m →Analysis: ref regarding structural elucidation |
I have raised a significant concern about this and related pages. I'd ask note to stay until someone moves material from here to "phenols" page, leaving this to truly cover polyphenols. Prof D |
||
| Line 1: | Line 1: | ||
Author note: |
|||
[[File:Quercetin.svg|thumb|[[Quercetin]], a typical flavonoid, is a polyphenol.]] |
|||
The titling of this section needs to be discussed at the level of a wikipedia administrator/bureaucrat, because it and its graphical and text content mistakenly and thoroughly ambiguate plant-derived "simple" [[phenols]] of all sorts with the truly higher molecular weight, more complex polyphenols. Polyphenols are true to their name in having "many" (e.g., 12-16) phenolic substructures -- as opposed to the 1-4 phenolic substructures currently displayed on this page (range 1-4, median = mean = 2.5). See, for instance, S. Quideau et al, 2011.<ref>Plant polyphenols: chemical properties, biological activities, and synthesis. Quideau S, Deffieux D, Douat-Casassus C, Pouységu L, Chromatographia, Volume 60, Supplement 1, S93-S100, Angew Chem Int Ed Engl. 2011, 50(3):586-621; doi: 10.1002/anie.201000044.</ref> |
|||
Hence, [[quercetin]] is not by any measure a typical polyphenol, nor are [[phenol]], hydroquinone, or other simple structures currently pictured in this article. In confusing these matters, the current article distorts the relationship between structure and biological function; all chemicals presenting a phenolic group are not polyphenols, and certainly do not afford the health benefits associated with, for instance, tea and wine polyphenol components. Phenol itself caustic to external exposure, and quite toxic if taken internally. As structural complexity increases as one proceeds from phenol, to simpler di-,tri- and oligophenols, through to polyphenols, one engages with industrial dyes, toxins, plant pigments, drugs, and an array of other functional classes, in addition to medically interesting and beneficial simple, oligo-, and poly-phenols. |
|||
The fact that the web literatures on nutriceuticals propagate the ambiguities to an extraordinary degree, and that less chemically informed or less rigorous scientific venues also fail to make structure-activity distinctions -- these underlie but do not excuse the confusion that the current page now propagates. Hence, the subject of this article needs to be reviewed alongside these two other other pages -- ("[[phenols]]" and "[phenol]]" -- and structural distinctions that underlie functional differences vis-a-vis in health, nutrition, and medicine need to be made, so that readers can draw appropriate practical information and understanding. |
|||
A '''polyphenol''' (noun, pronunciation \ˌpä-lē-ˈfē-ˌnōl, -fi-ˈ\<ref name=merriam-webster>[http://www.merriam-webster.com/dictionary/polyphenol Polyphenol on www.merriam-webster.com online dictionary]</ref>) is a member of a loosely defined group of chemical substances also known as ''phenoloids'',<ref>Detection of phenoloids in some Hungarian Inula and Centaurea species. A. Péter and G. Dósa, Acta Botanica Hungarica, Volume 44, Numbers 1-2 / March 2002, Pages 129-135, {{doi|10.1556/ABot.44.2002.1-2.9}}</ref><ref>LC-MS Analysis of Antioxidant Plant Phenoloids. I. Papp, P. Apáti, V. Andrasek, A. Blázovics, A. Balázs, L. Kursinszki, G. C. Kite, P. J. Houghton and Á. Kéry, Chromatographia, Volume 60, Supplement 1, S93-S100, {{doi|10.1365/s10337-004-0348-z}}</ref><ref>[http://cat.inist.fr/?aModele=afficheN&cpsidt=15401411 New chlorine-containing phenoloid from Curculigo capitulata. Ning Li, Tan Ning-Hua and Jun Zhou, Journal of Asian natural products research, 2004, vol. 6, no 1, pp. 7-10]</ref> ''phenols'' or ''[[phenolic]]s'', sometimes abbreviated PP. Occurring naturally and having biological functions, they are considered [[list of biomolecules|biomolecules]] which, in [[plant]]s, defines them as [[phytochemical]]s. Their number in nature is estimated to be several thousand different structures. For example, on [http://www.metabolome.jp/download/flavonoid/ FlavonoidViewer.jp], there are 6850 flavonoids sorted by mass. |
A '''polyphenol''' (noun, pronunciation \ˌpä-lē-ˈfē-ˌnōl, -fi-ˈ\<ref name=merriam-webster>[http://www.merriam-webster.com/dictionary/polyphenol Polyphenol on www.merriam-webster.com online dictionary]</ref>) is a member of a loosely defined group of chemical substances also known as ''phenoloids'',<ref>Detection of phenoloids in some Hungarian Inula and Centaurea species. A. Péter and G. Dósa, Acta Botanica Hungarica, Volume 44, Numbers 1-2 / March 2002, Pages 129-135, {{doi|10.1556/ABot.44.2002.1-2.9}}</ref><ref>LC-MS Analysis of Antioxidant Plant Phenoloids. I. Papp, P. Apáti, V. Andrasek, A. Blázovics, A. Balázs, L. Kursinszki, G. C. Kite, P. J. Houghton and Á. Kéry, Chromatographia, Volume 60, Supplement 1, S93-S100, {{doi|10.1365/s10337-004-0348-z}}</ref><ref>[http://cat.inist.fr/?aModele=afficheN&cpsidt=15401411 New chlorine-containing phenoloid from Curculigo capitulata. Ning Li, Tan Ning-Hua and Jun Zhou, Journal of Asian natural products research, 2004, vol. 6, no 1, pp. 7-10]</ref> ''phenols'' or ''[[phenolic]]s'', sometimes abbreviated PP. Occurring naturally and having biological functions, they are considered [[list of biomolecules|biomolecules]] which, in [[plant]]s, defines them as [[phytochemical]]s. Their number in nature is estimated to be several thousand different structures. For example, on [http://www.metabolome.jp/download/flavonoid/ FlavonoidViewer.jp], there are 6850 flavonoids sorted by mass. |
||
Revision as of 23:31, 1 February 2011
Author note:
The titling of this section needs to be discussed at the level of a wikipedia administrator/bureaucrat, because it and its graphical and text content mistakenly and thoroughly ambiguate plant-derived "simple" phenols of all sorts with the truly higher molecular weight, more complex polyphenols. Polyphenols are true to their name in having "many" (e.g., 12-16) phenolic substructures -- as opposed to the 1-4 phenolic substructures currently displayed on this page (range 1-4, median = mean = 2.5). See, for instance, S. Quideau et al, 2011.[1]
Hence, quercetin is not by any measure a typical polyphenol, nor are phenol, hydroquinone, or other simple structures currently pictured in this article. In confusing these matters, the current article distorts the relationship between structure and biological function; all chemicals presenting a phenolic group are not polyphenols, and certainly do not afford the health benefits associated with, for instance, tea and wine polyphenol components. Phenol itself caustic to external exposure, and quite toxic if taken internally. As structural complexity increases as one proceeds from phenol, to simpler di-,tri- and oligophenols, through to polyphenols, one engages with industrial dyes, toxins, plant pigments, drugs, and an array of other functional classes, in addition to medically interesting and beneficial simple, oligo-, and poly-phenols.
The fact that the web literatures on nutriceuticals propagate the ambiguities to an extraordinary degree, and that less chemically informed or less rigorous scientific venues also fail to make structure-activity distinctions -- these underlie but do not excuse the confusion that the current page now propagates. Hence, the subject of this article needs to be reviewed alongside these two other other pages -- ("phenols" and "[phenol]]" -- and structural distinctions that underlie functional differences vis-a-vis in health, nutrition, and medicine need to be made, so that readers can draw appropriate practical information and understanding.
A polyphenol (noun, pronunciation \ˌpä-lē-ˈfē-ˌnōl, -fi-ˈ\[2]) is a member of a loosely defined group of chemical substances also known as phenoloids,[3][4][5] phenols or phenolics, sometimes abbreviated PP. Occurring naturally and having biological functions, they are considered biomolecules which, in plants, defines them as phytochemicals. Their number in nature is estimated to be several thousand different structures. For example, on FlavonoidViewer.jp, there are 6850 flavonoids sorted by mass.
Problem of a comprehensive definition
Different definitions of the chemical nature of polyphenols have been made so far. The word is formed from poly-, from the Ancient Greek word πολύς (polus, meaning “many, much”) and the word phenol which is a molecule formed by a phenyl (-C6H5) group bonded to a hydroxyl (-OH) group.
On Merriam Webster and Babylon on line dictionaries, they are defined as molecules with more than one hydroxyl group on one or more phenol unit per molecule.[2][6] On the free dictionary online or on wiktionary, they are alcohols containing two or more benzene rings that each have at least one hydroxyl group attached.[7] Other sources give definitions insisting on their plant origin, antioxidant properties or health benefits.[8][9] The term has been in use since 1894 according to the Merriam-Webster 's International scientific vocabulary.[2]
Edwin Haslam proposed a definition of plant polyphenols based on the earlier proposals of Bate-Smith, Swain and White, which includes specific structural characteristics common to all phenolics having a tanning property. We could thus refer to it as the White–Bate-Smith–Swain–Haslam (WBSSH) definition.[10] According to this definition, a polyphenol is "a water-soluble phenolic compounds having molecular masses between 500 to 3000-4000 Da, possessing 12 to 16 phenolic hydroxyl groups and five to seven aromatic rings per 1000 relative molecular mass, giving the usual phenolic reactions such as the formation of intense blue-black complexes upon treatment with iron(III) salts, and expressing special properties such as the ability to precipitate alkaloids and proteins." It can be noted that this definition corresponds well with that of tannins.
The term "polyphenol" has been entered in the Medical Subject Headings (MeSH) database[11] of the National Library of Medicine in 1980 citing an article about sclerotization in the Surinam cockroach (Pycnoscelus surinamensis).[12] In the "note" section of the entry, it is written "GEN only; use the precise structure header, most commonly in the FLAVONOIDS group; this term only refers vaguely to phenolic (aromatic) hydroxyls; provinols are polyphenols from red wine".
In a limited sense, polyphenols are natural molecules, produced mostly by plants, while in a broader acceptance, the term polyphenols can be applied to all types of molecules containing phenols units including synthetic molecules. It can in this way also apply to polyphenolic proteins.
Chemistry
Polyphenols are normally made of the three elements carbon, hydrogen and oxygen. More than one phenolic subcomponents (benzenediols, benzenetriols) can be present on a given polyphenol.
 |
 |
 |
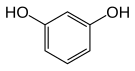 |
 |
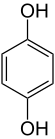 |
| Examples: coumaric acid derived lignins, kaempferol | Examples: catechin, quercetin, caffeic and ferulic acid derived lignins, hydroxytyrosol esters | Examples: gallocatechins (EGCG), tannins, myricetin, sinapyl alcohol derived lignins | Examples: resveratrol | Examples: almost all flavonoids | Examples: arbutin |
Polyphenols can also have other substituents than hydroxyl groups.
The molecules may contain heterocycles with the heteroatom being oxygen. Lactones (cyclic esters) can also be found. Polyphenol may contain a macrocycle as in the molecule cavicularin isolated from the liverwort Cavicularia densa.
In the case of polyphenols comprising two or more benzenic rings, a special interest can be assigned to the linker (molecular spacer) between those phenol groups. A direct carbon-carbon diphenyl link can be observed in some polyphenol, like hexahydroxydiphenic acid. In the case of depsides, the linker is made of a ester bond. Polyphenols can also contain benzene rings linked in a naphthalene structure like in the case of gossypol. Molecules comprising a furan cycle, like furanoflavonoids, or a tropone or tropolone cycle, like purpurogallin or theaflavin, also possess an extra non-benzenic aromatic ring.
Regarding degree of unsaturation, polyphenols follow the general "rings plus double bonds formulation" :
But as they generally do not contain nitrogen (N) nor halogens (X), the formula can be simplified in :
where
- R is the rings count as one degree of unsaturation
- DB is the double bonds count as one degree of unsaturation
- Triple bonds count as two degrees of unsaturation
- C is the number of carbons
- H is the number of hydrogens
Chemical properties
The majority of these compounds are solubles molecules but the smaller polyphenols can be volatiles.
Polyphenols are molecules producing autofluorescence, especially lignin and the phenolic part of suberin. Polyphenols spectral data show a typical UV absorbance characteristic of benzene aromaticity at 270 nm. However, according to Woodward's rules, bathochromic shifts often happen suggesting the presence of delocalised π electrons arising from a conjugation between the benzene and vinyls groups.[13]
Polyphenols are reactive species. A complex mixture of polyphenols, found in food for example, can undergo autoxidation during the ageing process. Simple polyphenols can lead to the formation of B-type procyanidins in wines[14] or in model solutions.[15][16] This is correlated to the non enzymatic browning color change characteristic of this process.[17] This phenomenon is also observed in foods like carrot purees.[18] ABTS may be used to characterise polyphenol oxidation products[19].
Many polyphenols present chirality within their molecule. An example of such molecules is catechin. Cavicularin is a unusual macrocycle because it was the first compound isolated from nature displaying optical activity due to the presence of planar chirality and axial chirality.
Polyphenols chemically interact with many other substances. Stacking, a chemical property of molecules with aromaticity, is seen occurring between polyphenols molecules. When studied in mass spectrometry, polyphenols easily form adduct ions with halogens. They can also interact with proteins (case of tannins) and other food matrices or with different forms of silica (mesoporous silica, fumed silica[20] or silica-based sol gels[21]).
Classification and nomenclature
In terms of chemical classification, polyphenols are generally divided into hydrolyzable tannins (gallic acid esters of glucose and other sugars or cyclitols) and phenylpropanoids, such as lignins, flavonoids, and condensed tannins. This division is derived from the variety of simple polyphenolic units derived from secondary plant metabolism as well as classical divisions based upon the relative importance of each base component to different fields of study. Tannin chemistry originated in the importance of tannic acid to the tanning industry; lignins to the chemistry of soil and plant structure; and flavonoids to the chemistry of plant secondary metabolites for plant defense, and flower color (e.g. from anthocyanins).
The largest and best studied polyphenols are the flavonoids, which include several thousand compounds, among them the flavonols, flavones, flavan-3ol (catechins), flavanones, anthocyanidins and isoflavonoids.[22]
| Base Unit: |  |
 |
 |
|---|---|---|---|
| Class/Polymer: | Hydrolyzable tannins | Flavonoid, Condensed tannins |
There is a classification based on number of carbons based on Harborne et al., published in 1980:[23]
| Number of carbon atoms | Basic skeleton | Class | Examples |
|---|---|---|---|
| 6 | C6 | Simple phenols, Benzoquinones | Catechol, Hydroquinone, 2,6-Dimethoxybenzoquinone |
| 7 | C6-C1 | Phenolic acids, Phenolic aldehydes | Gallic, salicylic acids |
| 8 | C6-C2 | Acetophenones, Tyrosine derivatives , Phenylacetic acids | 3-Acetyl-6-methoxybenzaldehyde, Tyrosol, p-Hydroxyphenylacetic acid |
| 9 | C6-C3 | Hydroxycinnamic acids, Phenylpropenes, Coumarins, Isocoumarins, Chromones | Caffeic, ferulic acids, Myristicin, Eugenol, Umbelliferone, aesculetin, Bergenon, Eugenin |
| 10 | C6-C4 | Naphthoquinones | Juglone, Plumbagin |
| 13 | C6-C1-C6 | Xanthonoids | Mangiferin |
| 14 | C6-C2-C6 | Stilbenoids, Anthraquinones | Resveratrol, Emodin |
| 15 | C6-C3-C6 | Chalcones, Flavonoids, Isoflavonoids, Neoflavonoids | Quercetin, cyanidin, Genistein |
| 18 | (C6-C3)2 | Lignans, Neolignans | Pinoresinol, Eusiderin |
| 30 | (C6-C3-C6)2 | Biflavonoids | Amentoflavone |
| n | (C6-C3)n, (C6)n, (C6-C3-C6)n | Lignins, Catechol melanins, Flavolans (Condensed tannins) |
The phenolic unit can be found dimerized or further polymerized, creating a new class of polyphenol. For example, ellagic acid is a dimer of gallic acid and forms the class of ellagitannins, or a catechin and a gallocatechin can combine to form the red compound theaflavin, a process which also results in the large class of brown thearubigins in tea.
Two polyphenols from two different categories, for instance a flavonoid and a lignan, can combine to form a hybrid class like the flavonolignans.
Chemical uses
Polyphenols, especially, tannins, can be used as precursors in green chemistry[24] notably to produce plastics or resins by polymerisation with[25] or without the use of formaldehyde[26] or adhesives for particleboards.[27]
Polyphenols are also used for the production of creosote to treat wood.
The aims are generally to make use of plant residues from grape, olive (called pomaces) or pecan shells left after processing.
Some polyphenols can be used as dyes or drugs. For instance, in the Indian subcontinent's ancient Ayurveda system of medicine, the pomegranate has extensively been used as a source of traditional remedies for thousands of years. The pomegranate peel, high in tannins and other polyphenols, or its juice, is employed in the dyeing of non-synthetic fabrics.[28]
Polyphenols can be synthetised chemically from phenol to produce a mixture of phenylene and oxyphenylene.[29]
Biology
Occurrence in nature
The most abundant polyphenols are the condensed tannins, found in virtually all families of plants, and comprising up to 50% of the dry weight of leaves. Phenolic compounds can also be found in mushroom basidiomycetes species[30] like Boletus edulis.
Some polyphenols produced by plants in case of pathogens attacks are called phytoalexins. Such compounds can be implied in the hypersensitive response of plants. High levels of polyphenols in some woods can explain their natural preservation against rot.[31]
Cucumbers grown on vermicompost may be less susceptible to striped cucumber beetle attack due to a higher induced level of polyphenols in the plant.
Polyphenols can be involved in allelopathic interactions in soil[32][33] or in water.[34] Juglone is an example of such a molecule inhibiting the growth of other plant species around walnut trees. The fruit pulp of the plant Liriope muscari contains phenolic compounds which inhibit its own seeds germination.
Polyphenols can be a source of pollution in sites near processing plants producing olive oil, coffee (see coffee wastewater) or paper. Laccases (found for instance in the fungal species Panellus stipticus) can be used in bioremediation.
Polyphenol content can be an element of chemotaxonomy.[35]
Furanocoumarins are phenolic and are non-toxic until activated by light. Furancoumarins block the transcription and repair of DNA. Therefore, they are considered phytotoxins.
Polyphenols can also be found in animals. In arthropods like insects[36] and crustaceans[37] polyphenols play a role in epicuticle hardening (sclerotization). Pryor showed in 1940 that the hardening of the protein component of insect cuticle is due to the tanning action of an agent produced by oxidation of a phenolic substance. In the analogous hardening of the cockroach ootheca, Pryor, Russell & Todd showed in 1946 that the phenolic substance concerned is 3:4-dihydroxybenzoic acid[38] (protocatechuic acid). The hardening of the cuticle is due to the presence of a polyphenol oxidase.[39] In crustaceans, there is a second oxidase activity leading to cuticle pigmentation.[40] There is apparently no polyphenol tanning occurring in arachnids cuticle.[41]
Biological uses
Furanoflavonoids like karanjin or rotenoids are used as acaricide or insecticide.
Metabolism
Biosynthesis
Some polyphenols are derived from the variety of simple polyphenolic units derived from secondary plant metabolism of the shikimate pathway[42]
Polyphenol oxidase (PPO) is an enzyme that catalyses the oxidation of o-diphenols to produce o-quinones. It is the rapid polymerisation of o-quinones to produce black, brown or red polyphenolic pigments that is the cause of fruit browning. In insects, PPO serves for the cuticle hardening.[43]
Laccase is a major enzyme that initiates the cleavage of hydrocarbon rings, which catalyzes the addition of a hydroxyl group to phenolic compounds. This enzyme can be found in fungi like Panellus stipticus, organisms able to break down lignin, a complex aromatic polymer in wood that is highly resistant to degradation by conventional enzyme systems.
Anthracyclines or hypericin[44] are derived from polyketides cyclisation.[45]
Glycosylation, acetylation and conjugation
In plants, the phenolic units are esterified or methylated.
The polyphenols are submitted to conjugaison. Many polyphenols are found in the glycoside form instead of the aglycone form. This glycosylation increases their solubility.[46]
In olive oil, tyrosol forms esters with fatty acids.[47]
Some acetylations involve terpenes like geraniol.[48]
Methylations can occur by the formation of an ether bond on hydroxyl groups forming O-methylated polyphenols. In the case of the O-methylated flavone tangeritin, all of the five hydroxyls are methylated, leaving no free hydroxyls of the phenol group. Methylations can also occur on directly on a carbon of the benzene ring like in the case of poriol, a C-methylated flavonoid.
In animals and humans, after ingestion, polyphenols become part of the xenobiotic metabolism. In subsequent phase II reactions, these activated metabolites are conjugated with charged species such as glutathione, sulfate, glycine or glucuronic acid. These reactions are catalysed by a large group of broad-specificity transferases.
Content in food

Polyphenols are considered antinutrients, compounds that interfere with the absorption of nutrients as a mechanism of plant defense against herbivores. Leaf protein concentrates used as animal food can also be rich in polyphenols.[50] Animals produce salivary proteins, like protein IB5, as herbivore adaptations to plant defense, to counter the tannins astringent effect. Some animals like the greater glider or the koala can digest low nutrient foliage, specifically eucalypt leaf matter, which contains a variety of phenolic and terpenoid compounds and a high concentration of lignified fiber. Some frugivores like birds are reported to not sense astringency.
In human food

Notable sources of polyphenols include berries, tea, beer, olive oil, chocolate / cocoa, coffee, pomegranates, popcorn, yerba mate, fruits and fruit based drinks (including cider and wine) and vegetables. Herbs and spices, nuts (walnuts, peanut) and algae are also potentially significant for supplying certain polyphenols. Such foods containing polyphenols are generally considered as health food.
Polyphenols can also be found in fatty matrices like olive oil.[51] Cloudy olive oil has the higher levels of polyphenols, or polar phenols that form a complex polyphenol-protein complex.
Polyphenols from algae (phlorotannins) may be used as additives to prevent lipid oxidation during fish preservation.[52]
Role of processing in phenolic composition
Phenolic and carotenoid compounds with antioxidant properties in vegetables have been found to be retained significantly better through steaming than through frying.[53]
Polyphenols in wine, beer and various nonalcoholic juice beverages can be removed using finings, substances that are usually added at or near the completion of the processing of brewing.
Marketing argument
The presence of polyphenols is a marketing argument to sell functional foods, dietary supplements or anti-aging creams.
Functional foods may contain polyphenols. For superfruit beverages, which may include extracts from fruits like açai or pomegranate, the detailed composition of polyphenols is usually not revealed on the nutrition label. Instead, there may be an ORAC value given for the in vitro antioxidant capacity of the product. Polyphenols-enriched drinks may actually deliver the intended blend of bioavailable polyphenols, which would normally require consumption of several different plant-derived foods.[54]
As a matter of pharmacovigilance, health benefits from using these products have not been scientifically confirmed or approved by regulatory authorities and may only be supported by preliminary research. Accordingly, there are no recommended Dietary Reference Intake levels established for polyphenols as exist for essential nutrients.
Potential health effects
Research on polyphenols
Bioavailability
Questions on the relationship between health benefits and polyphenols generally revolve around bioavailability.[55] Gallic acid and isoflavones are the most well-absorbed polyphenols, followed by catechins, flavanones, and quercetin glucosides, but with different kinetics. The least well-absorbed polyphenols are the proanthocyanidins, galloylated tea catechins, and anthocyanins.[56]
Compared with the effects of polyphenols in vitro, the effects in vivo, although the subject of ongoing research, are limited and vague. The reasons for this are 1) the absence of validated in vivo biomarkers, especially for inflammation or carcinogenesis; 2) long-term studies failing to demonstrate effects with a mechanism of action, specificity or efficacy; and 3) invalid applications of high, unphysiological test concentrations in the in vitro studies, which are subsequently irrelevant for the design of in vivo experiments.[57]
To overcome this problem of bioavailability, an alternative could be liposome encapsulation. For instance, liposomes have been prepared encapsulating baicalin[58] and tested in mice.[59]
In rats, polyphenols absorbed in the small intestine[60] may be bound in protein-polyphenol complexes modified by intestinal microflora enzymes,[61] allowing derivative compounds formed by ring-fission to be better absorbed.[62][63]
Matrix effect
The poor bioavailability of polyphenols in vivo may be due to a matrix effect. Casein found in milk added to a polyphenol-rich food like tea may reduce the absorbed polyphenols content[64] by the formation of a complex as demonstrated in vitro.[65][66] Moreover other substances such as caffeine can form insoluble complexes with polyphenols.[67]
Polyphenols may also interact with fibers like pectins and have a positive effect in large intestine accessibility.[68]
Analysis
The analysis techniques are those of phytochemistry : extraction, isolation, structural elucidation[69], then quantification.
Extraction
Extraction of polyphenols[70] can be performed using a solvent like water, hot water, methanol, methanol/formic acid, methanol/water/acetic or formic acid etc. Liquid liquid extraction can be also performed or countercurrent chromatography. Solid phase extraction can also be made on C18 sorbent cartridges. Other techniques are ultrasonic extraction, heat reflux extraction, microwave-assisted extraction,[71] critical carbon dioxide,[72] pressurized liquid extraction[73] or use of ethanol in an immersion extractor.[74] The extraction conditions (temperature, extraction time, ratio of solvent to raw material, solvent and concentrations) have to be optimized.
Mainly found in the fruit skins and seeds, high levels of polyphenols may reflect only the measured extractable polyphenol (EPP) content of a fruit which may also contain non-extractable polyphenols.[75]
Concentration can be made by ultrafiltration.[76] Purification can be achieved by preparative chromatography.
Analysis techniques

Phosphomolybdic acid is used as a reagent for staining phenolics in thin layer chromatography. Polyphenols can be studied by the mean of spectroscopy, especially in the ultra violet domain, by fractionation or paper chromatography. They can also be analysed by chemical characterisation.
Instrumental chemistry analyses include separation by high performance liquid chromatography (HPLC), and especially by reversed-phase liquid chromatography (RPLC), can be coupled to mass spectrometry. Purified compounds can be identified by the mean of nuclear magnetic resonance.
pKa of phenolic compounds can be calculated from the retention time in liquid chromatography.[77] In the same way, pKas from phenolic acids can thus be calculated[78]
Microscopy analysis
The DMACA reagent is an histological dye specific to polyphenols used in microscopy analyses. The autofluorescence of polyphenols can also be used, especially for localisation of lignin and suberin.
Quantification
A method for polyphenolic content quantification is volumetric titration. An oxidizing agent, permanganate, is used to oxidize known concentrations of a standard tannin solution, producing a standard curve. The tannin content of the unknown is then expressed as equivalents of the appropriate hydrolyzable or condensed tannin.[79]
Some methods for quantification of total polyphenol content are based on colorimetric measurements. Some tests are relatively specific to polyphenols (for instance the Porter's assay). Total phenols (or antioxidant effect) can be measured using the Folin-Ciocalteu reaction. Results are typically expressed as gallic acid equivalents. Polyphenols are seldom evaluated by antibodies technologies.[80]
Other tests measure the antioxidant capacity of a fraction. Some make use of the ABTS radical cation which is reactive towards most antioxidants including phenolics, thiols and vitamin C.[81] During this reaction, the blue ABTS radical cation is converted back to its colorless neutral form. The reaction may be monitored spectrophotometrically. This assay is often referred to as the Trolox equivalent antioxidant capacity (TEAC) assay. The reactivity of the various antioxidants tested are compared to that of Trolox, which is a vitamin E analog.
Other antioxidant capacity assays which use Trolox as a standard include the diphenylpicrylhydrazyl (DPPH), oxygen radical absorbance capacity (ORAC),[82] ferric reducing ability of plasma (FRAP)[83] assays or inhibition of copper-catalyzed in vitro human low-density lipoprotein oxidation.[84]
New methods including the use of biosensors can help monitor the content of polyphenols in food.[85]
Quantitation results produced by the mean of diode array detector-coupled HPLC are generally given as relative rather than absolute values as there is a lack of commercially available standards for every polyphenolic molecules.
Other techniques
Polyphenols can be studied in model solution or even by computational simulation.[86]
Chemometrics analyses on acquired data can be performed to compare samples from different origins.
Genetic analysis
Polyphenols metabolic pahways and enzymes can be studied by mean of transgenesis of genes from one plant species to another.[87]
In Vitis vinifera cells suspensions, polyphenol production is regulated by sugars levels.[88]
References
- ^ Plant polyphenols: chemical properties, biological activities, and synthesis. Quideau S, Deffieux D, Douat-Casassus C, Pouységu L, Chromatographia, Volume 60, Supplement 1, S93-S100, Angew Chem Int Ed Engl. 2011, 50(3):586-621; doi: 10.1002/anie.201000044.
- ^ a b c Polyphenol on www.merriam-webster.com online dictionary
- ^ Detection of phenoloids in some Hungarian Inula and Centaurea species. A. Péter and G. Dósa, Acta Botanica Hungarica, Volume 44, Numbers 1-2 / March 2002, Pages 129-135, doi:10.1556/ABot.44.2002.1-2.9
- ^ LC-MS Analysis of Antioxidant Plant Phenoloids. I. Papp, P. Apáti, V. Andrasek, A. Blázovics, A. Balázs, L. Kursinszki, G. C. Kite, P. J. Houghton and Á. Kéry, Chromatographia, Volume 60, Supplement 1, S93-S100, doi:10.1365/s10337-004-0348-z
- ^ New chlorine-containing phenoloid from Curculigo capitulata. Ning Li, Tan Ning-Hua and Jun Zhou, Journal of Asian natural products research, 2004, vol. 6, no 1, pp. 7-10
- ^ Polyphenol on dictionary.babylon.com on line dictionary
- ^ Polyphenol definition on www.thefreedictionary.com online dictionary
- ^ Polyphenol on www.cancer.gov/dictionary
- ^ Polyphenol on www.medterms.com on line dictionary
- ^ Why bother with Polyphenols, by Stéphane Quideau, on Groupe Polyphenols website
- ^ Polyphenols at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- ^ Mosconi Bernardini, P; Cersosimo, A (1979). "Histochemical and biophysical study of cuticle sclerotization in Pycnoscelus surinamensis L. (Blattaria)". Basic and applied histochemistry. 23 (3): 203–10. PMID 533516.
- ^ Rapid analysis of stilbenes and derivatives from downy mildew-infected grapevine leaves by liquid chromatography–atmospheric pressure photoionisation mass spectrometry. J. Bernard Jean-Denis, Roger Pezet and Raffaele Tabacchi, Journal of Chromatography A, Volume 1112, Issues 1-2, 21 April 2006, Pages 263-268, doi:10.1016/j.chroma.2006.01.060
- ^ Tandem mass spectrometry of the B-type procyanidins in wine and B-type dehydrodicatechins in an autoxidation mixture of (+)-catechin and (-)-epicatechin. Weixing Sun, Miller Jack M., Journal of mass spectrometry, 2003, vol. 38, no4, pp. 438-446
- ^ Identification of autoxidation oligomers of flavan-3-ols in model solutions by HPLC-MS/MS. Fei He, Qiu-Hong Pan, Ying Shi, Xue-Ting Zhang and Chang-Qing Duan, Journal of Mass Spectrometry, Volume 44 Issue 5, Pages 633 - 640, 2008, doi:10.1002/jms.1536
- ^ Nonenzymic autoxidative phenolic browning reactions in a caffeic acid model system. Johannes J. L. Cilliers and Vernon L. Singleton, J. Agric. Food Chem., 1989, 37 (4), pp 890–896, doi:10.1021/jf00088a013
- ^ Nonenzymic Autoxidative Reactions of Caffeic Acid in Wine. Johannes J. L. Cilliers 1 and Vernon L. Singleton, Am. J. Enol. Vitic. 41:1:84-86, 1990.
- ^ Phenolic Autoxidation Is Responsible for Color Degradation in Processed Carrot Puree. Talcott S. T. and Howard L. R., J. Agric. Food Chem., 1999, 47 (5), pp 2109–2115, doi:10.1021/jf981134n
- ^ ABTS radical-driven oxidation of polyphenols: Isolation and structural elucidation of covalent adducts. A.M. Osman, K.K.Y. Wong and A. Fernyhough, Biochemical and Biophysical Research Communications, Volume 346, Issue 1, 21 July 2006, Pages 321-329, doi:10.1016/j.bbrc.2006.05.118
- ^ Interactions between bioactive ferulic acid and fumed silica by UV–vis spectroscopy, FT-IR, TPD MS investigation and quantum chemical methods. T.V. Kulik, N.A. Lipkovska, V.N. Barvinchenko, B.B. Palyanytsya, O.A. Kazakova, O.A. Dovbiy and V.K. Pogorelyi, Journal of Colloid and Interface Science, Volume 339, Issue 1, 1 November 2009, Pages 60-68, doi:10.1016/j.jcis.2009.07.055
- ^ Encapsulation of fluorescence vegetable extracts within a templated sol–gel matrix. Ioana Lacatusu, Nicoleta Badea, Rodica Nita, Alina Murariu, Florin Miculescu, Ion Iosub and Aurelia Meghea, Optical Materials, Volume 32, Issue 6, April 2010, Pages 711-718, 2nd International Conference on Functional Materials and Devices, 2nd International Conference on Functional Materials and Devices, doi:10.1016/j.optmat.2009.09.001
- ^ Jamison, Jennifer R. Clinical Guide to Nutrition and Dietary Supplements in Disease Management. p. 525. ISBN 0-443-07193-4.
- ^ Harborne, J. B. (1980). "Plant phenolics". In Bell; Charlwood, B. V. (eds.). Encyclopedia of Plant Physiology, volume 8 Secondary Plant Products. Berlin Heidelberg New York: Springer-Verlag. pp. 329–395.
{{cite book}}: Missing pipe in:|editor1=(help); More than one of|editor1=and|editor1-last=specified (help) - ^ Polshettiwar, Vivek; Varma, Rajender S. (2008). "Greener and expeditious synthesis of bioactive heterocycles using microwave irradiation". Pure and Applied Chemistry. 80: 777–90. doi:10.1351/pac200880040777.
- ^ Reaction of polyphenols with formaldehyde. W. E. Hillis, Gerda Urbach, Journal of Applied Chemistry, Volume 9, Issue 12, pages 665–673, December 1959, doi:10.1002/jctb.5010091207
- ^ Fukuoka, Tokuma; Uyama, Hiroshi; Kobayashi, Shiro (2003). "Synthesis of Ultrahigh Molecular Weight Polyphenols by Oxidative Coupling". Macromolecules. 36: 8213–5. doi:10.1021/ma034803t.
- ^ Pizzi, A.; Valenezuela, J.; Westermeyer, C. (1994). "Low formaldehyde emission, fast pressing, pine and pecan tannin adhesives for exterior particleboard". Holz als Roh- und Werkstoff. 52: 311–5. doi:10.1007/BF02621421.
- ^ K. K. Jindal, R. C. Sharma (2004). Recent trends in horticulture in the Himalayas. Indus Publishing. ISBN 8173871620.
... bark of tree and rind of fruit is commonly used in ayurveda ... also used for dyeing ...
- ^ Oguchi, Takahisa; Tawaki, Shin-Ichiro; Uyama, Hiroshi; Kobayashi, Shiro (1999). "Soluble polyphenol". Macromolecular Rapid Communications. 20: 401–3. doi:10.1002/(SICI)1521-3927(19990701)20:7<401::AID-MARC401>3.0.CO;2-6.
- ^ Delsignore, A; Romeo, F; Giaccio, M (1997). "Content of phenolic substances in basidiomycetes". Mycological Research. 101: 552–6. doi:10.1017/S0953756296003206.
- ^ Hart, John H.; Hillis, W. E. (1974). "Inhibition of wood-rotting fungi by stilbenes and other polyphenols in Eucalyptus sideroxylon". Phytopathology. 64: 939–48. doi:10.1094/Phyto-64-939.
- ^ Blum, Udo; Shafer, Steven R.; Lehman, Mary E. (1999). "Evidence for Inhibitory Allelopathic Interactions Involving Phenolic Acids in Field Soils: Concepts vs. an Experimental Model". Critical Reviews in Plant Sciences. 18: 673–93. doi:10.1080/07352689991309441.
- ^ Popa, V; Dumitru, M; Volf, I; Anghel, N (2008). "Lignin and polyphenols as allelochemicals". Industrial Crops and Products. 27: 144–9. doi:10.1016/j.indcrop.2007.07.019.
- ^ Nakai, S (2000). "Myriophyllum spicatum-released allelopathic polyphenols inhibiting growth of blue-green algae Microcystis aeruginosa". Water Research. 34: 3026–32. doi:10.1016/S0043-1354(00)00039-7.
- ^ Li, Li; Li, Min-Hui; Xu, Li-Jia; Guo, Na; Wu-Lan, Ta-Na; Shi, Ren-Bing; Peng, Yong; Xiao, Pei-Gen (2010). "Distribution of seven polyphenols in several medicinal plants of Boraginaceae in China" (PDF). Journal of Medicinal Plants Research. 4 (12): 1216–21.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ The source of lipids and polyphenols for the insect cuticle: The role of fat body, oenocytes and oenocytoids. V.B. Wigglesworth, Tissue and Cell, Volume 20, Issue 6, 1988, Pages 919-932, doi:10.1016/0040-8166(88)90033-X
- ^ The Occurrence and Significance of Phenolic Hardening in the Newly Formed Cuticle of Crustacea decapoda. R. Dennell, Proc. R. Soc. Lond. B 30 September 1947 vol. 134 no. 877 485-503, doi:10.1098/rspb.1947.0027
- ^ The occurrence of phenolic substances in arthropods. R. H. Hackman, M. G. M. Pryor and A. R. Todd, Biochem J. 1948; 43(3): 474–477, PMC PMC1274717
- ^ The distribution of phenoloxidases and polyphenols during cuticle formation. M. Locke and N. Krishnan, Tissue and Cell, Volume 3, Issue 1, 1971, Pages 103-126, doi:10.1016/S0040-8166(71)80034-4
- ^ Phenolic Tanning and Pigmentation of the Cuticle in Carcinus maenas. G. Krishnan, Quarterly Journal of Microscopical Science, Vol s3-92, 333-342
- ^ The Epicuticle of an Arachnid, Palamneus swammerdami. G. Krishnan, Quarterly Journal of Microscopical Science, Vol s3-95, 371-381
- ^ Knaggs, Andrew R. (2001). "The biosynthesis of shikimate metabolites (1999)". Natural Product Reports. 18 (3): 334–55. doi:10.1039/b001717p. PMID 11476485.
- ^ Polyphenols and their quinone derivatives in the cuticle of the desert locust, Schistocerca gregaria (Forskål). S.R.A. Maleka, Comparative Biochemistry and Physiology, Volume 2, Issue 1, January 1961, Pages 35-50, doi:10.1016/0010-406X(61)90071-8
- ^ A Review of the Hypothetical Biogenesis and Regulation of Hypericin synthesis via the Polyketide Pathway in Hypericum perforatum and Experimental Methods Proposed to Evaluate the Hypothesis. Loren W. Walker, Portland State University , May, 1999
- ^ Polyketide Biosynthesis. Christian Hertweck, Angew. Chem. Int. Ed. 2009, 48, 4688-4716, doi:10.1002/anie.200806121
- ^ Polyphenol Glucosylating Activity in Cell Suspensions of Grape (Vitis vinifera). Mark N. Krasnow and Terence M. Murphy, J. Agric. Food Chem., 2004, 52 (11), pp 3467–3472, doi:10.1021/jf035234r
- ^ Lucas, Ricardo; Comelles, Francisco; Alcántara, David; Maldonado, Olivia S.; Curcuroze, Melanie; Parra, Jose L.; Morales, Juan C. (2010). "Surface-Active Properties of Lipophilic Antioxidants Tyrosol and Hydroxytyrosol Fatty Acid Esters: A Potential Explanation for the Nonlinear Hypothesis of the Antioxidant Activity in Oil-in-Water Emulsions". Journal of Agricultural and Food Chemistry. 58 (13): 8021–6. doi:10.1021/jf1009928. PMID 20524658.
- ^ Šmejkal, Karel; Grycová, Lenka; Marek, Radek; Lemière, Filip; Jankovská, Dagmar; Forejtníková, Hana; Vančo, Ján; Suchý, Václav (2007). "C-Geranyl Compounds from Paulownia tomentosa Fruits". Journal of Natural Products. 70 (8): 1244–8. doi:10.1021/np070063w. PMID 17625893.
- ^ Distribution of foliar formylated phloroglucinol derivatives amongst Eucalyptus species. B. M. Eschler, D. M. Pass, R. Willis and W. J. Foley, Biochemical Systematics and Ecology, Volume 28, Issue 9, November 2000, Pages 813-824, doi:10.1016/S0305-1978(99)00123-4
- ^ Rambourg, J. C.; Monties, B. (1983). "Determination of polyphenolic compounds in leaf protein concentrates of lucerne and their effect on the nutritional value". Qualitas Plantarum Plant Foods for Human Nutrition. 33: 169–72. doi:10.1007/BF01091304.
- ^ Gutfinger, T. (1981). "Polyphenols in olive oils". Journal of the American Oil Chemists Society. 58: 966–8. doi:10.1007/BF02659771.
- ^ Enhancing the quality of seafood products through new preservation techniques and seaweed-based antioxidants. Thesis, Tao Wang, 2009
- ^ Miglio C, Chiavaro E, Visconti A, Fogliano V, Pellegrini N (2008). "Effects of different cooking methods on nutritional and physicochemical characteristics of selected vegetables". J Agric Food Chem. 56: 139–47. PMID 18069785.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Bioavailability of multiple components following acute ingestion of a polyphenol-rich juice drink. Gina Borges, William Mullen, Adam Mullan, Michael E. J. Lean, Susan A. Roberts and Alan Crozier, Molecular Nutrition & Food Research, Special Issue: New Perspectives on Dietary Polyphenols, Volume 54, Issue Supplement 2, pages S268–S277, July 2010, doi:10.1002/mnfr.200900611
- ^ Bioavailability of the Polyphenols: Status and Controversies. Massimo D’Archivio, Carmelina Filesi, Rosaria Varì, Beatrice Scazzocchio and Roberta Masella, Int J Mol Sci. 2010; 11(4): 1321–1342, doi:10.3390/ijms11041321, PMC PMC2871118
- ^ Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Claudine Manach, Gary Williamson, Christine Morand, Augustin Scalbert and Christian Rémésy, American Journal of Clinical Nutrition, Vol. 81, No. 1, 230S-242S, January 2005, Dietary polyphenols and health: Proceedings of the 1st international conference on polyphenols and health
- ^ Williamson G, Manach C. Bioavailability and bioefficacy of polyphenols in humans. II. Review of 93 intervention studies. Am J Clin Nutr 81:(1) 243S-5S, 2005. Dietary polyphenols and health: Proceedings of the 1st International Conference on Polyphenols and Health
- ^ Study on Preparing Technique of Baicalin Liposomes. Wu Wenying, Xi Zhixia, Xue Hongan, Li Xinghua, Lei Jianlin and Li Jinjuan, China Pharmacist, 2004-09, Error: Bad DOI specified!
- ^ Study on preparation of baicalin liposome and its tissue distribution in mice. Wu Wen-ying, Xue Hong-an, Xi Zhi-xia, Li Xing-hua, Lei Jian-lin and Li Jin-juan, Chinese Journal of Biochemical Pharmaceutics, 2004-05 Error: Bad DOI specified!
- ^ Evaluation of polyphenol bioavailability in rat small intestine. Carbonaro M, Grant G and Pusztai A, Eur J Nutr. 2001 Apr;40(2):84-90, PMID 11518204
- ^ Protein-polyphenol interactions and in vivo digestibility of buckwheat groat proteins. Skrabanja V, Laerke HN and Kreft I, Pflugers Arch. 2000;440(5 Suppl):R129-31, PMID 11005640
- ^ Polyphenols and health: What compounds are involved? D. Del Rio, L.G. Costa, M.E.J. Lean and A. Crozier, Nutrition, Metabolism & Cardiovascular Diseases, Volume 20, Issue 1, Pages 1-6 (January 2010), doi:10.1016/j.numecd.2009.05.015
- ^ Intake and bioaccessibility of total polyphenols in a whole diet. Fulgencio Saura-Calixto, José Serrano and Isabel Goñi, Food Chemistry, Volume 101, Issue 2, 2007, Pages 492-501, doi:10.1016/j.foodchem.2006.02.006
- ^ Addition of milk prevents vascular protective effects of tea. Mario Lorenz, Nicoline Jochmann, Amélie von Krosigk, Peter Martus, Gert Baumann, Karl Stangl and Verena Stangl, Eur Heart J (2007) 28 (2): 219-223, doi:10.1093/eurheartj/ehl442
- ^ Polyphenol−β-Casein Complexes at the Air/Water Interface and in Solution: Effects of Polyphenol Structure. V. Aguié-Béghin, P. Sausse, E. Meudec, V. Cheynier and R. Douillard, J. Agric. Food Chem., 2008, 56 (20), pp 9600–9611, doi:10.1021/jf801672x
- ^ An Investigation of the interactions between milk proteins and tea polyphenols. P.J. Brown and W.B. Wright, Journal of Chromatography A, Volume 11, 1963, Pages 504-514, doi:10.1016/S0021-9673(01)80953-5
- ^ Balentine D. A., Harbowy M. E. and Graham H. N. (1998). G Spiller (ed.). Tea: the Plant and its Manufacture; Chemistry and Consumption of the Beverage.
{{cite book}}:|journal=ignored (help) - ^ Apple pectin and a polyphenol-rich apple concentrate are more effective together than separately on cecal fermentations and plasma lipids in rats. Aprikian O, Duclos V, Guyot S, Besson C, Manach C, Bernalier A, Morand C, Rémésy C and Demigné C, J Nutr. 2003 Jun;133(6):1860-5, PMID 12771330
- ^ Isolation and structure elucidation of the major individual polyphenols in carob fibre. R. W. Owen, R. Haubner, W. E. Hull, G. Erben, B. Spiegelhalder, H. Bartsch and B. Haber, Food and Chemical Toxicology, Volume 41, Issue 12, December 2003, Pages 1727-1738, doi:10.1016/S0278-6915(03)00200-X
- ^ Polyphenol extraction from foods. Maria teresa Escribano-Bailon and Celestino Santos-Buelga
- ^ Pan, X (2003). "Microwave-assisted extraction of tea polyphenols and tea caffeine from green tea leaves". Chemical Engineering and Processing. 42: 129–33. doi:10.1016/S0255-2701(02)00037-5.
- ^ Palma, M; Taylor, L (1999). "Extraction of polyphenolic compounds from grape seeds with near critical carbon dioxide". Journal of Chromatography A. 849 (1): 117–24. doi:10.1016/S0021-9673(99)00569-5. PMID 10444839.
- ^ Alonsosalces, R; Korta, E; Barranco, A; Berrueta, L; Gallo, B; Vicente, F (2001). "Pressurized liquid extraction for the determination of polyphenols in apple". Journal of Chromatography A. 933 (1–2): 37–43. doi:10.1016/S0021-9673(01)01212-2. PMID 11758745.
- ^ Sineiro, J.; Domínguez, H.; Núñez, M. J.; Lema, J. M. (1996). "Ethanol extraction of polyphenols in an immersion extractor. Effect of pulsing flow". Journal of the American Oil Chemists’ Society. 73: 1121–5. doi:10.1007/BF02523372.
- ^ Arranz, Sara; Saura-Calixto, Fulgencio; Shaha, Shika; Kroon, Paul A. (2009). "High Contents of Nonextractable Polyphenols in Fruits Suggest That Polyphenol Contents of Plant Foods Have Been Underestimated". Journal of Agricultural and Food Chemistry. 57 (16): 7298–303. doi:10.1021/jf9016652. PMID 19637929.
- ^ Nawaz, H; Shi, J; Mittal, G; Kakuda, Y (2006). "Extraction of polyphenols from grape seeds and concentration by ultrafiltration". Separation and Purification Technology. 48: 176–81. doi:10.1016/j.seppur.2005.07.006.
- ^ Prediction of pKa values of phenolic and nitrogen-containing compounds by computational chemical analysis compared to those measured by liquid chromatography. T. Hanai, K. Koizumi, T. Kinoshita, R. Arora and F. Ahmed, Journal of Chromatography A, Volume 762, Issues 1-2, 21 February 1997, Pages 55-61, 20th International Symposium on High Performance Liquid Phase Separation and Related Techniques, {{doi:10.1016/S0021-9673(96)01009-6}}
- ^ Spectrophotometric, potentiometric and chromatographic pKa values of polyphenolic acids in water and acetonitrile–water media. J.L. Beltrán, N. Sanli, G. Fonrodona, D. Barrón, G. Özkan and J. Barbosa, Analytica Chimica Acta 484 (2003) 253–264, doi:10.1016/S0003-2670(03)00334-9
- ^ Tannin-measuring techniques. Alice S. Tempel, Journal of Chemical Ecology, Volume 8, Number 10, 1289-1298, doi:10.1007/BF00987762
- ^ Monoclonal antibodies against tea polyphenols: A novel immunoassay to detect polyphenols in biological fluids. M. Gani, B. J. Mcguinness and A. P. Da Vies, Food and Agricultural Immunology, Volume 10, Issue 1 March 1998 , pages 13-22, doi:10.1080/09540109809354964
- ^ Walker, Richard B.; Everette, Jace D. (2009). "Comparative Reaction Rates of Various Antioxidants with ABTS Radical Cation". Journal of Agricultural and Food Chemistry. 57 (4): 1156–61. doi:10.1021/jf8026765. PMID 19199590.
- ^ ORAC and DPPH assay comparison to assess antioxidant capacity of tea infusions: Relationship between total polyphenol and individual catechin content. Roy Molay K, Koide,Motoki, Rao Theertham P, Okubo Tsutomu, Ogasawara Yutaka and Juneja Lekh R, International Journal of Food Sciences and Nutrition, Volume 61, Number 2, March 2010, pp. 109-124(16), PMID 20109129, doi:10.3109/09637480903292601
- ^ Antioxidant Activity of Dietary Polyphenols As Determined by a Modified Ferric Reducing/Antioxidant Power Assay. Raquel Pulido, Laura Bravo and Fulgencio Saura-Calixto, J. Agric. Food Chem., 2000, 48 (8), pp 3396–3402, doi:10.1021/jf9913458
- ^ Inhibition of Human Low-Density Lipoprotein Oxidation in Relation to Composition of Phenolic Antioxidants in Grapes (Vitis vinifera). Anne S. Meyer, Ock-Sook Yi, Debra A. Pearson, Andrew L. Waterhouse and Edwin N. Frankel, J. Agric. Food Chem., 1997, 45 (5), pp 1638–1643, doi:10.1021/jf960721a
- ^ Mello, L (2003). "HRP-based amperometric biosensor for the polyphenols determination in vegetables extract". Sensors and Actuators B: Chemical. 96: 636–45. doi:10.1016/j.snb.2003.07.008.
- ^ Glasser, Wolfgang G.; Glasser, Heidemarie R. (1974). "Simulation of Reactions with Lignin by Computer (Simrel). II. A Model for Softwood Lignin". Holzforschung. 28: 5–11, 1974. doi:10.1515/hfsg.1974.28.1.5.
- ^ Purple Canola: Arabidopsis PAP1 Increases Antioxidants and Phenolics in Brassica napus Leaves. Xiang Li, Ming-Jun Gao, Hong-Yu Pan, De-Jun Cui and Margaret Y. Gruber, J. Agric. Food Chem., 2010, 58 (3), pp 1639–1645, doi:10.1021/jf903527y
- ^ Regulation of polyphenol production in Vitis vinifera cell suspension cultures by sugars. F. Larronde, S. Krisa, A. Decendit, C. Chèze, G. Deffieux and J. M. Mérillon, Plant Cell Reports, Volume 17, Number 12, 946-950, doi:10.1007/s002990050515
External links
Databases
- phenol-explorer.eu by Augustin Scalbert, INRA Clermont-Ferrand, Unité de Nutrition Humaine (Human food unit)
- FlavonoidViewer.jp (Japanese, English) by Arita Group (Univ of Tokyo, RIKEN Plant Science Center, and Keio Univ), Nishioka Group (Kyoto and Keio Univ) and Kanaya Group (NAIST)
- Other tools
- KNApSACK
- massbank.jp, a high resolution Mass Spectral Database
- PubChem, NCBI
- liberherbarum.com, the incomplete reference-guide to Herbal medicine, Copyright © Erik Gotfredsen.
- metabolomics.jp (English, Japanese)
- KEGG: Kyoto Encyclopedia of Genes and Genomes
- ChEBI
- Comparative Toxicogenomics Database for toxicity


