Content deleted Content added
Updating {{drugbox}} (no changed fields - added verified revid - updated 'CAS_number_Ref', 'verifiedrevid') per Chem/Drugbox validation (report errors or bugs) |
Solid Thight (talk | contribs) No edit summary Tag: Visual edit |
||
| (19 intermediate revisions by 14 users not shown) | |||
| Line 1: | Line 1: | ||
{{short description|Chemical compound}} |
|||
{{Unreferenced stub|auto=yes|date=December 2009}} |
|||
{{Drugbox |
{{Drugbox |
||
| verifiedrevid = 461769300 |
| verifiedrevid = 461769300 |
||
| IUPAC_name = 1-cyclohexylaminopropan-2-yl benzoate |
| IUPAC_name = 1-cyclohexylaminopropan-2-yl benzoate |
||
| image = Hexylcaine. |
| image = Hexylcaine Structure.svg |
||
<!--Clinical data--> |
<!--Clinical data--> |
||
| tradename = |
| tradename = |
||
| pregnancy_category = |
| pregnancy_category = |
||
| legal_status = |
| legal_status = |
||
| routes_of_administration = |
| routes_of_administration = |
||
<!--Pharmacokinetic data--> |
<!--Pharmacokinetic data--> |
||
| bioavailability = |
| bioavailability = |
||
| Line 16: | Line 14: | ||
| metabolism = |
| metabolism = |
||
| elimination_half-life = <10 minutes |
| elimination_half-life = <10 minutes |
||
<!--Identifiers--> |
<!--Identifiers--> |
||
| IUPHAR_ligand = 7196 |
|||
| CASNo_Ref = {{cascite|correct|CAS}} |
|||
| CAS_number_Ref = {{cascite|correct|??}} |
| CAS_number_Ref = {{cascite|correct|??}} |
||
| CAS_number = 532-77-4 |
| CAS_number = 532-77-4 |
||
| ATC_prefix = |
| ATC_prefix = None |
||
| ATC_suffix = |
| ATC_suffix = |
||
| ATC_supplemental = |
| ATC_supplemental = |
||
| PubChem = 10770 |
| PubChem = 10770 |
||
| DrugBank_Ref = {{drugbankcite|correct|drugbank}} |
| DrugBank_Ref = {{drugbankcite|correct|drugbank}} |
||
| DrugBank = DB00473 |
|||
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
||
| ChemSpiderID = 10315 |
| ChemSpiderID = 10315 |
||
| Line 35: | Line 32: | ||
| ChEMBL_Ref = {{ebicite|correct|EBI}} |
| ChEMBL_Ref = {{ebicite|correct|EBI}} |
||
| ChEMBL = 1197 |
| ChEMBL = 1197 |
||
<!--Chemical data--> |
<!--Chemical data--> |
||
| C=16 | H=23 | N=1 | O=2 |
| C=16 | H=23 | N=1 | O=2 |
||
| molecular_weight = 261.359 g/mol |
|||
| smiles = O=C(OC(CNC1CCCCC1)C)c2ccccc2 |
| smiles = O=C(OC(CNC1CCCCC1)C)c2ccccc2 |
||
| InChI = 1/C16H23NO2/c1-13(12-17-15-10-6-3-7-11-15)19-16(18)14-8-4-2-5-9-14/h2,4-5,8-9,13,15,17H,3,6-7,10-12H2,1H3 |
|||
| InChIKey = DKLKMKYDWHYZTD-UHFFFAOYAY |
|||
| StdInChI_Ref = {{stdinchicite|correct|chemspider}} |
| StdInChI_Ref = {{stdinchicite|correct|chemspider}} |
||
| StdInChI = 1S/C16H23NO2/c1-13(12-17-15-10-6-3-7-11-15)19-16(18)14-8-4-2-5-9-14/h2,4-5,8-9,13,15,17H,3,6-7,10-12H2,1H3 |
| StdInChI = 1S/C16H23NO2/c1-13(12-17-15-10-6-3-7-11-15)19-16(18)14-8-4-2-5-9-14/h2,4-5,8-9,13,15,17H,3,6-7,10-12H2,1H3 |
||
| Line 47: | Line 40: | ||
| StdInChIKey = DKLKMKYDWHYZTD-UHFFFAOYSA-N |
| StdInChIKey = DKLKMKYDWHYZTD-UHFFFAOYSA-N |
||
}} |
}} |
||
'''Hexylcaine hydrochloride''', also called '''cyclaine''' ([[Merck & Co.|Merck]]) or '''osmocaine''', is a short-acting [[local anesthetic]]. It acts by inhibiting [[sodium channel]] conduction. Overdose can lead to [[headache]], [[tinnitus]], [[paresthesia|numbness and tingling]] around the mouth and tongue, [[convulsion]]s, [[respiratory arrest|inability to breathe]], and decreased heart function. |
'''Hexylcaine hydrochloride''', also called '''cyclaine''' ([[Merck & Co.|Merck]]) or '''osmocaine''', is a short-acting [[local anesthetic]]. It acts by inhibiting [[sodium channel]] conduction. Overdose can lead to [[headache]], [[tinnitus]], [[paresthesia|numbness and tingling]] around the mouth and tongue, [[convulsion]]s, [[respiratory arrest|inability to breathe]], and decreased heart function.<ref name="pmid13620024">{{cite journal | vauthors = Spellberg MA | title = Hexylcaine (cyclaine) as topical anesthetic in gastroscopy and esophagoscopy | journal = Gastroenterology | volume = 36 | issue = 1 | pages = 120–1 | date = January 1959 | pmid = 13620024 | doi = 10.1016/S0016-5085(59)80102-5| url = }}</ref> |
||
==Synthesis== |
==Synthesis== |
||
[[File:Hexylcaine synthesis.svg|thumb|center|500px|Synthesis:<ref>Cope, Arthur C.; Hancock, Evelyn M. (1944). "1-Alkylamino-2-propanols and their p-Nitro- and p-Aminobenzoates". Journal of the American Chemical Society 66 (9): 1453–1456. doi:10.1021/ja01237a010.</ref><ref>"Local Anesthetics". New England Journal of Medicine. 263 (19): 963–965.1960. doi:10.1056/NEJM196011102631912. </ref> Patent:<ref>Cope Arthur C, {{US patent|2486374}} (1949 to Sharp & Dohme Inc).</ref>]] |
|||
[[File:Hexylcaine synthesis.png|thumb|center|700px|Hexylcaine synthesis:<ref>{{Cite doi|10.1021/ja01237a010}}</ref>]] |
|||
The [[reductive amination]] between [[1-Amino-2-propanol]] [78-96-6] ('''1''') and [[cyclohexanone]] gives 1-Cyclohexylamino-2-propanol [103-00-4] ('''2'''). Treatment with [[benzoyl chloride]] gives the ester, completing the synthesis of Hexylcaine ('''3''').{{Citation needed|date=November 2023}} |
|||
==References== |
|||
{{Reflist}} |
|||
{{Local anesthetics}} |
{{Local anesthetics}} |
||
[[Category:Local anesthetics]] |
[[Category:Local anesthetics]] |
||
[[Category: |
[[Category:Benzoate esters]] |
||
[[Category:Cyclohexylamines]] |
|||
{{nervous-system-drug-stub}} |
{{nervous-system-drug-stub}} |
||
Latest revision as of 08:48, 14 November 2023
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Pharmacokinetic data | |
| Elimination half-life | <10 minutes |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C16H23NO2 |
| Molar mass | 261.365 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Hexylcaine hydrochloride, also called cyclaine (Merck) or osmocaine, is a short-acting local anesthetic. It acts by inhibiting sodium channel conduction. Overdose can lead to headache, tinnitus, numbness and tingling around the mouth and tongue, convulsions, inability to breathe, and decreased heart function.[1]
Synthesis
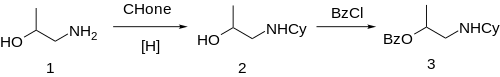
The reductive amination between 1-Amino-2-propanol [78-96-6] (1) and cyclohexanone gives 1-Cyclohexylamino-2-propanol [103-00-4] (2). Treatment with benzoyl chloride gives the ester, completing the synthesis of Hexylcaine (3).[citation needed]
References
- ^ Spellberg MA (January 1959). "Hexylcaine (cyclaine) as topical anesthetic in gastroscopy and esophagoscopy". Gastroenterology. 36 (1): 120–1. doi:10.1016/S0016-5085(59)80102-5. PMID 13620024.
- ^ Cope, Arthur C.; Hancock, Evelyn M. (1944). "1-Alkylamino-2-propanols and their p-Nitro- and p-Aminobenzoates". Journal of the American Chemical Society 66 (9): 1453–1456. doi:10.1021/ja01237a010.
- ^ "Local Anesthetics". New England Journal of Medicine. 263 (19): 963–965.1960. doi:10.1056/NEJM196011102631912.
- ^ Cope Arthur C, U.S. patent 2,486,374 (1949 to Sharp & Dohme Inc).
