→Possible impacts: tweak wording |
→Possible impacts: tidy last paragraph |
||
| Line 100: | Line 100: | ||
</ref>. Recent work examining a [[sedimentology|sediment core]] from the [[Atlantic Ocean|North Atlantic]] found that while the species composition of coccolithophorids has remained unchanged for the [[Industrial revolution|industrial]] period 1780 to 2004, the calcification of coccoliths has increased by up to 40% during the same time<ref name=Iglesias2008/>. |
</ref>. Recent work examining a [[sedimentology|sediment core]] from the [[Atlantic Ocean|North Atlantic]] found that while the species composition of coccolithophorids has remained unchanged for the [[Industrial revolution|industrial]] period 1780 to 2004, the calcification of coccoliths has increased by up to 40% during the same time<ref name=Iglesias2008/>. |
||
While the full [[ecology|ecological]] consequences of these changes in calcification are still uncertain, it appears likely that many calcifying species will be adversely affected. There is also a suggestion that the effect of acidification on coccolithophores |
While the full [[ecology|ecological]] consequences of these changes in calcification are still uncertain, it appears likely that many calcifying species will be adversely affected. There is also a suggestion that the effect of acidification on coccolithophores may have secondary effects on climate change, by decreasing the earth's [[albedo]] via their effects on [[CLAW hypothesis|oceanic cloud cover]]<ref name=rutt06>{{cite journal |
||
|last=Ruttiman|first=J.| |
|last=Ruttiman|first=J.| |
||
|year=2006 |
|year=2006 |
||
| Line 107: | Line 107: | ||
|journal=Nature|issn=0028-0836 |
|journal=Nature|issn=0028-0836 |
||
|volume=442|issue=7106|pages=978-980|doi=10.1038/442978a}} (Subscription required) |
|volume=442|issue=7106|pages=978-980|doi=10.1038/442978a}} (Subscription required) |
||
</ref>. Aside from calcification, organisms may suffer other adverse effects, either directly as reproductive or physiological effects (e.g. CO<sub>2</sub>-induced acidification of body fluids, known as [[hypercapnia]]), or indirectly through negative impacts on food resources<ref name=raven05/>. However, as with calcification, as yet there is not a full understanding of these processes in marine organisms or [[ecosystem]]s. |
|||
</ref>. |
|||
Leaving aside direct biological effects, it is expected that ocean acidification in the future will lead to a complete cessation of the burial of carbonate sediments for several centuries, leading to the enhancement of the ocean as a reservoir for {{co2}} with moderate (and potentially beneficial) implications for climate change<ref name=tyrrell08>{{cite journal | last=Tyrrell | first=T. | year=2008 | title=Calcium carbonate cycling in future oceans and its influence on future climates | journal=J. Plankton Res. | volume=30 | pages=141-156 }}</ref><ref name=ridgwell07>{{cite journal |
|||
| last=Ridgwell | first=A. | coauthors=Zondervan, I., Hargreaves, J.C., Bijma, J. and Lenton, T.M. | year=2007 | title=Assessing |
|||
the potential long-term increase of oceanic fossil fuel {{co2}} uptake due to {{co2}}-calcification feedback | journal=Biogeosciences | volume=4 | pages=481–492}}</ref>. |
|||
==Gallery== |
==Gallery== |
||
Revision as of 08:16, 24 April 2008
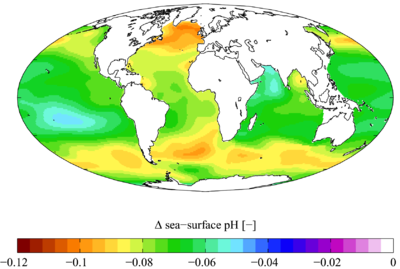
Ocean acidification is the name given to the ongoing decrease in the pH of the Earth's oceans, caused by their uptake of anthropogenic carbon dioxide from the atmosphere. Between 1751 and 1994 surface ocean pH is estimated to have decreased from approximately 8.179 to 8.104 (a change of -0.075).[1][2]
Carbon cycle
In the natural carbon cycle, the atmospheric concentration of carbon dioxide (CO2) represents a balance of fluxes between the oceans, terrestrial biosphere and the atmosphere. Human activities such as land-use changes, the combustion of fossil fuels, and the production of cement have led to a new flux of CO2 into the atmosphere. Some of this has remained in the atmosphere (where it is responsible for the rise in atmospheric concentrations), some is believed to have been taken up by terrestrial plants, and some has been absorbed by the oceans.
When CO2 dissolves, it reacts with water to form a balance of ionic and non-ionic chemical species : dissolved free carbon dioxide (CO2 (aq)), carbonic acid (H2CO3), bicarbonate (HCO3-) and carbonate (CO32-). The ratio of these species depends on factors such as seawater temperature and alkalinity (see the article on the ocean's solubility pump for more detail).
Acidification
| Time | pH | pH change | Source |
|---|---|---|---|
| Pre-industrial (1700s) | 8.179 | 0.000 | analysed field[2] |
| Recent past (1990s) | 8.104 | -0.075 | field[2] |
| 2050 (2×CO2 = 560 ppm) | 7.949 | -0.230 | model[1] |
| 2100 (IS92a)[3] | 7.824 | -0.355 | model[1] |
Dissolving CO2 in seawater also increases the hydrogen ion (H+) concentration in the ocean, and thus decreases ocean pH. The use of the term "ocean acidification" to describe this process was introduced in Caldeira and Wickett (2003)[4]. Since the industrial revolution began, it is estimated that surface ocean pH has dropped by slightly less than 0.1 units (on the logarithmic scale of pH), and it is estimated that it will drop by a further 0.3 - 0.5 units by 2100 as the ocean absorbs more anthropogenic CO2[4][1][5]. Note that, although the ocean is acidifying, its pH is still greater than 7 (that of neutral water), so the ocean could also be described as becoming less alkaline.
Possible impacts
Although the natural absorption of CO2 by the world's oceans helps mitigate the climatic effects of anthropogenic emissions of CO2, it is believed that the resulting decrease in pH will have negative consequences, primarily for oceanic calcifying organisms. These use the calcite or aragonite polymorphs of calcium carbonate to construct cell coverings or skeletons. Calcifiers span the food chain from autotrophs to heterotrophs and include organisms such as coccolithophores, corals, foraminifera, echinoderms, crustaceans and molluscs.
Under normal conditions, calcite and aragonite are stable in surface waters since the carbonate ion is at supersaturating concentrations. However, as ocean pH falls, so does the concentration of this ion, and when carbonate becomes under-saturated, structures made of calcium carbonate are vulnerable to dissolution. Research has already found that corals[6], coccolithophore algae[7][8][9][10], coralline algae[11], foraminifera[12], shellfish[13] and pteropods[1] experience reduced calcification or enhanced dissolution when exposed to elevated CO2. The Royal Society of London published a comprehensive overview of ocean acidification, and its potential consequences, in June 2005[5].
However, some studies have found different response to ocean acidification, with coccolithophore calcification and photosynthesis both increasing under elevated atmospheric pCO2[14][15][16], an equal decline in primary production and calcification in response to elevated CO2[17] or the direction of the response varying between species[18]. Recent work examining a sediment core from the North Atlantic found that while the species composition of coccolithophorids has remained unchanged for the industrial period 1780 to 2004, the calcification of coccoliths has increased by up to 40% during the same time[16].
While the full ecological consequences of these changes in calcification are still uncertain, it appears likely that many calcifying species will be adversely affected. There is also a suggestion that the effect of acidification on coccolithophores may have secondary effects on climate change, by decreasing the earth's albedo via their effects on oceanic cloud cover[19]. Aside from calcification, organisms may suffer other adverse effects, either directly as reproductive or physiological effects (e.g. CO2-induced acidification of body fluids, known as hypercapnia), or indirectly through negative impacts on food resources[5]. However, as with calcification, as yet there is not a full understanding of these processes in marine organisms or ecosystems.
Leaving aside direct biological effects, it is expected that ocean acidification in the future will lead to a complete cessation of the burial of carbonate sediments for several centuries, leading to the enhancement of the ocean as a reservoir for CO2 with moderate (and potentially beneficial) implications for climate change[20][21].
Gallery
 |
 |
 |
See also
- Biological pump
- Carbon dioxide sinks
- Continental shelf pump
- Global Ocean Data Analysis Project
- Seawater pH
- Solubility pump
References
- ^ a b c d e f Orr, James C. (2005). "Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms" (PDF). Nature. 437 (7059): 681–686. doi:10.1038/nature04095. ISSN 0028-0836.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ a b c Key, R.M. (2004). "A global ocean carbon climatology: Results from GLODAP". Global Biogeochemical Cycles. 18: GB4031. doi:10.1029/2004GB002247. ISSN 0886-6236.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Review of Past IPCC Emissions Scenarios, IPCC Special Report on Emissions Scenarios (ISBN 0521804930).
- ^ a b Caldeira, K. (2003). "Anthropogenic carbon and ocean pH" (PDF). Nature. 425 (6956): 365–365. doi:10.1038/425365a. ISSN 0028-0836.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ a b c Raven, J. A. et al. (2005). Ocean acidification due to increasing atmospheric carbon dioxide. Royal Society, London, UK.
- ^ Gattuso, J.-P. (1998). "Effect of calcium carbonate saturation of seawater on coral calcification". Global and Planetary Change. 18 (1–2): 37–46. doi:10.1016/S0921-8181(98)00035-6. ISSN 0921-8181.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Riebesell, Ulf (2000). "Reduced calcification of marine plankton in response to increased atmospheric CO2". Nature. 407 (6802): 364–367. doi:10.1038/35030078. ISSN 0028-0836.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) (Subscription required) - ^ Zondervan, I. (2001). "Decreasing marine biogenic calcification: a negative feedback on rising atmospheric pCO2". Global Biogeochem. Cycles. 15: 507–516.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Zondervan, I. (2002). "Effect of CO2 concentration on the PIC/POC ratio in the coccolithophore Emiliania huxleyi grown under light limiting conditions and different day lengths". J. Exp. Mar. Biol. Ecol. 272: 55–70.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Delille, B. (2005). "Response of primary production and calcification to changes of pCO2 during experimental blooms of the coccolithophorid Emiliania huxleyi". Global Biogeochem. Cycles. 19. doi:10.1029/2004GB002318.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help); line feed character in|journal=at position 7 (help) - ^ Kuffner, I.B. (2007). "Decreased abundance of crustose coralline algae due to ocean acidification". Nature Geoscience. 1: 114–117. doi:doi:10.1038/ngeo100.
{{cite journal}}: Check|doi=value (help); Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Phillips, Graham (2007-09-13). "Ocean Acidification – The BIG global warming story". ABC TV Science: Catalyst. Australian Broadcasting Corporation. Retrieved 2007-09-18.
{{cite news}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Gazeau, F. (2007). "Impact of elevated CO2 on shellfish calcification". Geophysical Research Letters. 34: L07603. doi:10.1029/2006GL028554. ISSN 0094-8276.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^
Buitenhuis, E.T. (1999). "Photosynthesis and calcification by Emiliania huxleyi (Prymnesiophyceae) as a function of inorganic carbon species". J. Phycology. 35: 949–959.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Nimer, N.A. (1993). "Calcification rate in Emiliania huxleyi Lohmann in response to light, nitrate and availability of inorganic carbon". New Phytologist. 123: 673–677.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ a b
Iglesias-Rodriguez, M.D. (2008). "Phytoplankton Calcification in a High-CO2 World". Science. 320: 336–340. doi:10.1126/science.1154122.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^
Sciandra, A. (2003). "Response of coccolithophorid Emiliania huxleyi to elevated partial pressure of CO2 under nitrogen limitation". Mar. Ecol. Prog. Ser. 261: 111–112.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^
Langer, G. (2006). "Species-specific responses of calcifying algae to changing seawater carbonate chemistry". Geochem. Geophys. Geosyst. 7. doi:0.1029/2005GC001227.
{{cite journal}}: Check|doi=value (help); Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Ruttiman, J. (2006). "Sick Seas". Nature. 442 (7106): 978–980. doi:10.1038/442978a. ISSN 0028-0836.
{{cite journal}}: Cite has empty unknown parameter:|1=(help) (Subscription required) - ^ Tyrrell, T. (2008). "Calcium carbonate cycling in future oceans and its influence on future climates". J. Plankton Res. 30: 141–156.
- ^ Ridgwell, A. (2007). "Assessing
the potential long-term increase of oceanic fossil fuel CO2 uptake due to CO2-calcification feedback". Biogeosciences. 4: 481–492.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help); line feed character in|title=at position 10 (help)
Further reading
- Cicerone, R. (2004). "The Ocean in a High CO2 World" (PDF). EOS, Transactions American Geophysical Union. 85 (37): 351–353. doi:10.1029/2004EO370007. ISSN 0096-3941.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help)
- Doney, S. C. (2006). "The Dangers of Ocean Acidification". Scientific American. 294: 58–65. ISSN 0036-8733.
{{cite journal}}: Cite has empty unknown parameter:|coauthors=(help), (Article preview only).
- Feely, R. A. (2004). "Impact of Anthropogenic CO2 on the CaCO3 System in the Oceans". Science. 305 (5682): 362–366. doi:10.1126/science.1097329. ISSN 0036-8075.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help)
- Henderson, Caspar (2006-08-05). "Ocean acidification: the other CO2 problem". NewScientist.com news service.
{{cite news}}: Check date values in:|date=(help)
- Jacobson, M. Z. (2005). "Studying ocean acidification with conservative, stable numerical schemes for nonequilibrium air-ocean exchange and ocean equilibrium chemistry". Journal of Geophysical Research - Atmospheres. 110: D07302. doi:10.1029/2004JD005220. ISSN 0148-0227.
{{cite journal}}: Cite has empty unknown parameter:|coauthors=(help)
- Kleypas, J.A., R.A. Feely, V.J. Fabry, C. Langdon, C.L. Sabine, and L.L. Robbins. (2006). Impacts of Ocean Acidification on Coral Reefs and Other Marine Calcifiers: A Guide for Further Research, report of a workshop held 18-20 April 2005, St. Petersburg, FL, sponsored by NSF, NOAA and the U.S. Geological Survey, 88pp.
- Kolbert, E. (2006). The Darkening Sea: Carbon emissions and the ocean. The New Yorker magazine. 20 November 2006.
- Sabine, C. L. (2004). "The Oceanic Sink for Anthropogenic CO2". Science. 305 (5682): 367–371. doi:10.1126/science.1097403. ISSN 0036-8075.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help)
- Stone, R. (2007). "A World Without Corals?". Science. 316 (5825): 678–681. doi:10.1126/science.316.5825.678. ISSN 0036-8075.
{{cite journal}}: Cite has empty unknown parameter:|coauthors=(help)
External links
- Announcement for Royal Society of London report
- Orr et al. (2005) supplementary material
- The Acid Ocean – the Other Problem with CO2 Emission, David Archer, a RealClimate discussion
- Task Force on Ocean Acidification in the Pacific, including recent presentations on ocean acidification, Pacific Science Association
- "Coral Bones" - an investigation into the future of coral reefs
- "Growing Acidity of Oceans May Kill Corals", Washington Post
- Ocean Acidification - a multimedia, interactive site from The World Ocean Observatory
- Dropping pH in the Oceans Causing a Rising Tide of Alarm by Tundi Agardy, The World Ocean Observatory
- Regularly-updated "blog" of ocean acidification publications and news
- The Ocean Acidification Network: An Information Network for the International Scientific Community
- CO2-04: Effect of Elevated CO2 on Phytoplankton project of Australia's Antarctic Climate and Ecosystems Cooperative Research Centre
Carbonate system calculators
The following packages calculate the state of the carbonate system in seawater (including pH):
- CO2SYS, a stand-alone executable (also available in a version for Microsoft Excel/VBA)
- seacarb, a R package for Windows, Mac OS X and Linux (also available here)
- CSYS, a Matlab script
