92.194.76.172 (talk) →Pharmacodynamics: Style |
|||
| (10 intermediate revisions by the same user not shown) | |||
| Line 56: | Line 56: | ||
<!-- Definition and medical uses --> |
<!-- Definition and medical uses --> |
||
'''Oxcarbazepine''' is an [[anticonvulsant]] drug primarily used in the treatment of [[epilepsy]].<ref name=":3" /> There is some evidence for oxcarbazepine as a [[mood stabilizer|mood-stabilizing]] agent and thus, it can be used as add-on therapy for [[bipolar disorder]] in patients that have failed or are unable to tolerate approved treatments.<ref name="Mazza_2007">{{Cite journal|title = Oxcarbazepine in bipolar disorder: a critical review of the literature|journal = Expert Opinion on Pharmacotherapy|date = 2007-04-01|issn = 1744-7666|pmid = 17376019|pages = 649–656|volume = 8|issue = 5|doi = 10.1517/14656566.8.5.649|first = Marianna|last = Mazza|first2 = Marco|last2 = Di Nicola|first3 = Giovanni|last3 = Martinotti|first4 = Colombo|last4 = Taranto|first5 = Gino|last5 = Pozzi|first6 = Gianluigi|last6 = Conte|first7 = Luigi|last7 = Janiri|first8 = Pietro|last8 = Bria|first9 = Salvatore|last9 = Mazza}}</ref><ref name=":2">{{cite journal |vauthors=Ghaemi SN, Berv DA, Klugman J, Rosenquist KJ, Hsu DJ |title = Oxcarbazepine treatment of bipolar disorder|journal = J Clin Psychiatry|volume = 64|issue = 8|pages = 943–5|date = August 2003|pmid = 12927010|doi = 10.4088/JCP.v64n0813|url = }}</ref> |
'''Oxcarbazepine''' is an [[anticonvulsant]] drug primarily used in the treatment of [[epilepsy]].<ref name=":3" /> There is some evidence for oxcarbazepine as a [[mood stabilizer|mood-stabilizing]] agent and thus, it can be used as add-on therapy for [[bipolar disorder]] in patients that have failed or are unable to tolerate approved treatments.<ref name="Mazza_2007">{{Cite journal|title = Oxcarbazepine in bipolar disorder: a critical review of the literature|journal = Expert Opinion on Pharmacotherapy|date = 2007-04-01|issn = 1744-7666|pmid = 17376019|pages = 649–656|volume = 8|issue = 5|doi = 10.1517/14656566.8.5.649|first = Marianna|last = Mazza|first2 = Marco|last2 = Di Nicola|first3 = Giovanni|last3 = Martinotti|first4 = Colombo|last4 = Taranto|first5 = Gino|last5 = Pozzi|first6 = Gianluigi|last6 = Conte|first7 = Luigi|last7 = Janiri|first8 = Pietro|last8 = Bria|first9 = Salvatore|last9 = Mazza}}</ref><ref name=":2">{{cite journal |vauthors=Ghaemi SN, Berv DA, Klugman J, Rosenquist KJ, Hsu DJ |title = Oxcarbazepine treatment of bipolar disorder|journal = J Clin Psychiatry|volume = 64|issue = 8|pages = 943–5|date = August 2003|pmid = 12927010|doi = 10.4088/JCP.v64n0813|url = }}</ref> |
||
<!-- Side effects --> |
|||
Common side effects include nausea, vomiting, dizziness, drowsiness, headache, [[diplopia|double vision]] and [[ataxia|trouble with walking]].<ref name=":3">{{Cite web|title = DailyMed - OXCARBAZEPINE- oxcarbazepine tablet|url = http://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=17325a80-fb9c-4a83-b4b4-98e0b999d852|website = dailymed.nlm.nih.gov|accessdate = 2015-11-11}}</ref> Although not common, [[anaphylaxis]] may occur.<ref name=":3" /> Due to its structural similarities to [[carbamazepine]] there is approximately a 25–30% chance of cross-reactivity between the two medications.<ref name=":3" /> |
|||
<!-- History, society and culture --> |
<!-- History, society and culture --> |
||
| Line 73: | Line 70: | ||
Currently, there is limited data analyzing the impact of oxcarbazepine on a human fetus.<ref name=":3" /> Animal studies have shown an increased incidence of fetal abnormalities in pregnant rats and rabbits treated with oxcarbazepine, during pregnancy.<ref name=":3" /> In addition, oxcarbazepine is structurally similar to carbamazepine (pregnancy category: D) which is considered to be [[Teratology|teratogenic]] in humans.<ref name=":3" /><ref>{{Cite web|title = DailyMed - CARBAMAZEPINE- carbamazepine tablet CARBAMAZEPINE- carbamazepine tablet, chewable|url = http://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=c13bc0b8-7900-4ef4-98ed-e1315a08d95d|website = dailymed.nlm.nih.gov|accessdate = 2015-11-12}}</ref> Oxcarbazepine should only be used in pregnancy if the benefits justify the risks.<ref name=":3" /> |
Currently, there is limited data analyzing the impact of oxcarbazepine on a human fetus.<ref name=":3" /> Animal studies have shown an increased incidence of fetal abnormalities in pregnant rats and rabbits treated with oxcarbazepine, during pregnancy.<ref name=":3" /> In addition, oxcarbazepine is structurally similar to carbamazepine (pregnancy category: D) which is considered to be [[Teratology|teratogenic]] in humans.<ref name=":3" /><ref>{{Cite web|title = DailyMed - CARBAMAZEPINE- carbamazepine tablet CARBAMAZEPINE- carbamazepine tablet, chewable|url = http://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=c13bc0b8-7900-4ef4-98ed-e1315a08d95d|website = dailymed.nlm.nih.gov|accessdate = 2015-11-12}}</ref> Oxcarbazepine should only be used in pregnancy if the benefits justify the risks.<ref name=":3" /> |
||
Pregnant persons on oxcarbazepine should be closely monitored, as plasma levels of |
Pregnant persons on oxcarbazepine should be closely monitored, as plasma levels of licarbazepine has been shown to potentially decrease during pregnancy.<ref name=":3" /> |
||
| ⚫ | |||
| ⚫ | Oxcarbazepine and |
||
==Side effects== |
==Side effects== |
||
The most common side effects associated with oxcarbazepine include:<ref name=":3" /> |
The most common side effects associated with oxcarbazepine include:<ref name=":3">{{Cite web|title = DailyMed - OXCARBAZEPINE- oxcarbazepine tablet|url = http://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=17325a80-fb9c-4a83-b4b4-98e0b999d852|website = dailymed.nlm.nih.gov|accessdate = 2015-11-11}}</ref> |
||
* dizziness |
* dizziness |
||
* [[somnolence|drowsiness]] or fatigue |
|||
* blurred or double vision (2.1%) |
|||
* double vision, otherwise abnormal vision |
|||
* fatigue (2.1%) |
|||
| ⚫ | |||
* headaches (2.9%) |
|||
* indigestion, or abdominal pain |
|||
| ⚫ | |||
* tremor, abnormal gait, or ataxia |
|||
* vomiting (2%) |
|||
* sleepiness (2.4%) |
|||
* there is evidence of difficulty in concentration and mental sluggishness |
|||
Other rare side effects of oxcarbazepine include:<ref name=":3" /> |
Other rare side effects of oxcarbazepine include:<ref name=":3" /> |
||
| Line 99: | Line 91: | ||
Measurement of serum sodium levels should be considered in maintenance treatment or if symptoms of hyponatremia develop.<ref name=":3" /> Some side effects (such as headache) are more pronounced shortly after a dose is taken and tend to fade with the passage of time (60 to 90 minutes). Other side effects include stomach pain, tremor, rash, diarrhea, constipation, decreased appetite and dry mouth. Photosensitivity is a potential side-effect and people could experience severe [[sunburn]]s as a result of sun exposure.<ref name=":3" /> |
Measurement of serum sodium levels should be considered in maintenance treatment or if symptoms of hyponatremia develop.<ref name=":3" /> Some side effects (such as headache) are more pronounced shortly after a dose is taken and tend to fade with the passage of time (60 to 90 minutes). Other side effects include stomach pain, tremor, rash, diarrhea, constipation, decreased appetite and dry mouth. Photosensitivity is a potential side-effect and people could experience severe [[sunburn]]s as a result of sun exposure.<ref name=":3" /> |
||
Due to its structural similarities to [[carbamazepine]] there is approximately a 25–30% chance of cross-reactivity between the two medications.<ref name=":3" /> |
|||
| ⚫ | |||
| ⚫ | Oxcarbazepine and its active metabolite |
||
| ⚫ | |||
In addition, oxcarbazepine and MHD are [[CYP3A4]] and [[CYP3A5]] inducers and thus, have the potential to decrease the plasma concentration and therapeutic efficacy of medications that are CYP3A substrates.<ref name=":3" /> Examples of CYP3A4 substrates include [[dihydropyridine]] [[Calcium channel blocker|calcium channel antagonist]] and [[Oral contraceptive pill|oral contraceptives]].<ref name=":3" /> Due to the increase in drug metabolism of CYP3A4 substrates, hormonal contraceptives might be less effective, placing females at a greater risk of pregnancy.<ref name=":3" /><ref name=":5" /> |
|||
| ⚫ | Oxcarbazepine and licarbazepine are both present in human breast milk and thus, some of the active drug can be transferred to a nursing infant.<ref name=":3" /> When considering whether to continue this medication in nursing mothers, the impact of the drug's side effect profile on the infant, should be weighed against its anti-epileptic benefit for the mother.<ref name=":3" /> |
||
| ⚫ | |||
Furthermore, oxcarbazepine has been shown to interact with other anticonvulsant medications when used in combination.<ref name=":3" /> For example, phenytoin and [[phenobarbital]] are known CYP inducers and thus, have the potential to decrease the plasma concentration of oxcarbazepine's active metabolite, MHD.<ref name=":3" /> |
|||
| ⚫ | Oxcarbazepine and its active metabolite licarbazepine have been shown to exhibit interactions with drugs that are metabolized via [[CYP2C19]], [[CYP3A4]] and [[CYP3A5]].<ref name=":3" /> Oxcarbazepine and licarbazepine are potent inhibitors of [[CYP2C19]] and thus have the potential to increase plasma concentration of drugs that are metabolized through this pathway.<ref name=":3" /> In addition, oxcarbazepine and licarbazepine are [[CYP3A4]] and [[CYP3A5]] inducers and thus have the potential to decrease the plasma concentration of CYP3A4 and CYP3A5 substrates.<ref name=":3" /> Furthermore, oxcarbazepine has, in turn, been shown to interact with CYP inducers, including other anticonvulsant medications.<ref name=":3" /> |
||
Review articles of specific Cytochrome P450 enzymes for a list of their respective ligands. |
|||
| ⚫ | |||
| ⚫ | Oxcarbazepine has great [[bioavailability]] |
||
| ⚫ | |||
| ⚫ | Both oxcarbazepine and |
||
== Pharmacology == |
== Pharmacology == |
||
Oxcarbazepine and |
Oxcarbazepine and licarbazepine exert their action by blocking voltage-sensitive sodium channels, thus leading to the stabilization of hyper excited neural membranes, suppression of repetitive neuronal firing and diminishment propagation of synaptic impulses.<ref name=":3" /> Furthermore, anticonvulsant effects of these compounds could be attributed to enhanced potassium conductance and modulation of high-voltage activated calcium channels.<ref name=":3" /> |
||
| ⚫ | Oxcarbazepine is a derivative of [[carbamazepine]], whith reduced impact on the [[liver]] of [[drug metabolism|metabolizing]] the drug, and also lacks the serious forms of [[anemia]] or [[agranulocytosis]] occasionally associated with carbamazepine.{{citation needed|date=November 2015}} Aside from this reduction in side effects, it is thought to have the same mechanism as carbamazepine — [[sodium channel]] inhibition (presumed to be the main mechanism of action) – and is generally used to treat the same conditions. Oxcarbazepine is a [[prodrug]] which is activated to [[licarbazepine]] in the liver.<ref name="Dulsat">{{cite journal |
||
| ⚫ | |||
| ⚫ | |||
Since the structure of oxcarbazepine is similar to carbamazepine, there is a consideration for genetic testing in patients who are of Asian descent (mainly populations of Han Chinese (2–12%), Thai (8%), Philippines (15%), Malaysia) due to the higher frequency of the HLA-B*1502 [[allele]].<ref name=":3" /> [[Human leukocyte antigen]] (HLA) allele B*1502 has been associated with an increased incidence of [[Stevens–Johnson syndrome]] and [[toxic epidermal necrolysis]].<ref name=":3" /> |
|||
==Structure pharmacology== |
|||
| ⚫ | Oxcarbazepine is a |
||
Oxcarbazepine is a [[prodrug]] which is activated to [[licarbazepine]] in the liver.<ref name="Dulsat">{{cite journal |
|||
| author = Dulsat, C., Mealy, N., Castaner, R., Bolos, J. |
| author = Dulsat, C., Mealy, N., Castaner, R., Bolos, J. |
||
| title = Eslicarbazepine acetate |
| title = Eslicarbazepine acetate |
||
| Line 132: | Line 113: | ||
| doi = 10.1358/dof.2009.034.03.1352675 |
| doi = 10.1358/dof.2009.034.03.1352675 |
||
}}</ref> |
}}</ref> |
||
| ⚫ | |||
| ⚫ | Both oxcarbazepine and licarbazepine were found to show anticonvulsant properties in seizure models done on animals.<ref name=":3" /> These compounds had protective functions whenever tonic extension seizures were induced electrically, but such protection was less apparent whenever seizures were induced chemically.<ref name=":3" /> There was no observable tolerance during a four weeks course of treatment with daily administration of oxcarbazepine or licarbazepine in electroshock test on mice and rats.<ref name=":3" /> The antiepileptic activity can be attributed to licarbazepine.<ref name=":3" /> |
||
| ⚫ | |||
| ⚫ | Oxcarbazepine has great [[bioavailability]] upon oral administration.<ref name=":3" /> Once absorbed, oxcarbazepine is largely metabolized to its pharmacologically active 10-monohydroxy metabolite [[licarbazepine]] (sometimes called ''MHD'', monohydroxy derivate).<ref name=":3" /> In a study in humans, the 2% of oxcarbazepine remained unchanged, 70% was metabolised to licarbazepine, and the rest were minor metabolites.<ref name=":3" /> The [[half-life]] of oxcarbazepine is considered to be about 2 hours, whereas licarbazepine has a half-life of nine hours. |
||
| ⚫ | |||
| ⚫ | |||
[[Human leukocyte antigen]] (HLA) allele B*1502 has been associated with an increased incidence of [[Stevens–Johnson syndrome]] and [[toxic epidermal necrolysis]] in patients treated with carbamazepine, and thus those treated with oxcarbazepine might have similar risks.<ref name=":3" /> Patients who are of Asian descent are more likely to carry this genetic variant, especially Malaysians, Han Chinese (2–12%), Thai (8%), and Philippines (15%).<ref name=":3" /> Therefore, it has been suggested to consider genetic testing in these patients prior to initiation of treatment.<ref name=":3" /> |
|||
== Chemistry == |
|||
Oxcarbazepine is a structural derivative of [[carbamazepine]], with a [[ketone]] in place of the [[carbon–carbon double bond]] on the [[dibenzazepine]] ring at the 10 position (10-keto). |
|||
==History== |
==History== |
||
Revision as of 09:50, 18 October 2017
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ɒks.kɑːrˈbæz.ɪˌpiːn/ |
| Trade names | Trileptal |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601245 |
| Routes of administration | By mouth (tablets or liquid)[1] |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | >95% |
| Metabolism | Liver (cytosolic enzymes and glucuronic acid) |
| Elimination half-life | 1–5 hours (healthy adults) |
| Excretion | Kidney (<1%)[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.044.702 |
| Chemical and physical data | |
| Formula | C15H12N2O2 |
| Molar mass | 252.268 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Oxcarbazepine is an anticonvulsant drug primarily used in the treatment of epilepsy.[2] There is some evidence for oxcarbazepine as a mood-stabilizing agent and thus, it can be used as add-on therapy for bipolar disorder in patients that have failed or are unable to tolerate approved treatments.[3][4]
Oxcarbazepine is marketed as Trileptal by Novartis and available in some countries as a generic drug.[5] There is also an extended-release formulation marketed as Oxtellar XR by Supernus Pharmaceuticals.[6]
Medical uses
Oxcarbazepine is an anticonvulsant used to reduce the occurrence of epileptic episodes, and is not intended to cure epilepsy.[7] Oxcarbazepine is used alone or in combination with other medications for the treatment of focal (partial) seizures in adults.[2] In pediatric populations, it can be used by itself for the treatment of partial seizures for children 4 years and older, or in combination with other medications for children 2 years and older.[2]
In addition, oxcarbazepine has been shown to improve mood and reduce anxiety; therefore, it is a potential option for add-on therapy in the treatment of bipolar disorder.[3][4]
Pregnancy
Oxcarbazepine is listed as Pregnancy Category C.[2]
Currently, there is limited data analyzing the impact of oxcarbazepine on a human fetus.[2] Animal studies have shown an increased incidence of fetal abnormalities in pregnant rats and rabbits treated with oxcarbazepine, during pregnancy.[2] In addition, oxcarbazepine is structurally similar to carbamazepine (pregnancy category: D) which is considered to be teratogenic in humans.[2][8] Oxcarbazepine should only be used in pregnancy if the benefits justify the risks.[2]
Pregnant persons on oxcarbazepine should be closely monitored, as plasma levels of licarbazepine has been shown to potentially decrease during pregnancy.[2]
Side effects
The most common side effects associated with oxcarbazepine include:[2]
- dizziness
- drowsiness or fatigue
- double vision, otherwise abnormal vision
- nausea and vomiting
- indigestion, or abdominal pain
- tremor, abnormal gait, or ataxia
Other rare side effects of oxcarbazepine include:[2]
- severe low blood sodium (1%)
- anaphylaxis / angioedema
- hypersensitivity (especially if experienced with carbamazepine previously [25-30% cross-reactivity])
- toxic epidermal necrolysis (TEN, 0.5 to 6 cases per million-person years)
- Stevens–Johnson syndrome (SJS, 0.5 to 6 cases per million-person years)
- thoughts of suicide (<1%)
Measurement of serum sodium levels should be considered in maintenance treatment or if symptoms of hyponatremia develop.[2] Some side effects (such as headache) are more pronounced shortly after a dose is taken and tend to fade with the passage of time (60 to 90 minutes). Other side effects include stomach pain, tremor, rash, diarrhea, constipation, decreased appetite and dry mouth. Photosensitivity is a potential side-effect and people could experience severe sunburns as a result of sun exposure.[2]
Due to its structural similarities to carbamazepine there is approximately a 25–30% chance of cross-reactivity between the two medications.[2]
Nursing mothers
Oxcarbazepine and licarbazepine are both present in human breast milk and thus, some of the active drug can be transferred to a nursing infant.[2] When considering whether to continue this medication in nursing mothers, the impact of the drug's side effect profile on the infant, should be weighed against its anti-epileptic benefit for the mother.[2]
Interactions
Oxcarbazepine and its active metabolite licarbazepine have been shown to exhibit interactions with drugs that are metabolized via CYP2C19, CYP3A4 and CYP3A5.[2] Oxcarbazepine and licarbazepine are potent inhibitors of CYP2C19 and thus have the potential to increase plasma concentration of drugs that are metabolized through this pathway.[2] In addition, oxcarbazepine and licarbazepine are CYP3A4 and CYP3A5 inducers and thus have the potential to decrease the plasma concentration of CYP3A4 and CYP3A5 substrates.[2] Furthermore, oxcarbazepine has, in turn, been shown to interact with CYP inducers, including other anticonvulsant medications.[2]
Review articles of specific Cytochrome P450 enzymes for a list of their respective ligands.
Pharmacology
Oxcarbazepine and licarbazepine exert their action by blocking voltage-sensitive sodium channels, thus leading to the stabilization of hyper excited neural membranes, suppression of repetitive neuronal firing and diminishment propagation of synaptic impulses.[2] Furthermore, anticonvulsant effects of these compounds could be attributed to enhanced potassium conductance and modulation of high-voltage activated calcium channels.[2] Oxcarbazepine is a derivative of carbamazepine, whith reduced impact on the liver of metabolizing the drug, and also lacks the serious forms of anemia or agranulocytosis occasionally associated with carbamazepine.[citation needed] Aside from this reduction in side effects, it is thought to have the same mechanism as carbamazepine — sodium channel inhibition (presumed to be the main mechanism of action) – and is generally used to treat the same conditions. Oxcarbazepine is a prodrug which is activated to licarbazepine in the liver.[9]
Pharmacodynamics
Both oxcarbazepine and licarbazepine were found to show anticonvulsant properties in seizure models done on animals.[2] These compounds had protective functions whenever tonic extension seizures were induced electrically, but such protection was less apparent whenever seizures were induced chemically.[2] There was no observable tolerance during a four weeks course of treatment with daily administration of oxcarbazepine or licarbazepine in electroshock test on mice and rats.[2] The antiepileptic activity can be attributed to licarbazepine.[2]
Pharmacokinetics
Oxcarbazepine has great bioavailability upon oral administration.[2] Once absorbed, oxcarbazepine is largely metabolized to its pharmacologically active 10-monohydroxy metabolite licarbazepine (sometimes called MHD, monohydroxy derivate).[2] In a study in humans, the 2% of oxcarbazepine remained unchanged, 70% was metabolised to licarbazepine, and the rest were minor metabolites.[2] The half-life of oxcarbazepine is considered to be about 2 hours, whereas licarbazepine has a half-life of nine hours.
Pharmacogenetics

Human leukocyte antigen (HLA) allele B*1502 has been associated with an increased incidence of Stevens–Johnson syndrome and toxic epidermal necrolysis in patients treated with carbamazepine, and thus those treated with oxcarbazepine might have similar risks.[2] Patients who are of Asian descent are more likely to carry this genetic variant, especially Malaysians, Han Chinese (2–12%), Thai (8%), and Philippines (15%).[2] Therefore, it has been suggested to consider genetic testing in these patients prior to initiation of treatment.[2]
Chemistry
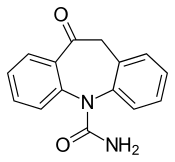
Oxcarbazepine is a structural derivative of carbamazepine, with a ketone in place of the carbon–carbon double bond on the dibenzazepine ring at the 10 position (10-keto).
History

First made in 1965,[citation needed] it was patent-protected by Geigy in 1969 through DE 2011087. It was approved for use as an anticonvulsant in Denmark in 1990, Spain in 1993, Portugal in 1997, and eventually for all other EU countries in 1999. It was approved in the US in 2000.[5] In September 2010, Novartis pleaded guilty to marketing Trileptal for the unapproved uses of neuropathic pain and bipolar disorder.[10]
See also
References
- ^ a b "Oxcarbazepine Drug Label".
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae af "DailyMed - OXCARBAZEPINE- oxcarbazepine tablet". dailymed.nlm.nih.gov. Retrieved 2015-11-11.
- ^ a b Mazza, Marianna; Di Nicola, Marco; Martinotti, Giovanni; Taranto, Colombo; Pozzi, Gino; Conte, Gianluigi; Janiri, Luigi; Bria, Pietro; Mazza, Salvatore (2007-04-01). "Oxcarbazepine in bipolar disorder: a critical review of the literature". Expert Opinion on Pharmacotherapy. 8 (5): 649–656. doi:10.1517/14656566.8.5.649. ISSN 1744-7666. PMID 17376019.
- ^ a b Ghaemi SN, Berv DA, Klugman J, Rosenquist KJ, Hsu DJ (August 2003). "Oxcarbazepine treatment of bipolar disorder". J Clin Psychiatry. 64 (8): 943–5. doi:10.4088/JCP.v64n0813. PMID 12927010.
- ^ a b "Trileptal® (oxcarbazepine) tablets and oral suspension". www.pharma.us.novartis.com. Retrieved 2015-11-11.
- ^ "Neurology Portfolio | Supernus Pharmaceuticals". www.supernus.com. Retrieved 2015-11-11.
- ^ Information, National Center for Biotechnology; Pike, U. S. National Library of Medicine 8600 Rockville; MD, Bethesda; Usa, 20894. "Oxcarbazepine (By mouth) - National Library of Medicine - PubMed Health". mmdn/DNX1023. Retrieved 2015-11-02.
{{cite web}}:|first4=has numeric name (help)CS1 maint: numeric names: authors list (link) - ^ "DailyMed - CARBAMAZEPINE- carbamazepine tablet CARBAMAZEPINE- carbamazepine tablet, chewable". dailymed.nlm.nih.gov. Retrieved 2015-11-12.
- ^ Dulsat, C., Mealy, N., Castaner, R., Bolos, J. (2009). "Eslicarbazepine acetate". Drugs of the Future. 34 (3): 189. doi:10.1358/dof.2009.034.03.1352675.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ "Novartis Pharmaceuticals Corp. to Pay More Than $420 Million to Resolve Off-label Promotion and Kickback Allegations | OPA | Department of Justice". www.justice.gov. Retrieved 2015-11-11.
