103185003/https://time.com/4588458/martin-shkreli-response-sydney-students-daraprim/|url-status = live}}</ref> and on [[Australian Broadcasting Corporation|ABC Australia]],<ref name = SGS-ABC /> the [[BBC]],<ref name = "BBC01122016" /> and [[CNN]].<ref>{{cite news|vauthors = Roberts E|title = 'Pharma Bro' Martin Shkreli meets his match in a group of Australian schoolboys|url = https://edition.cnn.com/2016/12/01/health/daraprim-oz/index.html|access-date = 23 April 2019|date = 1 December 2016|work = [[CNN]]|archive-date = 23 April 2019|archive-url = https://web.archive.org/web/20190423132358/https://edition.cnn.com/2016/12/01/health/daraprim-oz/index.html|url-status = live}}</ref> |
||
On 22 October 2015, [[Imprimis Pharmaceuticals]] announced it had made available compounded and customizable formulations of pyrimethamine and leucovorin in capsules to be taken by mouth starting as low as $99 |
On 22 October 2015, [[Imprimis Pharmaceuticals]] announced it had made available compounded and customizable formulations of pyrimethamine and leucovorin in capsules to be taken by mouth starting as low as $99 for a 100-count bottle in the United States.<ref>{{Cite web|url = https://www.cnbc.com/2015/11/30/express-scripts-imprimis-to-offer-daraprim-alternative.html|title = Express Scripts, Imprimis to offer $1 Daraprim alternative|website = [[CNBC]]|date = December 2015|access-date = 1 April 2020|archive-date = 7 August 2020|archive-url = https://web.archive.org/web/20200807091406/https://www.cnbc.com/2015/11/30/express-scripts-imprimis-to-offer-daraprim-alternative.html|url-status = live}}</ref> Pyrimethamine was approved as a generic in the United States in February 2020.<ref name="FDA PR 20200228">{{cite press release | title=FDA Approves First Generic of Daraprim | website=U.S. [[Food and Drug Administration]] (FDA) | date=28 February 2020 | url=https://www.fda.gov/news-events/press-announcements/fda-approves-first-generic-daraprim | access-date=28 February 2020 | archive-date=29 February 2020 | archive-url=https://web.archive.org/web/20200229070016/https://www.fda.gov/news-events/press-announcements/fda-approves-first-generic-daraprim | url-status=live }}</ref> |
||
In January 2020, the FTC filed a case against Vyera "alleging an elaborate anticompetitive scheme to preserve a monopoly for the life-saving drug, Daraprim".<ref>{{Cite web|url=https://www.ftc.gov/enforcement/cases-proceedings/161-0001/vyera-pharmaceuticals-llc|title=Vyera Pharmaceuticals, LLC|date=January 27, 2020|website=[[Federal Trade Commission]]|access-date=9 December 2021|archive-date=9 December 2021|archive-url=https://web.archive.org/web/20211209054320/https://www.ftc.gov/enforcement/cases-proceedings/161-0001/vyera-pharmaceuticals-llc|url-status=live}}</ref> A settlement was reached in December 2021. According to [[AP News]], the settlement "requires Vyera and Phoenixus to provide up to $40 million in relief over 10 years to consumers who allegedly were fleeced by their actions and requires them to make Daraprim available to any potential generic competitor at the cost of producing the drug."<ref>{{Cite web|url=https://apnews.com/article/business-health-federal-trade-commission-martin-shkreli-98e0a8b6be3dc6901153610756cbb58a|title='Pharma Bro' firm reaches $40M settlement in gouging case|date=December 8, 2021|website=[[AP News]]|access-date=9 December 2021|archive-date=9 December 2021|archive-url=https://web.archive.org/web/20211209054310/https://apnews.com/article/business-health-federal-trade-commission-martin-shkreli-98e0a8b6be3dc6901153610756cbb58a|url-status=live}}</ref> According to [[Law360]], company executive Kevin Mulleady "agreed to a seven-year ban on working for or holding more than an 8% share in most pharmaceutical companies."<ref>{{Cite web|url=https://www.law360.com/articles/1446580/former-shkreli-co-inks-deal-worth-up-to-40m-with-ftc-ags|title=Former Shkreli Co. Inks Deal Worth Up To $40M With FTC, AGs - Law360|website=[[Law360]]|date=December 7, 2021|vauthors=Koenig B|access-date=9 December 2021|archive-date=9 December 2021|archive-url=https://web.archive.org/web/20211209054310/https://www.law360.com/articles/1446580/former-shkreli-co-inks-deal-worth-up-to-40m-with-ftc-ags|url-status=live}}</ref> |
|||
| ⚫ | |||
| ⚫ | In India, |
||
| ⚫ | |||
In the UK, the same drug is available from [[GlaxoSmithKline|GSK]] at a cost of US$20 (£13) for 30 tablets (about $0.66 each).<ref>{{Cite web|title = What's a fair price for a drug?|url = https://www.bbc.com/news/health-34322720|publisher = [[BBC News Online]]|accessdate = 2015-09-23|url-status = live|archive-url = https://web.archive.org/web/20150924214307/http://www.bbc.com/news/health-34322720|archive-date = 2015-09-24}}</ref> |
|||
| ⚫ | In India, multiple combinations of generic pyrimethamine are available for a price ranging from US$0.04 to US$0.10 each (3–7 rupees).<ref name=CL2015>{{cite web | vauthors = Miseta E |url=http://www.clinicalleader.com/doc/high-drug-prices-should-we-blame-pharma-or-the-fda-0001 |title=High Drug Prices: Should We Blame Pharma Or The FDA? |date=29 September 2015 | work = Clinical Leader |url-status=live|archive-url=https://web.archive.org/web/20150930155628/http://www.clinicalleader.com/doc/high-drug-prices-should-we-blame-pharma-or-the-fda-0001|archive-date=30 September 2015}}</ref><ref>{{Cite web|title =Medline India – Sulfadoxine with Pyrimethamine |url = http://www.medlineindia.com/antibiotic/sulfadoxine_with_pyrimethamine.htm|website = www.medlineindia.com|access-date = 2015-09-22|url-status = live|archive-url = https://web.archive.org/web/20150924051026/http://www.medlineindia.com/antibiotic/sulfadoxine_with_pyrimethamine.htm|archive-date = 2015-09-24}}</ref><ref>{{cite web|url=http://www.ibtimes.co.uk/daraprim-like-drug-costs-less-0-07-india-1521144|title=It is Cheaper for an American patient to fly out to India and buy a year's supply of the medication than buy a single Daraprim tablet in the US|date=25 September 2015|url-status=live|archive-url=https://web.archive.org/web/20150928225100/http://www.ibtimes.co.uk/daraprim-like-drug-costs-less-0-07-india-1521144|archive-date=2015-09-28}}</ref><ref name=MarginalRevolution>{{cite web|url=http://marginalrevolution.com/marginalrevolution/2015/09/generic-drug-regulation.html|title=There is no reason why the United States cannot have as vigorous a market in generic pharmaceuticals as does India|date=24 September 2015|url-status=live|archive-url=https://web.archive.org/web/20151006001053/http://marginalrevolution.com/marginalrevolution/2015/09/generic-drug-regulation.html|archive-date=2015-10-06}}</ref> |
||
In |
In the UK, the same drug is available from [[GlaxoSmithKline|GSK]] at a cost of US$20 (£13) for 30 tablets (about $0.66 each).<ref>{{Cite news | vauthors = Roberts M |title = What's a fair price for a drug?|work = BBC News|date = 22 September 2015|url = https://www.bbc.com/news/health-34322720|access-date = 2015-09-23|url-status = live|archive-url = https://web.archive.org/web/20150924214307/http://www.bbc.com/news/health-34322720|archive-date = 24 September 2015 }}</ref> |
||
In |
In Australia, the drug is available in most pharmacies at a cost of US$9.35 (A$12.99) for 50 tablets (around US$0.18 each).<ref>{{Cite web|title = Chemist Warehouse|url = http://www.chemistwarehouse.com.au/Buy/56166/Daraprim-25mg-Tablets-50|website = www.chemistwarehouse.com.au|access-date = 2015-12-12|url-status = live|archive-url = https://web.archive.org/web/20151222092834/http://www.chemistwarehouse.com.au/Buy/56166/Daraprim-25mg-Tablets-50|archive-date = 2015-12-22}}</ref> |
||
In |
In [[Brazil]], the drug is available for R$0.07 a pill, or about US$0.02.<ref>{{Cite web|title = Remédio que teve aumento de 5.000% nos EUA custa R$ 0,07 no Brasil (e não vai aumentar)|url = http://www.brasilpost.com.br/2015/09/23/remedio-aumento-daraprim_n_8186462.html|website = brasilpost.com.br|access-date = 2015-09-23|url-status = live|archive-url = https://web.archive.org/web/20150925095222/http://www.brasilpost.com.br/2015/09/23/remedio-aumento-daraprim_n_8186462.html|archive-date = 2015-09-25}}</ref> |
||
In [[Switzerland]], the drug is available for US$9.45 (CHF9.05) for 30 tablets (around US$0.32 a piece).<ref>{{Cite web|title = Swiss Compendium information about Daraprim|url = http://compendium.ch/prod/daraprim-cpr-25-mg/fr|website = compendium.ch| |
In [[Switzerland]], the drug is available for US$9.45 (CHF9.05) for 30 tablets (around US$0.32 a piece).<ref>{{Cite web|title = Swiss Compendium information about Daraprim|url = http://compendium.ch/prod/daraprim-cpr-25-mg/fr|website = compendium.ch|access-date = 2017-09-01|archive-date = 29 August 2021|archive-url = https://web.archive.org/web/20210829163806/https://compendium.ch/fr/product/17866-daraprim-tabl-25-mg|url-status = live}}</ref> |
||
==Research== |
==Research== |
||
In 2011, researchers discovered that pyrimethamine can increase [[Beta-hexosaminidase|β-hexosaminidase]] activity, thus potentially slowing down the progression of late-onset [[Tay–Sachs disease]].<ref>{{cite journal| |
In 2011, researchers discovered that pyrimethamine can increase [[Beta-hexosaminidase|β-hexosaminidase]] activity, thus potentially slowing down the progression of late-onset [[Tay–Sachs disease]].<ref>{{cite journal | vauthors = Osher E, Fattal-Valevski A, Sagie L, Urshanski N, Amir-Levi Y, Katzburg S, Peleg L, Lerman-Sagie T, Zimran A, Elstein D, Navon R, Stern N, Valevski A | display-authors = 6 | title = Pyrimethamine increases β-hexosaminidase A activity in patients with Late Onset Tay Sachs | journal = Molecular Genetics and Metabolism | volume = 102 | issue = 3 | pages = 356–363 | date = March 2011 | pmid = 21185210 | doi = 10.1016/j.ymgme.2010.11.163 }}</ref> It is being evaluated in clinical trials as a treatment for [[amyotrophic lateral sclerosis]].<ref>{{ClinicalTrialsGov|NCT01083667|SOD1 Inhibition by Pyrimethamine in Familial Amyotrophic Lateral Sclerosis (ALS)}}</ref> |
||
== See also == |
== See also == |
||
* [[Sulfadoxine/pyrimethamine]] |
* [[Sulfadoxine/pyrimethamine]] |
||
==References== |
== References == |
||
{{Reflist}} |
{{Reflist}} |
||
==External links== |
== External links == |
||
{{Scholia|topic}} |
{{Scholia|topic}} |
||
* {{cite web | url = https://druginfo.nlm.nih.gov/drugportal/name/pyrimethamine | publisher = U.S. National Library of Medicine | work = Drug Information Portal | title = Pyrimethamine }} |
* {{cite web | url = https://druginfo.nlm.nih.gov/drugportal/name/pyrimethamine | publisher = U.S. National Library of Medicine | work = Drug Information Portal | title = Pyrimethamine }} |
||
| Line 207: | Line 207: | ||
[[Category:Aminopyrimidines]] |
[[Category:Aminopyrimidines]] |
||
[[Category:Antimalarial agents]] |
[[Category:Antimalarial agents]] |
||
[[Category: |
[[Category:Chlorobenzene derivatives]] |
||
[[Category:Drugs developed by GSK plc]] |
|||
[[Category:Medical controversies]] |
[[Category:Medical controversies]] |
||
[[Category:Protozoal dihydrofolate reductase inhibitors]] |
[[Category:Protozoal dihydrofolate reductase inhibitors]] |
||
Latest revision as of 01:38, 4 February 2024
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌpɪrɪˈmɛθəmɪn/ |
| Trade names | Daraprim, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601050 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | well-absorbed |
| Protein binding | 87% |
| Metabolism | Liver |
| Elimination half-life | 96 hours |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.331 |
| Chemical and physical data | |
| Formula | C12H13ClN4 |
| Molar mass | 248.71 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 233 to 234 °C (451 to 453 °F) |
| |
| |
| (verify) | |
Pyrimethamine, sold under the brand name Daraprim among others, is a medication used with leucovorin (leucovorin is used to decrease side effects of pyrimethamine; it does not have intrinsic anti-parasitic activity) to treat the parasitic diseases toxoplasmosis and cystoisosporiasis.[3][4] It is also used with dapsone as a second-line option to prevent Pneumocystis jiroveci pneumonia in people with HIV/AIDS.[3] It was previously used for malaria but is no longer recommended due to resistance.[3] Pyrimethamine is taken by mouth.[3]
Common side effects include gastrointestinal upset, severe allergic reactions, and bone marrow suppression.[3] It should not be used by people with folate deficiency that has resulted in anemia.[3] There is concern that it may increase the risk of cancer.[3] While occasionally used in pregnancy it is unclear if pyrimethamine is safe for the baby.[1] Pyrimethamine is classified as a folic acid antagonist.[3] It works by inhibiting folic acid metabolism and therefore the making of DNA.[3]
Pyrimethamine was discovered in 1952 and came into medical use in 1953.[3][5] It is on the World Health Organization's List of Essential Medicines.[6] It was approved as a generic in the United States in February 2020.[7]
Medical uses
Pyrimethamine is typically given with a sulfonamide and folinic acid.[8]
It is used for the treatment of toxoplasmosis, actinomycosis, and isosporiasis, and for the treatment and prevention of Pneumocystis jirovecii pneumonia.[3]
Toxoplasmosis
Pyrimethamine is also used in combination with sulfadiazine to treat active toxoplasmosis. The two drugs bind the same enzymatic targets as the drugs trimethoprim and sulfamethoxazole - dihydrofolate reductase and dihydropteroate synthase, respectively.[citation needed]
Pyrimethamine has also been used in several trials to treat retinochoroiditis.[9]
Pregnancy consideration
Pyrimethamine is labeled as pregnancy category C in the United States.[1][10] To date, not enough evidence on its risks in pregnancy or its effects on the fetus is available.[10][11]
Malaria
It is primarily active against Plasmodium falciparum, but also against Plasmodium vivax.[12] Due to the emergence of pyrimethamine-resistant strains of P. falciparum, pyrimethamine alone is seldom used now. In combination with a long-acting sulfonamide such as sulfadiazine, it was widely used, such as in Fansidar, though resistance to this combination is increasing.[12]
Contraindications
Pyrimethamine is contraindicated in people with folate-deficiency anaemia.[8]
Side effects
When higher doses are used, as in the treatment of toxoplasmosis, pyrimethamine can cause gastrointestinal symptoms such as nausea, vomiting, glossitis, anorexia, and diarrhea.[11][13] A rash, which can be indicative of a hypersensitivity reaction, is also seen, particularly in combination with sulfonamides.[11] Central nervous system effects include ataxia, tremors, and seizures.[13] Hematologic side effects such as thrombocytopenia, leukopenia, and anemia can also occur.[13]
Interactions
Other antifolate agents such as methotrexate and trimethoprim may potentiate the antifolate actions of pyrimethamine, leading to potential folate deficiency, anaemia, and other blood dyscrasias.[8]
Mechanism of action
Pyrimethamine interferes with the regeneration of tetrahydrofolic acid from dihydrofolate by competitively inhibiting the enzyme dihydrofolate reductase.[14] Tetrahydrofolic acid is essential for DNA and RNA synthesis in many species, including protozoa.[14] It has also been found to reduce the expression of SOD1, a key protein involved in amyotrophic lateral sclerosis.[15][16]
Other medications
Pyrimethamine is typically given with folinic acid and sulfadiazine.[10]
- Sulfonamides (e.g. sulfadiazine) inhibit dihydropteroate synthetase, an enzyme that participates in folic acid synthesis from para-aminobenzoic acid. Hence, sulfonamides work synergistically with pyrimethamine by blocking a different enzyme needed for folic acid synthesis.
- Folinic acid (leucovorin) is a folic acid derivative converted to tetrahydrofolate, the primary active form of folic acid, in vivo, without relying on dihydrofolate reductase. Folinic acid reduces side effects related to folate deficiency in the patient.
Mechanism of resistance
Resistance to pyrimethamine is widespread. Mutations in the malarial gene for dihydrofolate reductase may reduce its effectiveness.[17] These mutations decrease the binding affinity between pyrimethamine and dihydrofolate reductase via loss of hydrogen bonds and steric interactions.[18]
History
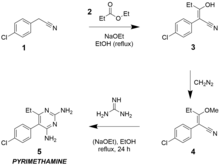
Nobel Prize-winning American scientist Gertrude Elion developed the drug at Burroughs-Wellcome (now part of GlaxoSmithKline) to combat malaria.[19] Pyrimethamine has been available since 1953.[20] In 2010, GlaxoSmithKline sold the marketing rights for Daraprim to CorePharma. Impax Laboratories sought to buy CorePharma in 2014, and completed the acquisition, including Daraprim, in March 2015.[21] In August 2015, the rights were bought by Turing Pharmaceuticals.[22] Turing subsequently became infamous for a price hike controversy when it raised the price of a dose of the drug in the U.S. market from US$13.50 to US$750, a 5,500% increase.[23]
Society and culture
Economics
In the United States in 2015, Turing Pharmaceuticals was criticized for increasing the price 50-fold, from US$13.50 to $750 a tablet,[24] leading to a cost of $75,000 for a course of treatment reported at one hospital.[25]
United States
In the United States, in 2015, with Turing Pharmaceuticals' acquisition of the US marketing rights for Daraprim tablets,[26] Daraprim became a single-source and specialty pharmacy item, and the price was increased.[27] The cost of a monthly course for a person on 75 mg dose rose to about $75,000/month at one hospital, or $750 per tablet while it was previously priced at $13.50.[28]
Outpatients could no longer obtain the medication from a community pharmacy, but only through a single dispensing pharmacy, Walgreens Specialty Pharmacy, and institutions could no longer order from their general wholesaler, but had to set up an account with the Daraprim Direct program.[27][29] Presentations from Retrophin, a company formerly headed by Martin Shkreli, CEO of Turing, from which Turing acquired the rights to Daraprim, suggested that a closed distribution system could prevent generic competitors from legally obtaining the drugs for the bioequivalence studies required for FDA approval of a generic drug.[29]
Shkreli defended the price hike by saying, "If there was a company that was selling an Aston Martin at the price of a bicycle, and we buy that company and we ask to charge Toyota prices, I don't think that that should be a crime."[30][31] As a result of the backlash, Shkreli hired a crisis public relations firm to help explain his fund's move.[32] Turing Pharmaceuticals announced on 24 November 2015, "that it would not reduce the list price of that drug after all", but they would offer patient assistance programs.[33] New York Times journalist Andrew Pollack noted that these programs "are standard for companies selling extremely high-priced drugs. They enable the patients to get the drug while pushing most of the costs onto insurance companies and taxpayers."[33]
The price increase was criticized by physician groups such as HIV Medicine Associates and Infectious Diseases Society of America.[34]
In 2016, a group of high school students from Sydney Grammar supported by the University of Sydney prepared pyrimethamine as an illustration that the synthesis is comparatively easy and the price-hike unjustifiable. His team produced 3.7 g for US$20, which would have been worth between US$35,000 and US$110,000 in the United States at the time.[35] Shkreli said the schoolboys were not competition, likely because the necessary bioequivalence studies require a sample of the existing medication provided directly by the company, and not simply purchased from a pharmacy, which Turing could decline to provide.[36][37] Nonetheless, the students' work was featured in The Guardian[36] and Time magazine,[38] and on ABC Australia,[35] the BBC,[37] and CNN.[39]
On 22 October 2015, Imprimis Pharmaceuticals announced it had made available compounded and customizable formulations of pyrimethamine and leucovorin in capsules to be taken by mouth starting as low as $99 for a 100-count bottle in the United States.[40] Pyrimethamine was approved as a generic in the United States in February 2020.[7]
In January 2020, the FTC filed a case against Vyera "alleging an elaborate anticompetitive scheme to preserve a monopoly for the life-saving drug, Daraprim".[41] A settlement was reached in December 2021. According to AP News, the settlement "requires Vyera and Phoenixus to provide up to $40 million in relief over 10 years to consumers who allegedly were fleeced by their actions and requires them to make Daraprim available to any potential generic competitor at the cost of producing the drug."[42] According to Law360, company executive Kevin Mulleady "agreed to a seven-year ban on working for or holding more than an 8% share in most pharmaceutical companies."[43]
Other countries
In India, multiple combinations of generic pyrimethamine are available for a price ranging from US$0.04 to US$0.10 each (3–7 rupees).[44][45][46][47]
In the UK, the same drug is available from GSK at a cost of US$20 (£13) for 30 tablets (about $0.66 each).[48]
In Australia, the drug is available in most pharmacies at a cost of US$9.35 (A$12.99) for 50 tablets (around US$0.18 each).[49]
In Brazil, the drug is available for R$0.07 a pill, or about US$0.02.[50]
In Switzerland, the drug is available for US$9.45 (CHF9.05) for 30 tablets (around US$0.32 a piece).[51]
Research
In 2011, researchers discovered that pyrimethamine can increase β-hexosaminidase activity, thus potentially slowing down the progression of late-onset Tay–Sachs disease.[52] It is being evaluated in clinical trials as a treatment for amyotrophic lateral sclerosis.[53]
See also
References
- ^ a b c "Pyrimethamine (Daraprim) Use During Pregnancy". Drugs.com. 31 July 2019. Archived from the original on 3 December 2016. Retrieved 28 February 2020.
- ^ "Daraprim Tablets – Summary of Product Characteristics (SmPC)". (emc). 19 February 2020. Archived from the original on 29 February 2020. Retrieved 28 February 2020.
- ^ a b c d e f g h i j k "Pyrimethamine". The American Society of Health-System Pharmacists. Archived from the original on 2 December 2016. Retrieved 2 December 2016.
- ^ Hamilton R (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 54. ISBN 9781284057560.
- ^ Sylvie M, Pierre C, Jean M (2008). Biodiversity of Malaria in the world. John Libbey Eurotext. p. 6. ISBN 9782742009633. Archived from the original on 14 January 2023. Retrieved 10 September 2017.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ a b "FDA Approves First Generic of Daraprim". U.S. Food and Drug Administration (FDA) (Press release). 28 February 2020. Archived from the original on 29 February 2020. Retrieved 28 February 2020.
- ^ a b c Rossi S, ed. (2013). Australian Medicines Handbook (2013 ed.). Adelaide: The Australian Medicines Handbook Unit Trust. ISBN 978-0-9805790-9-3.
- ^ Pradhan E, Bhandari S, Gilbert RE, Stanford M (May 2016). "Antibiotics versus no treatment for toxoplasma retinochoroiditis". The Cochrane Database of Systematic Reviews. 2016 (5): CD002218. doi:10.1002/14651858.CD002218.pub2. PMC 7100541. PMID 27198629.
- ^ a b c "Daraprim- pyrimethamine tablet". DailyMed. 31 August 2017. Archived from the original on 3 August 2020. Retrieved 28 February 2020.
- ^ a b c "Pyrimethamine | FDA Label - Tablet". AIDSinfo. Archived from the original on 18 January 2017. Retrieved 7 November 2016.
- ^ a b Brayfield A, ed. (13 December 2013). "Pyrimethamine". Martindale: The Complete Drug Reference. Pharmaceutical Press. Archived from the original on 29 August 2021. Retrieved 12 April 2014.
- ^ a b c "Daraprim Side Effects in Detail". www.drugs.com. Archived from the original on 8 November 2016. Retrieved 7 November 2016.
- ^ a b "Product Information Daraprim Tablets". TGA eBusiness Services. Aspen Pharmacare Australia Pty Ltd. 5 December 2011. p. 1. Archived from the original on 5 October 2016. Retrieved 12 April 2014.
- ^ Limpert AS, Mattmann ME, Cosford ND (2013). "Recent progress in the discovery of small molecules for the treatment of amyotrophic lateral sclerosis (ALS)" (PDF). Beilstein Journal of Organic Chemistry. 9: 717–732. doi:10.3762/bjoc.9.82. PMC 3678841. PMID 23766784. Archived (PDF) from the original on 13 April 2014.
- ^ Lange DJ, Andersen PM, Remanan R, Marklund S, Benjamin D (April 2013). "Pyrimethamine decreases levels of SOD1 in leukocytes and cerebrospinal fluid of ALS patients: a phase I pilot study". Amyotrophic Lateral Sclerosis & Frontotemporal Degeneration. 14 (3): 199–204. doi:10.3109/17482968.2012.724074. PMID 22985433. S2CID 39846211.
- ^ Gatton ML, Martin LB, Cheng Q (June 2004). "Evolution of resistance to sulfadoxine-pyrimethamine in Plasmodium falciparum". Antimicrobial Agents and Chemotherapy. 48 (6): 2116–2123. doi:10.1128/AAC.48.6.2116-2123.2004. PMC 415611. PMID 15155209.
- ^ Sirichaiwat C, Intaraudom C, Kamchonwongpaisan S, Vanichtanankul J, Thebtaranonth Y, Yuthavong Y (January 2004). "Target guided synthesis of 5-benzyl-2,4-diamonopyrimidines: their antimalarial activities and binding affinities to wild type and mutant dihydrofolate reductases from Plasmodium falciparum". Journal of Medicinal Chemistry. 47 (2): 345–354. doi:10.1021/jm0303352. PMID 14711307.
- ^ Vasudevan DM, Sreekumari S, Vaidyanathan K (2013). Textbook of Biochemistry for Medical Students. JP Medical Ltd. p. 491. ISBN 9789350905302. OCLC 843532694. Archived from the original on 8 September 2017. Retrieved 15 January 2016.
- ^ Cha AE (22 September 2015). "CEO who raised price of old pill more than $700 calls journalist a 'moron' for asking why". The Washington Post. Archived from the original on 23 September 2015.
- ^ Pollack A (20 September 2015). "Drug Goes From $13.50 a Tablet to $750, Overnight". The New York Times. Archived from the original on 16 December 2015. Retrieved 17 December 2015.
- ^ LaMattina J (21 September 2015). "Here's A Way For Pharma To Prevent Outrageous Generic Price Increases – And Help Its Reputation". Forbes. Archived from the original on 23 September 2015.
- ^ Kliff S (22 September 2015). "Vox Explainers: A Drug Company Raised a Pill's Price 5,500 Percent Because, in America, It Can". Vox. Archived from the original on 10 December 2015. Retrieved 9 December 2015.
- ^ Pollack A (20 September 2015). "Drug Goes From $13.50 a Tablet to $750, Overnight". The New York Times. Archived from the original on 16 December 2015. Retrieved 31 March 2020.
Turing immediately raised the price to $750 a tablet from $13.50, bringing the annual cost of treatment for some patients to hundreds of thousands of dollars.
- ^ Alpern JD, Song J, Stauffer WM (May 2016). "Essential Medicines in the United States--Why Access Is Diminishing". The New England Journal of Medicine. 374 (20): 1904–1907. doi:10.1056/nejmp1601559. PMID 27192669.
Heavy scrutiny followed, and although Turing agreed to reduce the price, the drug remains prohibitively expensive for many patients. Recently, at our hospital, an immigrant patient with a new diagnosis of HIV– AIDS and toxoplasmosis couldn't receive first-line therapy because of cost: the price for 100 pills was $75,000.
- ^ "Turing Pharmaceuticals AG Acquires U.S. Marketing Rights to Daraprim (pyrimethamine)". Turing Pharmaceuticals AG. PR Newswire Association LLC. 10 August 2015. Archived from the original on 22 September 2015.
- ^ a b Mahoney MV (17 July 2015). "New Pyrimethamine Dispensing Program: What Pharmacists Should Know". PharmacyTimes. Archived from the original on 6 September 2015.
- ^ Pollack A (20 September 2015). "Drug Goes From $13.50 a Tablet to $750, Overnight". The New York Times. Archived from the original on 25 September 2015. Retrieved 21 September 2015.
- ^ a b Lowe D (11 September 2014). "The Most Unconscionable Drug Price Hike I Have Yet Seen". In the Pipeline. Archived from the original on 7 June 2016.
- ^ Ramsey L (22 September 2015). "A pharma CEO tried to defend his decision to jack up the price of a critical drug by 5,000% – and it backfired". Business Insider. Archived from the original on 10 October 2015.
- ^ "Company hikes price of popular drug". Reuters. 22 September 2015. Archived from the original on 2 October 2015.
- ^ Tannahill J (9 October 2015). "PR Man Allan Ripp Representing The "Most Hated Man in America"". EverythingPR. Archived from the original on 14 October 2015.
- ^ a b Pollack A (24 November 2015). "Turing Refuses to Lower List Price of Toxoplasmosis Drug". New York Times. Archived from the original on 8 September 2017. Retrieved 25 November 2015.
- ^ "Letter to Tom Evegan and Kevin Burnier" (PDF). Archived from the original (PDF) on 23 September 2015. Retrieved 23 September 2015.
- ^ a b Hunjan R (30 November 2016). "Daraprim drug's key ingredient recreated by high school students in Sydney for just $20". ABC News. Archived from the original on 4 April 2019. Retrieved 23 April 2019.
- ^ a b Davey M (1 December 2016). "Australian students recreate Martin Shkreli price-hike drug in school lab". The Guardian. Archived from the original on 1 December 2016. Retrieved 1 December 2016.
- ^ a b Dunlop G (1 December 2016). "Australian boys recreate life-saving drug". BBC News. Archived from the original on 1 December 2016. Retrieved 1 December 2016.
- ^ Lui K (2 December 2016). "Watch Martin Shkreli Respond to the School Kids Who Recreated His Drug for $2 a Dose". Time. Archived from the original on 3 November 2020. Retrieved 23 April 2019.
- ^ Roberts E (1 December 2016). "'Pharma Bro' Martin Shkreli meets his match in a group of Australian schoolboys". CNN. Archived from the original on 23 April 2019. Retrieved 23 April 2019.
- ^ "Express Scripts, Imprimis to offer $1 Daraprim alternative". CNBC. December 2015. Archived from the original on 7 August 2020. Retrieved 1 April 2020.
- ^ "Vyera Pharmaceuticals, LLC". Federal Trade Commission. 27 January 2020. Archived from the original on 9 December 2021. Retrieved 9 December 2021.
- ^ "'Pharma Bro' firm reaches $40M settlement in gouging case". AP News. 8 December 2021. Archived from the original on 9 December 2021. Retrieved 9 December 2021.
- ^ Koenig B (7 December 2021). "Former Shkreli Co. Inks Deal Worth Up To $40M With FTC, AGs - Law360". Law360. Archived from the original on 9 December 2021. Retrieved 9 December 2021.
- ^ Miseta E (29 September 2015). "High Drug Prices: Should We Blame Pharma Or The FDA?". Clinical Leader. Archived from the original on 30 September 2015.
- ^ "Medline India – Sulfadoxine with Pyrimethamine". www.medlineindia.com. Archived from the original on 24 September 2015. Retrieved 22 September 2015.
- ^ "It is Cheaper for an American patient to fly out to India and buy a year's supply of the medication than buy a single Daraprim tablet in the US". 25 September 2015. Archived from the original on 28 September 2015.
- ^ "There is no reason why the United States cannot have as vigorous a market in generic pharmaceuticals as does India". 24 September 2015. Archived from the original on 6 October 2015.
- ^ Roberts M (22 September 2015). "What's a fair price for a drug?". BBC News. Archived from the original on 24 September 2015. Retrieved 23 September 2015.
- ^ "Chemist Warehouse". www.chemistwarehouse.com.au. Archived from the original on 22 December 2015. Retrieved 12 December 2015.
- ^ "Remédio que teve aumento de 5.000% nos EUA custa R$ 0,07 no Brasil (e não vai aumentar)". brasilpost.com.br. Archived from the original on 25 September 2015. Retrieved 23 September 2015.
- ^ "Swiss Compendium information about Daraprim". compendium.ch. Archived from the original on 29 August 2021. Retrieved 1 September 2017.
- ^ Osher E, Fattal-Valevski A, Sagie L, Urshanski N, Amir-Levi Y, Katzburg S, et al. (March 2011). "Pyrimethamine increases β-hexosaminidase A activity in patients with Late Onset Tay Sachs". Molecular Genetics and Metabolism. 102 (3): 356–363. doi:10.1016/j.ymgme.2010.11.163. PMID 21185210.
- ^ Clinical trial number NCT01083667 for "SOD1 Inhibition by Pyrimethamine in Familial Amyotrophic Lateral Sclerosis (ALS)" at ClinicalTrials.gov
External links
- "Pyrimethamine". Drug Information Portal. U.S. National Library of Medicine.
