LaughingVulcan (talk | contribs) Rearrangement of article (complete one sentence in introduction, move uses to own subheading from intro) |
vandalized months ago no one notices/cares just like hundreds of other instances. close to quiting wikipedia |
||
| Line 8: | Line 8: | ||
Cyalume was invented by Michael M. Rauhut and Laszlo J. Bollyky of [[American Cyanamid]] based on work by Edwin A. Chandross of [[Bell Labs]]. <ref>{{cite journal | title = What's that stuff? Light Sticks | journal = [[Chemical & Engineering News]] | author = Elizabeth Wilson | date = 18 Jan 1999 | volume = 77 | issue = 3 | pages = 65 | url = http://www.cem.msu.edu/~cem333/CENLightSticks.html | format = reprint}}</ref> Other early work on chemoluminescence was carried out at the same time, by researchers under [[Herbert Richter]] at [[China Lake Naval Weapons Center]]. <ref>{{cite web | author = Rood, S. A. | work = [http://scholar.lib.vt.edu/theses/available/etd-5298-191116/ Government Laboratory Technology Transfer: Process and Impact Assessment] (Doctoral Dissertation) | title = Chapter 4 Post-Legislation Cases | url = |
Cyalume was invented by Michael M. Rauhut and Laszlo J. Bollyky of [[American Cyanamid]] based on work by Edwin A. Chandross of [[Bell Labs]]. <ref>{{cite journal | title = What's that stuff? Light Sticks | journal = [[Chemical & Engineering News]] | author = Elizabeth Wilson | date = 18 Jan 1999 | volume = 77 | issue = 3 | pages = 65 | url = http://www.cem.msu.edu/~cem333/CENLightSticks.html | format = reprint}}</ref> Other early work on chemoluminescence was carried out at the same time, by researchers under [[Herbert Richter]] at [[China Lake Naval Weapons Center]]. <ref>{{cite web | author = Rood, S. A. | work = [http://scholar.lib.vt.edu/theses/available/etd-5298-191116/ Government Laboratory Technology Transfer: Process and Impact Assessment] (Doctoral Dissertation) | title = Chapter 4 Post-Legislation Cases | url = |
||
http://scholar.lib.vt.edu/theses/available/etd-5298-191116/unrestricted/cases2.pdf}}</ref><ref>{{cite news | title = The great glow stick controversy (Forum Section) | author = Steve Givens | date = 27 Jul 2005 | url = http://www.studlife.com/media/storage/paper337/news/2005/07/27/Forum/The-Great.Glow.Stick.Controversy-965113.shtml?norewrite200608160150&sourcedomain=www.studlife.com | publisher = Student Life}}</ref> Richard Taylor Van Zandt is the registered inventor on the {{US patent|4064428}} filed on November 1, 1976 for the original "Chemical Light Device". |
http://scholar.lib.vt.edu/theses/available/etd-5298-191116/unrestricted/cases2.pdf}}</ref><ref>{{cite news | title = The great glow stick controversy (Forum Section) | author = Steve Givens | date = 27 Jul 2005 | url = http://www.studlife.com/media/storage/paper337/news/2005/07/27/Forum/The-Great.Glow.Stick.Controversy-965113.shtml?norewrite200608160150&sourcedomain=www.studlife.com | publisher = Student Life}}</ref> Richard Taylor Van Zandt is the registered inventor on the {{US patent|4064428}} filed on November 1, 1976 for the original "Chemical Light Device". |
||
[[Image:Lightstick disassembly.jpg|thumb|center|600px|Disassembly of a chemoluminescent glowstick showing original lightstick at left, then the opened glowstick with peroxide mixture poured into a graduated cylinder and glass ampoule of fluorophore removed, then all three under UV illumination showing fluorophore fluorescence and plastic container fluorescence, then chemoluminescence of mixed substances in the graduated cylinder and finally on the right the mixture put back into the original plastic container showing the slightly different (more orange) color of light emission.]] |
|||
== Uses == |
== Uses == |
||
Glowsticks are used for many purposes. They may be used by [[marching band]] conductors for night time performances, and also used for entertainment at parties (especially [[Rave party|raves]]), concerts, and dance clubs. [[Glowsticking]] refers to the use of glowsticks in [[dancing]]. A further application are light effects, especially [[balloon-carried light effect]]s. They are also used as light sources and light markers by [[military|miltary forces]], campers, and [[recreational diving|recreational divers]] doing [[night diving]]. Glowsticks are considered the only safe light source immediately following an earthquake, due to the fact that they do not use any kind of electricity to work, and there is no danger of sparking. |
Glowsticks are used for many purposes. They may be used by [[marching band]] conductors for night time performances, and also used for entertainment at parties (especially [[Rave party|raves]]), concerts, and dance clubs. [[Glowsticking]] refers to the use of glowsticks in [[dancing]]. A further application are light effects, especially [[balloon-carried light effect]]s. They are also used as light sources and light markers by [[military|miltary forces]], campers, and [[recreational diving|recreational divers]] doing [[night diving]]. Glowsticks are considered the only safe light source immediately following an earthquake, due to the fact that they do not use any kind of electricity to work, and there is no danger of sparking. |
||
Revision as of 02:53, 4 July 2007

A glowstick or lightstick is a transparent plastic tube which contains chemical substances capable to produces light through a chemical reaction induced chemiluminescence which does not require an electrical power source.
Glowsticks are waterproof, do not use batteries, and are inexpensive and disposable. They can tolerate high pressures, such as those found underwater. Because they do not have batteries or contain electrified filaments like normal flashlights, they are safe for use in explosive environments. In many situations they are preferable to use as opposed to other light sources, despite the disadvantage of not being renewable or rechargable.
History
Cyalume was invented by Michael M. Rauhut and Laszlo J. Bollyky of American Cyanamid based on work by Edwin A. Chandross of Bell Labs. [1] Other early work on chemoluminescence was carried out at the same time, by researchers under Herbert Richter at China Lake Naval Weapons Center. [2][3] Richard Taylor Van Zandt is the registered inventor on the U.S. patent 4,064,428 filed on November 1, 1976 for the original "Chemical Light Device".

Uses
Glowsticks are used for many purposes. They may be used by marching band conductors for night time performances, and also used for entertainment at parties (especially raves), concerts, and dance clubs. Glowsticking refers to the use of glowsticks in dancing. A further application are light effects, especially balloon-carried light effects. They are also used as light sources and light markers by miltary forces, campers, and recreational divers doing night diving. Glowsticks are considered the only safe light source immediately following an earthquake, due to the fact that they do not use any kind of electricity to work, and there is no danger of sparking.
Special glowstick formulas emitting infrared radiation are used in conjunction with night vision devices.
Dangers
Glowsticks contain hydrogen peroxide, and phenol is produced as a by-product. Therefore, it is advisable to keep the mixture away from skin, and food if the glowstick case splits, or breaks.
Chemistry
The glowstick contains two chemicals and a suitable fluorescent dye (sensitizer, or fluorophor). The chemicals in the plastic tube are a mixture of the dye and a derivate of phenyl oxalate ester (also called Cyalume). The chemical inside the glass vial is concentrated (about 35%) hydrogen peroxide. By mixing the peroxide with the phenyl oxalate ester, a chemical reaction takes place; the ester is oxidized, yielding two molecules of phenol and one molecule of peroxyacid ester. The peroxyacid decomposes spontaneously to carbon dioxide, releasing energy that excites the dye, which then deexcites by releasing a photon. The wavelength of the photon—the color of the emitted light—depends on the structure of the dye.

Other chemicals are usually present in the mixture as well; eg. sodium salicylate as a catalyst, and bis(2-ethylhexyl) phthalate (DEHP) as a solvent. Instead of cyalume, bis(2,4,5-trichlorophenyl-6-carbopentoxyphenyl)oxalate (CPPO) is used in practical formulations, as it gives significantly better quantum yield. A similar reaction can be achieved with bis(2,4,6-trichlorophenyl)oxalate (TCPO).

The dyes used in glowsticks usually exhibit fluorescence when exposed to ultraviolet radiation. Therefore even a spent glowstick will shine under a black light.
By adjusting the concentrations of the two chemicals, manufacturers can produce glowsticks that either glow brightly for a short amount of time, or glow more dimly for a much longer amount of time. At maximum concentration (typically only found in laboratory settings), mixing the chemicals results in a furious reaction, producing large amounts of light for only a few seconds.
Heating a glowstick causes the reaction to proceed faster and the glowstick to glow brighter, but for a shorter period of time. Cooling a glowstick slows the reaction and causes it to last longer, but the light is dimmer. This can be demonstrated by refrigerating or freezing an active glowstick; when it warms up again, it will resume glowing.
Fluorophors used
- 9,10-diphenylanthracene (DPA) emits blue light
- 1-chloro-9,10-diphenylanthracene (1-chloro(DPA)) and 2-chloro-9,10-diphenylanthracene (2-chloro(DPA)) emit blue-green light with higher efficiency than unsubstituted DPA, dihydro(DPA) is purple
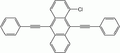
- 9,10-bis(phenylethynyl)anthracene (BPEA) emits green light
- 1-chloro-9,10-bis(phenylethynyl)anthracene emits yellow-green light, used in 30-minute high-intensity Cyalume sticks
- 2-chloro-9,10-bis(phenylethynyl)anthracene emits green light, used in 12-hour low-intensity Cyalume sticks
- 1,8-dichloro-9,10-bis(phenylethynyl)anthracene emits yellow light, used in Cyalume sticks
- Rubrene emits yellow.
- 2,4-di-tert-butylphenyl 1,4,5,8-tetracarboxynaphthalene diamide emits deep red light, together with DPA is used to produce white or hot-pink light, depending on their ratio
- Rhodamine B emits red light. Is rarely used as it breaks down in contact with phenyl oxalate, shortening the shelf life of the mixture
- 5,12-bis(phenylethynyl)naphthacene emits orange light
-
9,10-diphenylanthracene yields blue light
-
9,10-bis(phenylethynyl) anthracene yields green light
-
1-chloro- 9,10-bis(phenylethynyl) anthracene yields yellow light
-
rubrene (5,6,11,12-tetraphenyl naphthacene) yields yellow light
-
5,12-bis(phenylethynyl) naphthacene yields orange light
-
Rhodamine 6G yields orange light
-
Rhodamine B yields red light
References
- ^ Elizabeth Wilson (18 Jan 1999). "What's that stuff? Light Sticks" (reprint). Chemical & Engineering News. 77 (3): 65.
- ^ Rood, S. A. "Chapter 4 Post-Legislation Cases" (PDF). Government Laboratory Technology Transfer: Process and Impact Assessment (Doctoral Dissertation).
{{cite web}}: External link in|work= - ^ Steve Givens (27 Jul 2005). "The great glow stick controversy (Forum Section)". Student Life.