expand history |
redo construction section |
||
| Line 25: | Line 25: | ||
There is no information on the effects of smoking HnB devices during pregnancy, as of 2018.<ref name="pregnancy"/> However, they are nicotine-containing products.<ref name=WHO2018/> It is unsafe to use any product containing [[nicotine#Fetal development and breastfeeding|nicotine during pregnancy and breastfeeding]],<ref name=notsafe>{{Cite journal| doi = 10.1093/toxsci/kfq103| issn = 1096-6080| volume = 116| issue = 2| pages = 364–374| last1 = Bruin| first1 = Jennifer E.| last2 = Gerstein| first2 = Hertzel C.| last3 = Holloway| first3 = Alison C.| title = Long-Term Consequences of Fetal and Neonatal Nicotine Exposure: A Critical Review| journal = Toxicological Sciences| date = 2010-04-02| pmid = 20363831| pmc = 2905398|quote=Overall, the evidence provided in this review overwhelmingly indicates that nicotine should no longer be considered the ‘‘safe’’ component of cigarette smoke. In fact, many of the adverse postnatal health outcomes associated with maternal smoking during pregnancy may be attributable, at least in part, to nicotine alone.}}</ref> as nicotine harms the fetus.<ref>{{Cite journal| doi = 10.3109/00016341003605719| pmid = 20225987| pmc = 5881107| issn = 1600-0412| volume = 89| issue = 4| pages = 454–464| last1 = England| first1 = Lucinda J.| last2 = Kim| first2 = Shin Y.| last3 = Tomar| first3 = Scott L.| last4 = Ray| first4 = Cecily S.| last5 = Gupta| first5 = Prakash C.| last6 = Eissenberg| first6 = Thomas| last7 = Cnattingius| first7 = Sven| last8 = Bernert| first8 = John T.| last9 = Tita| first9 = Alan Thevenet N.| last10 = Winn| first10 = Deborah M.| last11 = Djordjevic| first11 = Mirjana V.| last12 = Lambe| first12 = Mats| last13 = Stamilio| first13 = David| last14 = Chipato| first14 = Tsungai| last15 = Tolosa| first15 = Jorge E.| title = Non-cigarette tobacco use among women and adverse pregnancy outcomes| journal = Acta Obstetricia et Gynecologica Scandinavica|date=31 December 2010|quote=The use of any products containing nicotine likely will have adverse effects of fetal neurological development. }}</ref> Nicotine use during pregnancy increases the child's risk of [[respiratory problem]]s, [[Neurobehavioral disorder|neurobehavioral defects]], obesity, [[type 2 diabetes]], [[hypertension]], and infertility.<ref name=Schraufnagel2014>{{cite journal |vauthors=Schraufnagel DE, Blasi F, Drummond MB, Lam DC, Latif E, Rosen MJ, Sansores R, Van Zyl-Smit R |title=Electronic cigarettes. A position statement of the forum of international respiratory societies |journal=American Journal of Respiratory and Critical Care Medicine |volume=190 |issue=6 |pages=611–8 |date=September 2014 |pmid=25006874 |doi=10.1164/rccm.201407-1198PP |url=https://www.thoracic.org/newsroom/press-releases/journal/articles/FIRS-E-cigarette-position-paper.pdf}}</ref> |
There is no information on the effects of smoking HnB devices during pregnancy, as of 2018.<ref name="pregnancy"/> However, they are nicotine-containing products.<ref name=WHO2018/> It is unsafe to use any product containing [[nicotine#Fetal development and breastfeeding|nicotine during pregnancy and breastfeeding]],<ref name=notsafe>{{Cite journal| doi = 10.1093/toxsci/kfq103| issn = 1096-6080| volume = 116| issue = 2| pages = 364–374| last1 = Bruin| first1 = Jennifer E.| last2 = Gerstein| first2 = Hertzel C.| last3 = Holloway| first3 = Alison C.| title = Long-Term Consequences of Fetal and Neonatal Nicotine Exposure: A Critical Review| journal = Toxicological Sciences| date = 2010-04-02| pmid = 20363831| pmc = 2905398|quote=Overall, the evidence provided in this review overwhelmingly indicates that nicotine should no longer be considered the ‘‘safe’’ component of cigarette smoke. In fact, many of the adverse postnatal health outcomes associated with maternal smoking during pregnancy may be attributable, at least in part, to nicotine alone.}}</ref> as nicotine harms the fetus.<ref>{{Cite journal| doi = 10.3109/00016341003605719| pmid = 20225987| pmc = 5881107| issn = 1600-0412| volume = 89| issue = 4| pages = 454–464| last1 = England| first1 = Lucinda J.| last2 = Kim| first2 = Shin Y.| last3 = Tomar| first3 = Scott L.| last4 = Ray| first4 = Cecily S.| last5 = Gupta| first5 = Prakash C.| last6 = Eissenberg| first6 = Thomas| last7 = Cnattingius| first7 = Sven| last8 = Bernert| first8 = John T.| last9 = Tita| first9 = Alan Thevenet N.| last10 = Winn| first10 = Deborah M.| last11 = Djordjevic| first11 = Mirjana V.| last12 = Lambe| first12 = Mats| last13 = Stamilio| first13 = David| last14 = Chipato| first14 = Tsungai| last15 = Tolosa| first15 = Jorge E.| title = Non-cigarette tobacco use among women and adverse pregnancy outcomes| journal = Acta Obstetricia et Gynecologica Scandinavica|date=31 December 2010|quote=The use of any products containing nicotine likely will have adverse effects of fetal neurological development. }}</ref> Nicotine use during pregnancy increases the child's risk of [[respiratory problem]]s, [[Neurobehavioral disorder|neurobehavioral defects]], obesity, [[type 2 diabetes]], [[hypertension]], and infertility.<ref name=Schraufnagel2014>{{cite journal |vauthors=Schraufnagel DE, Blasi F, Drummond MB, Lam DC, Latif E, Rosen MJ, Sansores R, Van Zyl-Smit R |title=Electronic cigarettes. A position statement of the forum of international respiratory societies |journal=American Journal of Respiratory and Critical Care Medicine |volume=190 |issue=6 |pages=611–8 |date=September 2014 |pmid=25006874 |doi=10.1164/rccm.201407-1198PP |url=https://www.thoracic.org/newsroom/press-releases/journal/articles/FIRS-E-cigarette-position-paper.pdf}}</ref> |
||
== |
== Construction == |
||
<gallery mode=packed heights=295px> |
|||
[[File:Verkohlte Pizza 2013-04-01-2663.jpg|thumb|This pizza was baked for four hours. Like the tobacco in these products, it has [[charring|charred]], and it has released pyrolysis products and particulate matter, but most of the carbon has not oxidized and it has not been reduced to [[ash]] (see [[carbonization]]).]] |
|||
File:Low-temperature cigarette disassembled.png|Tobacco stick; above, disassembled, below, intact.<ref name=PieperMallock2018/> A: Reconstituted tobacco film, made of dried tobacco [[suspension (chemistry)|suspension]].<ref name=PieperMallock2018/> 70% tobacco, [[humectants]] (water and [[glycerol|glycerin]]) to encourage aerosol formation, binding agents, and aroma agents.<ref name=PieperMallock2018/> B: Hollow [[acetate]] tube.<ref name=PieperMallock2018/> C: [[Polymer]] film filter cools the aerosol.<ref name=PieperMallock2018/> D: Soft [[cellulose acetate]] mouthpiece, which mimics the feel of a traditional cigarette.<ref name=PieperMallock2018/>|alt=Tobacco stick; above, disassembled, below, intact. A: Reconstituted tobacco film, made of dried tobacco suspension. 70% tobacco, humectants (water and glycerin) to encourage aerosol formation, binding agents, and aroma agents. B: Hollow acetate tube. C: Polymer film filter cools the aerosol. D: Soft cellulose acetate mouthpiece, which mimics the feel of a traditional cigarette. |
|||
</gallery> |
|||
Nicotine is released from tobacco heated above 150 °C.<ref name=ForsterLiu2015>{{cite journal|last1=Forster|first1=Mark|last2=Liu|first2=Chuan|last3=Duke|first3=Martin G|last4=McAdam|first4=Kevin G|last5=Proctor|first5=Christopher J|title=An experimental method to study emissions from heated tobacco between 100-200°C|journal=Chemistry Central Journal|volume=9|issue=1|year=2015|issn=1752-153X|doi=10.1186/s13065-015-0096-1|pmc=4418098|pmid=25941536}}</ref> Combustible tobacco cigarettes reach about 900 °C during a puff and smoulder at about 400 °C between puffs.<ref name=St.HelenJacobIII2018/> The burning process, substances emitted and their levels vary at different temperatures.<ref name=St.HelenJacobIII2018/> Distillation, the process during which nicotine and aromas are transferred from tobacco to smoke, occurs below 300 °C; [[pyrolysis]] occurs at about 300 °C–700 °C, entails the decomposition of biopolymers, proteins, and other organic materials and generates the majority of substances emitted in smoke; and combustion occurs above 750 °C and results in the generation of [[carbon dioxide]], [[carbon monoxide]], and water.<ref name=St.HelenJacobIII2018/> HeatSticks are heated to a maximum of 350 °C, a temperature sufficient to enable pyrolytic decomposition of some organic materials.<ref name=St.HelenJacobIII2018/> Formation of toxic volatile organic compounds, including formaldehyde, acetaldehyde and acrolein, via dehydration and oxidation of the humectants, propylene glycol and glycerin, have been reported in e-cigarette aerosols at similar temperatures as IQOS.<ref name=St.HelenJacobIII2018/> In addition, flavoring chemicals in e-cigarettes undergo thermal degradation and contribute significantly to levels of toxic aldehydes emitted in e-cigarette aerosol.<ref name=St.HelenJacobIII2018/> Since the constituents of HeatSticks may be different from that of combustible cigarettes, including flavorants and additives, it is plausible that the IQOS aerosol may contain substances not present in tobacco smoke.<ref name=St.HelenJacobIII2018>{{cite journal|last1=St.Helen|first1=Gideon|last2=Jacob III|first2=Peyton|last3=Nardone|first3=Natalie|last4=Benowitz|first4=Neal L|title=IQOS: examination of Philip Morris International’s claim of reduced exposure|journal=Tobacco Control|volume=27|issue=Suppl 1|year=2018|pages=s30–s36|issn=0964-4563|doi=10.1136/tobaccocontrol-2018-054321|pmid=30158205}}{{CC-notice|cc=by4|url=https://tobaccocontrol.bmj.com/content/27/Suppl_1/s30.long|author(s)=Gideon St.Helen, Peyton Jacob III, Natalie Nardone, and Neal L Benowitz}}</ref> The IQOS HeatSticks do not generate a flame, they are [[charring|charred]] following use.<ref name=Davis2017>{{cite journal|last1=Davis|first1=Barbara|last2=Williams|first2=Monique|last3=Talbot|first3=Prue|title=iQOS: evidence of pyrolysis and release of a toxicant from plastic|journal=Tobacco Control|date=20 February 2018|pages=tobaccocontrol–2017–054104|doi=10.1136/tobaccocontrol-2017-054104 |doi-broken-date=2018-10-25|issn=0964-4563|url=https://tobaccocontrol.bmj.com/content/early/2018/02/20/tobaccocontrol-2017-054104|pmid=29535257}}</ref> Heat-not-burn tobacco products may or may not generate [[tobacco smoke|smoke]].<ref name=Katz2017>{{cite journal|last1=Katz|first1=Mitchell H.|title=No Smoke—Just Cancer-Causing Chemicals|journal=JAMA Internal Medicine|volume=177|issue=7|date=July 2017|pages=1052|issn=2168-6106|doi=10.1001/jamainternmed.2017.1425|pmid=28531245}}</ref> |
|||
The heat-not-burn tobacco product consists of 3 components with different functions.<ref name=PieperMallock2018/> These include the tobacco stick with processed tobacco, a pen-like heater (holder), in which the tobacco stick is inserted, which is then heated by means of an electrically controlled heating element, and a charger (the charger), which recharges the heater after use.<ref name=PieperMallock2018/> The heat-not-burn tobacco products automatically stops the heating process after 6 minutes or 14 moves, so that pyrolysis products and pollutant release are limited in time as well as by a maximum number of puffs per stick.<ref name=PieperMallock2018/> The tobacco stick contains a compressed tobacco film as well as several filter elements.<ref name=PieperMallock2018/> The tobacco film consists of a dried tobacco suspension that has been rolled up into a paper-thin brown tobacco foil.<ref name=PieperMallock2018/> This consists of about 70% tobacco as well as humectants, binders and flavorings.<ref name=PieperMallock2018/> Water and glycerin are used as humectants to prevent drying out and to promote aerosol formation upon heating.<ref name=PieperMallock2018/> The filter elements consist of 2 independent systems: A polymer film filter that cools the aerosol, followed by a soft cellulose acetate mouthpiece filter that mimics the sensory aspects of a traditional cigarette.<ref name=PieperMallock2018/> |
|||
Nicotine is released from tobacco heated above 140°C.<ref>{{cite journal | last1=Forster | first1=Mark | last2=Liu | first2=Chuan | last3=Duke | first3=Martin | title=An experimental method to study emissions from heated tobacco between 100-200°C | journal=Chemistry Central Journal | date=16 April 2015 | volume=9 | pages=20 | pmid=25941536 | pmc=4418098 | doi=10.1186/s13065-015-0096-1 }}</ref> Heating tobacco causes [[pyrolysis]]; organic material breaks down, releasing most of the substances found in regular cigarette smoke.<ref name=constituents_pyrolysis_quote/><ref name="char"/> At higher temperatures, the [[carbon]] increasingly combines with atmospheric [[oxygen]], releasing [[carbon monoxide]] and [[carbon dioxide]].<ref name=constituents_pyrolysis_quote/> It is possible to heat to a temperature hot enough to cause pyrolysis, but not hot enough to oxidize and release the carbon.<ref name=constituents_pyrolysis_quote>{{cite journal |doi=10.1136/tobaccocontrol-2018-054321 |pmid=30158205 |title=IQOS: Examination of Philip Morris International's claim of reduced exposure |journal=Tobacco Control |volume=27 |issue=Suppl 1 |pages=tobaccocontrol–2018–054321 |year=2018 |last1=St.Helen |first1=Gideon |last2=Jacob Iii |first2=Peyton |last3=Nardone |first3=Natalie |last4=Benowitz |first4=Neal L |quote=PMI’s MRTP application fails to address the important question of whether the aerosol generation process for IQOS produces toxic substances not found in the smoke of combustible cigarettes, which could have been answered through non-targeted chemical analysis. Combustible tobacco cigarettes reach about 900°C during a puff and smoulder at about 400°C between puffs.[23] The burning process, substances emitted and their levels vary at different temperatures.[38] Distillation, the process during which nicotine and aromas are transferred from tobacco to smoke, occurs below 300°C; pyrolysis occurs at about 300°C–700°C, entails the decomposition of biopolymers, proteins, and other organic materials and generates the majority of substances emitted in smoke; and combustion occurs above 750°C and results in the generation of carbon dioxide, CO and water.[38] HeatSticks are heated to a maximum of 350°C,[19] a temperature sufficient to enable pyrolytic decomposition of some organic materials. Formation of toxic volatile organic compounds, including formaldehyde, acetaldehyde and acrolein, via dehydration and oxidation of the humectants, propylene glycol and glycerin, have been reported in e-cigarette aerosols at similar temperatures as IQOS.[39–42] In addition, flavouring chemicals in e-cigarettes undergo thermal degradation and contribute significantly to levels of toxic aldehydes emitted in e-cigarette aerosol.[43] Since the constituents of HeatSticks may be different from that of combustible cigarettes, including flavourants and additives, it is plausible that the IQOS aerosol may contain substances not present in tobacco smoke.}}<!--Please note that this article is licensed under CC-BY 4.0, and the text is thus usable in Wikipedia; quote added as content was challenged--></ref> This [[Charcoal#History|ancient]] process is called [[charring]],<ref name="char">{{cite journal |pmid=29535257 |url=https://tobaccocontrol.bmj.com/content/early/2018/02/20/tobaccocontrol-2017-054104 |laysummary=https://www.bmj.com/company/newsroom/heat-not-burn-smokeless-tobacco-product-may-not-be-as-harm-free-as-claimed/ |laysource=BMJ |year=2018 |last1=Davis |first1=Barbara |title=IQOS: Evidence of pyrolysis and release of a toxicant from plastic |journal=Tobacco Control |pages=tobaccocontrol-2017-054104 |last2=Williams |first2=Monique|last3=Talbot |first3=Prue |doi=10.1136/tobaccocontrol-2017-054104 |doi-broken-date=2018-10-31 |quote=Charring due to pyrolysis (a form of organic matter thermochemical decomposition) was observed in the tobacco plug after use. When the manufacturer’s cleaning instructions were followed, both charring of the tobacco plug and melting of the polymer-film filter increased. Headspace analysis of the polymer-film filter revealed the release of formaldehyde cyanohydrin at 90°C, which is well below the maximum temperature reached during normal usage.}}</ref> and the carbon-rich residue is called [[char]].<ref name="char_character">{{cite journal |last1=Sharma |first1=Ramesh K. |last2=Wooten |first2=Jan B. |last3=Baliga |first3=Vicki L. |last4=Martoglio-Smith |first4=Pamela A. |last5=Hajaligol |first5=Mohammad R. |title=Characterization of char from the pyrolysis of tobacco |journal=Journal of Agricultural and Food Chemistry |date=13 February 2002 |volume=50 |issue=4 |pages=771–783 |pmid=11829644 |issn=0021-8561}}</ref> |
|||
Heat-not-burn tobacco products are a [[battery (electricity)|battery]]-powered systems that produce nicotine-containing emissions by heating tobacco.<ref name=PieperMallock2018/> For this purpose, tobacco sticks are placed in a corresponding heater and heated to about 250–350 °C (around 500 °F.<ref name=Caputi2016/>).<ref name=PieperMallock2018/> This results in nicotine-containing emissions, which are inhaled via a mouthpiece with a filter segment.<ref name=PieperMallock2018/> Heat-not-burn tobacco products heat tobacco leaves at a lower temperature than traditional cigarettes.<ref name=LiSaad2018/> Heat-not-burn tobacco products usually heat up tobacco, rather than use liquids.{{sfn|McNeill|2018|p=201}} Another type of heat-not-burn tobacco product is the loose-leaf tobacco [[vaporizer (inhalation device)|vaporizer]] that entails putting loose-leaf tobacco into a chamber, which is electrically heated using an element.<ref name=LopezHiler2016/> Some use product-specific customized cigarettes.<ref name=WHO2018/> They are not e-cigarettes.<ref name=WHO2018/> They can overlap with e-cigarettes such as combining an e-cigarette and a heat-not-burn tobacco product, for using tobacco or [[construction of electronic cigarettes#E-cigarette liquid|e-liquid]].{{sfn|MHNZ|2017|p=4}} |
|||
HnB products vary, but can heat to these charring temperatures<!--temperatures hot enough to release smoke, but not most of the carbon-->.<ref name=constituents_pyrolysis_quote/><ref name="char"/><ref name=WHO2018/> The composition of the smoke is changed by lower temperatures: levels of some substances fall, while levels of others rise.<ref name=constituents>{{cite journal |doi=10.1136/tobaccocontrol-2018-054321 |pmid=30158205 |title=IQOS: Examination of Philip Morris International's claim of reduced exposure |journal=Tobacco Control |volume=27 |issue=Suppl 1 |pages=s30–s36 |year=2018 |last1=St.Helen |first1=Gideon |last2=Jacob Iii |first2=Peyton |last3=Nardone |first3=Natalie |last4=Benowitz |first4=Neal L. }}</ref> While the nicotine is the main addictive component in tobacco, some pyrolysis products of tobacco are thought to reinforce addiction (such as [[acetaldehyde]], [[norharman]], and [[Harmane|harman]]),<ref name=Bruijnzeel2012>{{cite journal |doi=10.1016/j.neubiorev.2012.02.015 |pmid=22405889 |pmc=3340450 |title=Tobacco addiction and the dysregulation of brain stress systems |journal=Neuroscience & Biobehavioral Reviews |volume=36 |issue=5 |pages=1418–1441 |year=2012 |last1=Bruijnzeel |first1=Adrie W. }}</ref><ref>{{cite journal |doi=10.1093/ntr/nts192 |pmid=22990226 |title=Abuse Potential of Non-Nicotine Tobacco Smoke Components: Acetaldehyde, Nornicotine, Cotinine, and Anabasine |journal=Nicotine & Tobacco Research |volume=15 |issue=3 |pages=622–632 |year=2013 |last1=Hoffman |first1=A. C. |last2=Evans |first2=S. E. }}</ref> some of which have been found in lower levels in the smoke of HnB products.<ref>{{cite journal |doi=10.1111/add.14365 |pmid=29920842 |title=Carbonyl emissions from a novel heated tobacco product (IQOS): Comparison with an e-cigarette and a tobacco cigarette |journal=Addiction |volume=113 |issue=11 |pages=2099–2106 |year=2018 |last1=Farsalinos |first1=Konstantinos E. |last2=Yannovits |first2=Nikoletta |last3=Sarri |first3=Theoni |last4=Voudris |first4=Vassilis |last5=Poulas |first5=Konstantinos |last6=Leischow |first6=Scott J. }}</ref> |
|||
These products are marketed as "heat-not-burn"<ref name="marketing">{{cite journal |last1=Staal |first1=Yvonne CM |last2=van de Nobelen |first2=Suzanne |last3=Havermans |first3=Anne |last4=Talhout |first4=Reinskje |title=New Tobacco and Tobacco-Related Products: Early Detection of Product Development, Marketing Strategies, and Consumer Interest |journal=JMIR Public Health and Surveillance |date=28 May 2018 |volume=4 |issue=2 |pages=60–6 |doi=10.2196/publichealth.7359 |pmid=5996176 |pmc=5996176 |issn=2369-2960}}</ref> and "smoke-free".<ref name=prohibited>{{Cite news| issn = 1170-0777| title = Tobacco company charged over importing prohibited product| work = NZ Herald| accessdate = 2018-06-06| date = 2017-05-18| url = https://www.nzherald.co.nz/nz/news/article.cfm?c_id=1&objectid=11858402}}</ref> However, independent researchers who tested a common "heat-not-burn" device explicitly disagreed with the claim that they are smokeless,<ref name=better_query/><ref name=npr_FDA>{{Cite web| title = FDA Panel Gives Qualified Support To Claims For 'Safer' Smoking Device| work = NPR.org| accessdate = 2018-06-04| url = https://www.npr.org/sections/health-shots/2018/01/25/580213742/fda-panel-gives-qualified-support-to-claims-for-safer-smoking-device}}</ref> arguing that the emitted aerosol is smoke, as it contains pyrolysis products.<ref>{{cite journal |doi=10.1001/jamainternmed.2017.5861 |pmid=29114801 |title=Perplexing Conclusions Concerning Heat-Not-Burn Tobacco Cigarettes—Reply |journal=JAMA Internal Medicine |volume=177 |issue=11 |pages=1699–1700 |year=2017 |last1=Auer |first1=Reto |last2=Cornuz |first2=Jacques |last3=Berthet |first3=Aurélie }}</ref> Independent researchers studying the [[aerosols]] produced by heat-not-burn products commonly call those aerosols "smoke".<ref>{{cite journal |last1=Mallock |first1=Nadja |last2=Böss |first2=Lisa |last3=Burk |first3=Robert |last4=Danziger |first4=Martin |last5=Welsch |first5=Tanja |last6=Hahn |first6=Harald |last7=Trieu |first7=Hai-Linh |last8=Hahn |first8=Jürgen |last9=Pieper |first9=Elke |last10=Henkler-Stephani |first10=Frank |last11=Hutzler |first11=Christoph |last12=Luch |first12=Andreas |title=Levels of selected analytes in the emissions of "heat not burn" tobacco products that are relevant to assess human health risks |journal=Archives of Toxicology |date=2018 |volume=92 |issue=6 |pages=2145–2149 |doi=10.1007/s00204-018-2215-y |pmid=29730817 |pmc=6002459 |issn=0340-5761}}, and sources therein</ref> Independent research has also disputed the claim that the products are "heat-not-burn" devices.<ref name="char"/><ref name=strategy>{{cite journal |doi=10.1136/tobaccocontrol-2018-054340 |pmid=30209207 |pmc=6202178 |title=Heated tobacco products: Another tobacco industry global strategy to slow progress in tobacco control |journal=Tobacco Control |volume=27 |issue=Suppl 1 |pages=s111–s117 |year=2018 |last1=Bialous |first1=Stella A. |last2=Glantz |first2=Stanton A. }}</ref> |
|||
== History == |
== History == |
||
Revision as of 16:29, 9 January 2019
An electrically-heated smoking system, also known as a heated tobacco product or heat-not-burn tobacco product (HNB), uses an electric heating element[1] to char tobacco,[2] at a lower temperature than a conventional cigarette.[3] The result is a smoke that contains nicotine, other chemicals, and particulates.[1][4]: Table 1 These products may match some of the behavioral aspects of smoking.[5] Some tobacco companies claim these products are less harmful to consumers than other types of cigarettes, but there is no reliable evidence to support these claims.[3][6]
Some of these products take loose-leaf tobacco in a heated chamber; others require product-specific cigarettes.[7][1] Products heating cigarettes using an external heat source first came to market in 1988, however they were not a commercial success.[3] These products are currently being re-introduced by large tobacco companies.[8]
Health effects

There is not enough research to evaluate the level of harm of these products.[9] A 2016 Cochrane review found that it was unclear whether using these products instead of conventional cigarettes would "substantially alter the risk of harm".[10] Also in 2016, the WHO noted that some scientists believe these products to be as harmful as traditional cigarettes, and stated that no convincing evidence had been presented for industry claims of lowered risk and health benefits. Independent research is not available to support these claims; they are based on industry-funded research.[6] Independent 2018 reanalysis of data from industry research has found deficiencies and omissions in the evidence used to support the industry's claims.[11][4][12]
Action on Smoking and Health stated in 2016 that due to "the tobacco industry's long record of deceit" regarding the health risks involving smoking, it is important to conduct independent studies into the health effects of these products.[13] Carlos Jiménez, director of research on smoking at the Spanish Society of Pneumonology and Thoracic Surgery, criticized the industry data available in 2017.[14]
The effects of second-hand exposure are unknown.[1]
Addiction and quitting
Such products are believed to be just as addictive as conventional cigarettes.[15] Nearly half of people using these products had never used conventional cigarettes,[16] according to a small survey done in Italy.[17] This has caused concern that the products might cause nicotine addiction rather than reduce harm to those who already smoke.[16]
There is not enough evidence to know if HnB products help with quitting smoking.[18] In one manufacturer-led study, smokers using Hnb products mostly did not stop using regular cigarettes; they mostly used both, although the HnB products were supplied to them for free.[11]
Pregnancy
There is no information on the effects of smoking HnB devices during pregnancy, as of 2018.[18] However, they are nicotine-containing products.[1] It is unsafe to use any product containing nicotine during pregnancy and breastfeeding,[19] as nicotine harms the fetus.[20] Nicotine use during pregnancy increases the child's risk of respiratory problems, neurobehavioral defects, obesity, type 2 diabetes, hypertension, and infertility.[21]
Construction
-
Tobacco stick; above, disassembled, below, intact.[22] A: Reconstituted tobacco film, made of dried tobacco suspension.[22] 70% tobacco, humectants (water and glycerin) to encourage aerosol formation, binding agents, and aroma agents.[22] B: Hollow acetate tube.[22] C: Polymer film filter cools the aerosol.[22] D: Soft cellulose acetate mouthpiece, which mimics the feel of a traditional cigarette.[22]
Nicotine is released from tobacco heated above 150 °C.[23] Combustible tobacco cigarettes reach about 900 °C during a puff and smoulder at about 400 °C between puffs.[24] The burning process, substances emitted and their levels vary at different temperatures.[24] Distillation, the process during which nicotine and aromas are transferred from tobacco to smoke, occurs below 300 °C; pyrolysis occurs at about 300 °C–700 °C, entails the decomposition of biopolymers, proteins, and other organic materials and generates the majority of substances emitted in smoke; and combustion occurs above 750 °C and results in the generation of carbon dioxide, carbon monoxide, and water.[24] HeatSticks are heated to a maximum of 350 °C, a temperature sufficient to enable pyrolytic decomposition of some organic materials.[24] Formation of toxic volatile organic compounds, including formaldehyde, acetaldehyde and acrolein, via dehydration and oxidation of the humectants, propylene glycol and glycerin, have been reported in e-cigarette aerosols at similar temperatures as IQOS.[24] In addition, flavoring chemicals in e-cigarettes undergo thermal degradation and contribute significantly to levels of toxic aldehydes emitted in e-cigarette aerosol.[24] Since the constituents of HeatSticks may be different from that of combustible cigarettes, including flavorants and additives, it is plausible that the IQOS aerosol may contain substances not present in tobacco smoke.[24] The IQOS HeatSticks do not generate a flame, they are charred following use.[25] Heat-not-burn tobacco products may or may not generate smoke.[26]
The heat-not-burn tobacco product consists of 3 components with different functions.[22] These include the tobacco stick with processed tobacco, a pen-like heater (holder), in which the tobacco stick is inserted, which is then heated by means of an electrically controlled heating element, and a charger (the charger), which recharges the heater after use.[22] The heat-not-burn tobacco products automatically stops the heating process after 6 minutes or 14 moves, so that pyrolysis products and pollutant release are limited in time as well as by a maximum number of puffs per stick.[22] The tobacco stick contains a compressed tobacco film as well as several filter elements.[22] The tobacco film consists of a dried tobacco suspension that has been rolled up into a paper-thin brown tobacco foil.[22] This consists of about 70% tobacco as well as humectants, binders and flavorings.[22] Water and glycerin are used as humectants to prevent drying out and to promote aerosol formation upon heating.[22] The filter elements consist of 2 independent systems: A polymer film filter that cools the aerosol, followed by a soft cellulose acetate mouthpiece filter that mimics the sensory aspects of a traditional cigarette.[22]
Heat-not-burn tobacco products are a battery-powered systems that produce nicotine-containing emissions by heating tobacco.[22] For this purpose, tobacco sticks are placed in a corresponding heater and heated to about 250–350 °C (around 500 °F.[27]).[22] This results in nicotine-containing emissions, which are inhaled via a mouthpiece with a filter segment.[22] Heat-not-burn tobacco products heat tobacco leaves at a lower temperature than traditional cigarettes.[28] Heat-not-burn tobacco products usually heat up tobacco, rather than use liquids.[29] Another type of heat-not-burn tobacco product is the loose-leaf tobacco vaporizer that entails putting loose-leaf tobacco into a chamber, which is electrically heated using an element.[7] Some use product-specific customized cigarettes.[1] They are not e-cigarettes.[1] They can overlap with e-cigarettes such as combining an e-cigarette and a heat-not-burn tobacco product, for using tobacco or e-liquid.[30]
History
As early as the 1960s, the tobacco companies developed alternative tobacco products with the goal of supplementing the cigarette market with products.[31] The first commercial heat-not-burn product was the R. J. Reynolds Premier,[32] a smokeless cigarette launched in 1988 and described as difficult to use.[33] Many smokers disliked the taste.[34] It was shaped like a traditional cigarette, and when heated the smoldered charcoal moved past processed tobacco containing more than 50 percent glycerin to create an aerosol of nicotine.[35] It did require some combustion.[36] In 1989,[37] after spending $325 million,[38] R. J. Reynolds pulled it from the market months later after organizations recommended to the US Food and Drug Administration (FDA) to restrict it or classify it as a drug.[39]
The Premier product concept went on to be further developed and re-launched as Eclipse[37] in the mid-1990s,[40] and was available in limited distribution as of 2015.[41] Reynolds American stated that the Revo was a "repositioning" of its Eclipse.[42] R. J. Reynolds' Revo was withdrawn in 2015.[41]
Philip Morris International launched a cigarette in 1998 that was placed into an electronic heating device as Accord.[43] Also in 1998 the company launched Accord in Osaka, Japan, calling it Oasis.[44] The battery-powered product was the size of a pager.[45] The product was marketed as "low-smoke".[44] Advertisements stating reduced risk were drafted for Accord in the US, but were never released.[44] An attempt was made in 2007 by Kenneth Podraza, who was the Vice President of Research and Development at Philip Morris in the US, to get the Surgeon General of the United States to endorse it.[44] It has not been shown that the Surgeon General replied to Podraza's letter.[44] Few people started using the Accord, and most users also continued to use traditional cigarettes.[44] The Accord ceased production in 2006, eight years after it came on the market.[44]
In 2007 Philip Morris International launched Heatbar;[46] which was very similar to the Accord.[44] The Heatbar was around the size of a mobile phone and was said to heat specifically designed cigarettes rather than burning them.[47] The only benefit was to lower second-hand smoke, which lead to Heatbar being discontinued.[48] Heatbar did not obtain any significant user reception.[49] Accord and Heatbar are predecessors of Philip Morris International's heat-not-burn tobacco products.[50] Heat-not-burn tobacco products were not a commercial success, and most of them were quickly taken off the market following their debut.[27]
In recent years leading up to 2018, increased tobacco control measures have directed the tobacco industry to develop alternative tobacco products, such as heat-not-burn tobacco products.[22] There has been a global decline in tobacco consumption that, if continued, will negatively impact the tobacco industry's profits.[31] This decline led the industry to invent and market new products, such as heat-not-burn tobacco products.[31] The introduction of these heat-not-burn tobacco products may also have been a response to the growing popularity of e-cigarettes beginning around 2007 after independent companies introduced them before the major multinational tobacco companies entered the e-cigarettes market.[31] Furthermore, the global decline of cigarette consumption and decrease in adult smoking prevalence (from 24% in 2007 to 21% in 2015), combined with the success of tobacco control, including implementation of the WHO Framework Convention on Tobacco Control, may also have lead the tobacco companies to consider alternative products to protect their profits and political interests.[31] The ubiquitousness of e-cigarettes and growing dissatisfaction with they do not provide a throat-hit may present an opportunity for heat-not-burn tobacco products.[27] These products are have been introduced by large tobacco companies.[8] Philip Morris International anticipates a future without traditional cigarettes, but campaigners and industry analysts call into question the probability of traditional cigarettes being dissolved, by either e-cigarettes or other products like IQOS.[51]
Products
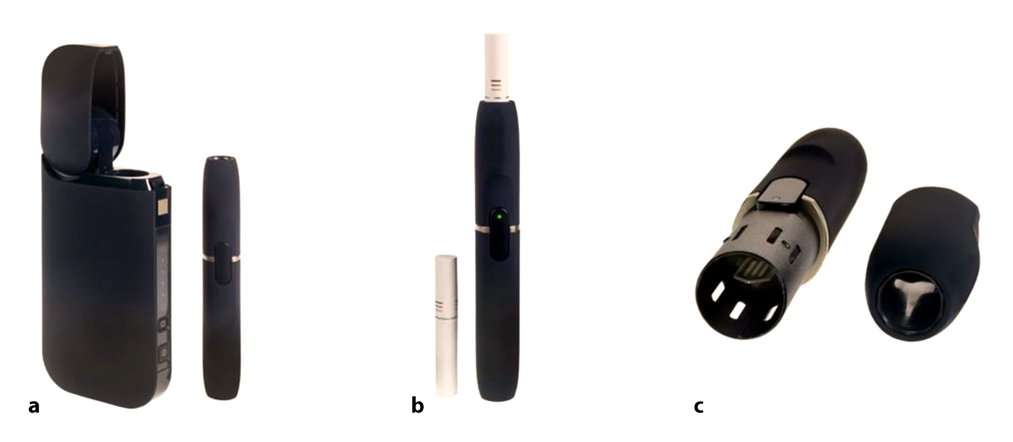
-
Electrically-heated tobacco system. a) Charger (left) and holder (right), b) Tobacco stick (left) and holder with tobacco stick inserted (right), c) Disassembled holder, with heating element visible (left) and the holder's lid (right).[22]
The products use a heating-system where the tobacco is heated and aerosolized.[1] In addition to nicotine, they contain additives not derived from tobacco, and are frequently flavored.[1] The evidence indicates that the concentrations of nicotine in mainstream heat-not-burn tobacco products aerosol are less than what is found in cigarette smoke.[52] Smokers regularly reported heat-not-burn tobacco product use to be less satisfying than smoking a cigarette.[53] The heat-not-burn tobacco products that were tested provided more nicotine in the aerosol than a cigalike e-cigarette but not as much nicotine compared with a tank style e-cigarette.[53] They are designed to be similar to their combustible counterparts[42] and they provide some of the behavioral aspects of smoking.[5] These products replicate the oral inhalation and exhalation, taste, rapid systemic delivery of nicotine, hand-to-mouth feel and throat hit sensations (depending on the temperature) that are similar to smoking traditional cigarettes.[28] There are different types of heat-not-burn tobacco products in the marketplace.[54]
3T

The 3T from Vapor Tobacco Manufacturing was launched in December 2014.[55] The product employs a patented, aqueous system whereby desired components are extracted into water.[56] The liquid is mixed with glycerin and aerosolized by an electronic heating system.[56] Their organic liquids are made from organic tobacco, organic glycerin, and water.[55]
Firefly 2
The Firefly developed the Firefly 2, which heats loose-leaf plant material and concentrates and is often used to vaporize marihuana.[57]
glo
In 2016, British American Tobacco launched a battery-powered heat-not-burn product called glo in Japan.[58] It is also sold in South Korea, Switzerland, and Russia.[59] It uses a heating element with a tobacco stick,[42] which looks similar to a short cigarette.[59] In May 2017 they released i-glo in Canada.[60] The glo iFuse debuted in Romania by British American Tobacco in 2015.[42] It uses a cartridge with a tobacco stick and a flavored nicotine liquid.[42] It is a hybrid of an heat-not-burn tobacco product and an e-cigarette.[24] It consists of a heating element, a liquid tank (like e-cigarettes) and a tobacco cavity through which the e-cigarette-like aerosol passes and is infused with tobacco flavor.[24] Bonnie Herzog, a senior analyst at Wells Fargo Securities stated that the proposed acquisition of R. J. Reynolds by British American Tobacco in 2016 would let them catch up in the technology competition.[61] The data on glo is limited.[62]
IQOS

The introduction of IQOS (/ˈaɪkoʊs/ EYE-kohs[63]) was announced on June 26, 2014.[64] Some have stated IQOS stands for "I quit ordinary smoking", according to The Express Tribune.[63] IQOS was not intended to be an acronym for "I Quit Ordinary Smoking," according to chief executive officer André Calantzopoulos of Philip Morris International.[65] The company explained that the name began with a lowercase "i," then changed into "IQ" together with "OS," has no specific meaning.[65] The product is marketed by Philip Morris International under the Marlboro and Parliament brands.[66] Although it is marketed as a novel product, it is very similar to the "Accord" product released by the same company in 1998; however, the IQOS products have more nicotine, more tar, and less tobacco.[44] They are heated to a lower temperature, and the kit costs about US$40 more in 2018 dollars.[44]
Initially launched in 2014 in Nagoya, Japan and Milan, Italy, IQOS is being gradually rolled out to other countries.[67] By the end of 2016, it was available in over twenty countries, with expansion plans into several more in 2017 as manufacturing capacity increases.[68] As of May 2018, IQOS is available in over 37 countries.[24] Philip Morris International began selling IQOS and HEETS modified cigarettes in two of its stores in Seoul, Korea on May 27, 2017.[69] On June 5, 2017, other retailers in Seoul including CU and Electromart began offering them.[69] As of 2018 IQOS is not approved for sale in the US.[70] Philip Morris International has projected that when 30 billions units are sold, IQOS would increase profits by $700 million.[71] In December 2017, the company launched TEEPS in the Dominican Republic.[72] It is a heat-not-burn product that looks similar to a traditional cigarette.[72]
In the UK, IQOS has been sold at an exclusive shop in London since December 2016 and later it became available online and at a few other retailers in London.[29] Outside of an IQOS retail shop in Canada, marketing included a sign with the message, 'Building a Smoke-Free Future'.[73] The packaging of IQOS is similar to iPhones and other upscale smartphones.[74] The IQOS is marketed as a "smoke-free" alternative to traditional cigarettes, and promoted as a way to lower risk from smoking.[75] Philip Morris International has been intensively promoting its IQOS product in Europe and Asia, as of 2017.[76] IQOS has over 1.4 million frequent users, according to the company, as of 2017.[65]
As of 2016, the company stated total investments made in the development and assessment of these products have exceeded $3 billion.[77] Phillip Morris spent €500 million on IQOS in 2016 alone.[78] Papastratos, Philip Morris International's division in Greece, intends to revamp a cigarette factory into a manufacturer of tobacco sticks for use with IQOS products.[79] Philip Morris International plans to invest in 2018 $320 million to build a second manufacturing plant in Dresden, Germany.[60] IQOS is probably going to be a major part of Philip Morris International's expansion strategy.[60]
The IQOS product consists of a charger around the size of a mobile phone and a holder that looks like a pen.[80] The product can collect personal data in regard to the smoking habits of the user.[81] Philip Morris International stated it only retrieves the data when the product is not working properly.[81] The disposable tobacco stick[82] called HeatSticks or HEETS in some places they are sold,[83] looks similar to a short cigarette.[59] The sticks contain processed tobacco and has been soaked in propylene glycol.[82] The stick is inserted into the holder which then heats it to temperatures up to 350 °C.[43] The smoke[82] and aerosol released contains nicotine, tobacco and other chemicals.[1] The amount of nicotine provided may be a little strong for light cigarette smokers.[84] The sticks are available in regular, balanced regular, menthol and mint flavor.[84] Users have reported less smell and odor on clothing.[36] Philip Morris International states that IQOS generates no smoke because the tobacco does not combust and the stick is heated rather than burned.[82] Even without fire, smoke can be produced.[82] Both IQOS and traditional cigarettes do not completely combust (pyrolysis) tobacco.[82] The emissions generated by IQOS contains substances from pyrolysis and thermogenic degradation that are identical to the constituents found in traditional tobacco cigarette smoke.[82] The emissions generated by IQOS contains the identical harmful constituents as cigarette smoke, including volatile organic compounds at comparable levels to cigarette smoke, polycyclic aromatic hydrocarbons at vast various ranges, and carbon monoxide.[85] All of these substances, on the basis of rigorous research of cigarette smoke, are known to cause significant harms to health.[85] A 2017 review found "little research on what substances are released after the device heats the tobacco-based paste. The physical effects on users are also not yet known."[76] Carlos Jiménez, director of research on smoking at the Spanish Society of Pneumonology and Thoracic Surgery stated in 2017 that the IQOS product is still harmful.[86] IQOS is likely less toxic than traditional cigarettes.[87]
A 2017 independent study of the IQOS states that "Dancing around the definition of smoke to avoid indoor-smoking bans is unethical" and called for more independent research, stating "Smokers and non-smokers need accurate information about toxic compounds released in IQOS smoke. This information should come from sources independent of the tobacco industry".[82] In 2017, according to two editors of the journal JAMA Internal Medicine, after publication of a research letter describing harmful chemicals in heat-not-burn tobacco products, people from Philip Morris International contacted the institutions where the researchers worked and questioned the methods used in the study; the editors described this as a form of "pressure to suppress discourse that could harm commercial interests".[88] Philip Morris International asked the University of Lausanne to retract the study.[89] A spokeswoman for the University of Lausanne stated in an email that following the release of their study, the heads of the University of Bern, Lausanne University Hospital, and University of Lausanne where the authors worked received letters from Phillip Morris International, criticizing the authors for using a flawed methodology in the study.[90] When contacted by a journalist from The Washington Post, the researchers declined to comment.[90] Philip Morris International had published an academic counter-argument on the Internet.[90] Philip Morris International told CBS This Morning in January 2018 that the IQOS product produces no smoke.[91] Reto Auer, who directed the 2017 study, said in an interview prior to the US FDA two-day hearing, "We disagree with the claim that it's smokeless."[92] The FDA stated in January 2018 that "There are significant analytical issues in the Auer et al. study, such as lack of testing reference samples, low number of replicates, lack of selectivity on some analytical methods."[93] In December 2017, Reuters published documents and testimonies of former employees detailing irregularities in the clinical trials conducted by Philip Morris International for the approval of the IQOS product by the US FDA.[94]
On December 5, 2016,[95] Philip Morris International submitted a multi-million page application[78] to the US FDA for IQOS to be authorized as a modified risk tobacco product.[95] Then in late March of 2017, Philip Morris International submitted to the US FDA for a premarket tobacco product application regarding its iQOS product.[60] In May 2017, Philip Morris International had received notice that the US FDA had started a lengthy scientific review process for the IQOS product.[60] On May 24, 2017, the US FDA had published papers acknowledging Philip Morris International's modified risk tobacco product application regarding the IQOS product.[96]
The advisory panel appointed by the US FDA reviewed Philip Morris International’s application in January 2018.[92] Matthew Myers, representing the Campaign for Tobacco-Free Kids, told the US FDA advisory panel that "It is high-tech. It is sleek. It is designed in exactly the way that would appeal to young people."[92] Jeff Fortenbacher, CEO of Access Health, stated that "Patients who smoke clearly need more tools to help them quit."[92] The advisory panel voted in favor of the claim that IQOS reduced users' exposure to harmful chemicals.[92] The US FDA advisory panel stated that Philip Morris International did not demonstrate that the product reduces the risks of diseases associated with tobacco use.[97] Philip Morris International's claim that "switching completely to iQOS presents less risk of harm than continuing to smoke cigarettes" did not gain support by the US FDA advisory panel.[92] The panel also "expressed concerns about the lack of data" on risk relative to traditional cigarettes.[98] The US FDA reviewed Phillip Morris International's data, independent studies, including a 2017 study on IQOS and a posted comment on Phillip Morris International's website about the 2017 study, a December 8, 2017 amendment to the application by Phillip Morris International on the same topic, and the US FDA's own laboratory testing data.[93] The FDA intends to continue to review Philip Morris International's research.[99]
Philip Morris International intends to convert its customers in Japan to using heat-not-burn products.[100] The IQOS products are sold as an alternative to regular cigarettes.[88] Philip Morris International conducted studies using their IQOS product.[74] The participants in the studies who were given the IQOS product were not likely to quit using traditional cigarettes.[74] In 2016 PMI acknowledged that the IQOS product is probably as addictive as tobacco smoking.[13] IQOS is sold with a warning that the best option is to avoid tobacco use altogether.[101]
Comparison to traditional cigarettes
| Analyzed Substance | IQOS product; Amount, Mean (SD) | Duplicate tests for given assay | Traditional cigarettes; Amount, Mean (SD) | Duplicate tests for given assay | Percentage (%) of the substance in each IQOS compared to traditional cigarettes |
|---|---|---|---|---|---|
| Volatile organic substances, μg per cigarette: 1 | |||||
| Acetaldehyde | 133 (35) | 5 | 610: 2 | 1 | 22 |
| Acetone | 12.0 (12.9) | 5 | 95.5 (13.5) | 2 | 13 |
| Acroleine | 0.9 (0.6) | 2 | 1.1 | 1 | 82 |
| Benzaldehyde | 1.2 (1.4) | 5 | 2.4 (2.6) | 2 | 50 |
| Crotonaldehyde | 0.7 (0.9) | 5 | 17.4 | 1 | 4 |
| Formaldehyde | 3.2 (2.7) | 5 | 4.3 (0.4) | 2 | 74 |
| Isovaleraldehyde | 3.5 (3.1) | 5 | 8.5 (10.8) | 2 | 41 |
| Propionaldehyde | 7.8 (4.3) | 5 | 29.6 (36.6) | 2 | 26 |
| Polycyclic aromatic hydrocarbons, ng per cigarette: 3 | |||||
| Naphthalene | 1.6 (0.5) | 4 | 1105 (269) | 7 | 0.1 |
| Acenaphthylene | 1.9 (0.6) | 4 | 235 (39) | 7 | 0.8 |
| Acenaphthene | 145 (54) | 4 | 49 (9) | 7 | 295 |
| Fluorene | 1.5 (0.6) | 4 | 371 (56) | 7 | 0.4 |
| Anthracene | 0.3 (0.1) | 4 | 130 (18) | 7 | 0.2 |
| Phenanthrene | 2.0 (0.2) | 4 | 292 (44) | 7 | 0.7 |
| Fluoranthene | 7.3 (1.1) | 4 | 123 (18) | 7 | 6 |
| Pyrene | 6.4 (1.1) | 4 | 89 (15) | 7 | 7 |
| Benz[a]anthracene | 1.8 (0.4) | 4 | 33 (4.2) | 7 | 6 |
| Chrysene | 1.5 (0.3) | 4 | 48 (6.2) | 7 | 3 |
| Benzo[b]fluoranthene | 0.5 (0.2) | 4 | 24 (2.9) | 7 | 2 |
| Benzo[k]fluoranthene | 0.4 (0.2) | 4 | 4.3 (2.8) | 7 | 9 |
| Benzo[a]pyrene | 0.8 (0.1) | 4 | 20 (2.9) | 7 | 4 |
| Indeno[1,2,3-cd]pyrene | ND | 4 | NA | NA | NA |
| Benzo[ghi]perylene | ND | 4 | NA | NA | NA |
| Dibenzo[a,h]anthracene | ND | 4 | NA | NA | NA |
| Inorganics, ppm in the mainstream smoked: 4 | |||||
| Carbon dioxide | 3057 (532) | 5 | >9000 | 3 | NA |
| Carbon monoxide | 328 (76) | 5 | >2000 | 3 | NA |
| Nitric oxide | 5.5 (1.5) | 5 | 89.4 (71.6) | 3 | 6 |
| Other evaluations | |||||
| Nicotine, μg per cigarette: 1 | 301 (213) | 4 | 361 | 1 | 84 |
| Temperature, °C | 330 (10) | 2 | 684 (197) | 1 | NA |
| Number of puffs | 12.6 (2.4) | 32 | 13.3 (3.1) | 6 | NA |
Abbreviations: NA, not analyzed; ND, not detected.[82] : 1 The techniques applied were presented earlier in Varlet et al([102]) to analyze volatile organic compounds and nicotine.[82] : 2 Due to there being one duplicate test, no SD can be determined.[82] : 3 The values presented were illustrated from Vu et al([103]) for the ISO smoking regimen and for an average of the 35 highest selling US traditional cigarette brands.[82] : 4 Carbon dioxide was assessed with a Testo 535 (Testo), and carbon monoxide and nitric oxide were assessed with a Pac 7000 that identified carbon monoxide (Draeger).[82] The apparatus calculated the smoke whenever generated from the syringe pump.[82]
∗A 2017 analysis comparing IQOS to popular US sold traditional cigarettes.[82]
iSmoke OneHitter
iSmoke OneHitter by iSmoke was launched in 2015.[104] It can be used as a loose-leaf tobacco vaporizer.[105] It has a chamber that can be filled with up to 800-milligrams of tobacco.[105]
IUOC
IUOC, marketed by Shenzhen Yukan Technology Co., Limited from China, can be used with any brand of cigarettes.[106] Users insert the entire cigarette into the product.[106] It does not use a tobacco stick.[106] The temperature reaches up to 395 °C when in use.[106] IUOC can smoke around 10 cigarettes per charge.[106]
lil
Korea Tobacco & Ginseng Corporation announced on June 8, 2017 that they will launch a heat-not-burn tobacco product in September 2017.[107] The heat-not-burn tobacco product named lil formally launched in November 2017.[108] The battery-operated product employs heat to the tobacco leaves.[108]
Pax 2
PAX Labs, formerly known as Ploom,[109] sells PAX vaporizers.[110] In 2010 they launched Ploom, a butane-powered product used for the heating tobacco or botanical products.[111] Later models replaced butane heating with an electric system.[112] After its initial partnership with Japan Tobacco was abandoned, the company became known as Pax Labs.[113] The Pax 2 uses loose-leaf tobacco.[42] The surface of the Pax 2 remains cool, while the oven heats to temperatures up to 455 °F.[114] It has four temperature options.[114]
Ploom Tech
In January 2016, Japan Tobacco released Ploom Tech.[115] Japan Tobacco's Ploom has been withdrawn from the US.[42] The Ploom brand, however, remained with Japan Tobacco and the product itself has been replaced with a different product called Ploom Tech, in which an aerosol passes through a capsule of granulated tobacco leaves.[116] Sales are being expanded throughout Japan in 2017.[117] They intend to spend $500 million to increase their heated tobacco manufacturing capacity by late 2018.[118] Studies on Japan Tobacco International's Ploom product has not been found.[62]
V2 Pro
V2 originally released their vaporizer line named V2 Pro in July 2014.[119] The initial product was named Series 3.[119] Series 3 comes with 3 cartridges including a loose-leaf cartridge, which heats the material by conduction.[120] It comes with a battery and USB changer, among other things.[120] Pro Series 3X also by V2 can be used with dry material.[121] It has three different air flow options that can be adjusted with a slight turn of the mouthpiece.[121] Series 7 comes with a loose-leaf cartridge, among other things.[122] Series 7 lets the user change the temperature by using a single button.[119]
Prevalence
Since mid-2017, heat-not-burn tobacco products were sold in 27 countries.[123] As of 2017, the market for such products is anticipated to expand by 60% over the next 10 to 15 years.[124]
Marketing
The term "heat-not-burn" refers to tobacco heated (at ~350 °C) by an electrically-powered element or carbon, not combusted (at ~800 °C).[28] Terms used in marketing of cigarette-like products that "heat rather than burn" are referring to the product as "reduced risk" and "innovative."[57] Marketing slogans like "heat-not-burn" cannot be a substitute for science.[82] Heat-not-burn tobacco products are not typically marketed as a harmless substitute to smoking.[125]
The tobacco companies are using a series of claims in the marketing of heat-not-burn tobacco products.[31] Both in websites and statements to the media and investors, heat-not-burn tobacco products are presented as less harmful but not risk-free.[31] In a few instances, marketing materials claim that heat-not-burn tobacco products are potentially helpful to smokers who want to quit.[31] Some media accounts of product launches state that heat-not-burn tobacco products reduce the levels of harmful tobacco components by 90%–95% compared with traditional cigarettes, while others emphasise the lack of odor or visible emissions as part of marketing campaigns.[31] As of April 2018, there is no evidence to confirm this claimed 90%–95% lower level of harm.[31] Other marketing claims highlight that these products produce no smoke, that is, are smoke-free.[31] Implied in these claims, in advertisements and stores globally, is that smokers should switch from traditional cigarettes to these new, allegedly less harmful, products.[31] The introduction of the latest generation of heat-not-burn tobacco products appears to be the latest chapter in the decades-old tobacco industry strategy of working to create partnerships with governments and health advocates, presenting these alleged 'harm reduction' products as an option to address the tobacco epidemic.[31]
The tobacco companies use heat-not-burn tobacco products as part of their broader political and public relations activities to position them as 'partners' to address the tobacco epidemic rather than as the vectors that are causing it.[31] This is a similar strategy previously used by the tobacco industry to promote itself as a partner of public health in reducing the harms of tobacco, while obfuscating the scientific evidence pointing that harm reduction is achieved through tobacco control policies that decrease consumption.[31]
Regulation
Current smoking bans may not have been extended to include such products.[126] In the majority of the countries in which they have been sold, heat-not-burn tobacco products have been taxed at a lower rate than traditional cigarettes.[127]
"There is concern that heat-not-burn tobacco will skirt local ordinances that prevent smoking in public areas," Mitchell H. Katz, director of the Los Angeles County Health Agency, wrote in 2017.[126] Action on Smoking and Health stated in 2016 that "unless and until independent evidence shows that IQOS and similar products are substantially less harmful than smoking then these products should be regulated in the same way as other tobacco products."[13] Tobacco control activist Stanton Glantz stated that the US FDA should halt new tobacco products until tobacco companies stop selling traditional cigarettes.[128] It is recommended that indoor-smoking bans for traditional cigarettes be extended to heat-not-burn tobacco products.[82]
In the United States, these products fall under the jurisdiction of the Food and Drug Administration as amended by the Family Smoking Prevention and Tobacco Control Act of 2016.[7]
Advertisement for the IQOS product itself is not regulated under the European Union Tobacco Products Directive.[42] Advertising for IQOS' tobacco stick may fall under the European Union Tobacco Products Directive.[42] The UK government has been looking into creating a separate category for taxing heat-not-burn tobacco products.[29]
Due to the alleged belief in heat-not-burn tobacco harm reduction in Italy, these products are exempted from the fiscal regimes of tobacco products.[129] Heat-not-burn tobacco products enjoy the same tax reduction as e-cigarettes, which is half that of traditional cigarettes.[129] Moreover, the enforcement of various tobacco control regulations is only minimally adopted for heated tobacco products in Italy.[129] First of all, health warnings are required to cover only 30% of the heat-not-burn tobacco product packaging (instead of 65% for traditional cigarettes), without pictorial images.[129] Second, comprehensive smoke-free regulations prohibiting smoking in all public places and workplaces do not apply to heat-not-burn tobacco products.[129] Finally, advertising and promotions are not banned for these new products.[129] This is evident by the presence in several strategic Italian cities of the "IQOS embassy" and "IQOS boutique", which are fancy concept stores where IQOS is promoted as a status symbol and people can try it for free.[129] Therefore, the most recognized tobacco control policies (ie, price/tax increase, smoking bans, advertising bans, and health warnings) have been compromised for heat-not-burn tobacco products in Italy.[129]
Heat-not-burn tobacco products are not restricted for sale in Israel by the Ministry of Health.[130] Justice Ministry in Israel agreed with the view of three voluntary organizations that the IQOS is a tobacco product, and the product should be regulated in the same manner as tobacco products.[131] In Israel IQOS are now taxed at the same rate as traditional cigarettes.[132]
Ploom, IQOS, and glo fall under the Tobacco Business Act as tobacco products in Japan because they consist of tobacco leaf.[133] Ploom and IQOS are governed by the Tobacco Industries Act regulations as tobacco products in Japan.[134] The Liberal Democratic Party will deliberate over increasing the tax rate for heat-not-burn tobacco products in April 2018.[135]
Electronic tobacco products using dry material are regulated as e-cigarettes in South Korea by the Ministry of Health and Welfare.[136] Korea regulates e-cigarettes differently than traditional cigarettes for tax reasons.[137] As a result, IQOS are taxed at a decreased rate, compared to the 75% incurred on normal cigarettes.[137] Emerging tobacco products are banned in Singapore by the Ministry of Health.[138]
After IQOS launched a marketing campaign in New Zealand in December 2016, the Ministry of Health stated in 2017 that the refill sticks are not legal for sale in New Zealand under the Smoke-free Environments Act 1990.[139] A representative for the company in New Zealand stated that IQOS product complies with the Smoke-Free Environments Act.[140] Three meetings between Ministry of Health officials and people from the tobacco industry were held from May 30, 2017 through June 2, 2017 to "discuss regulation of new tobacco and nicotine-delivery products".[141] Later on, in August 2017, the government stated they would initiate a review process before products are sold for heat-not-burn tobacco products such as IQOS.[141] In 2018, Philip Morris International and the Ministry of Health were in a court over the legality of selling IQOS in New Zealand.[142] A New Zealand court decided in March 2018 that the HEETs sticks used in the IQOS product are legal to sell in New Zealand.[143] Individuals can import heat-not-burn tobacco products to New Zealand for personal use.[144] As of 2016, 19 countries have permitted the sale of IQOS.[82]
Notes
Bibliography
- McNeill, A; Brose, LS; Calder, R; Bauld, L; Robson, D (February 2018). "Evidence review of e-cigarettes and heated tobacco products 2018" (PDF). UK: Public Health England. pp. 1–243.
- "Regulatory Impact Statement: Regulation of smokeless tobacco and nicotine-delivery products" (PDF). Ministry of Health (New Zealand). 2017. pp. 1–52.
- "Further development of the partial guidelines for implementation of Articles 9 and 10 of the WHO FCTC" (PDF). World Health Organization. 12 July 2016. pp. 1–11.
References
- ^ a b c d e f g h i j "Heated tobacco products (HTPs) information sheet". World Health Organization. 2018.
In order to produce the nicotine-infused vapor, HTPs heat tobacco up to 350°C (lower than 600°C as in conventional cigarettes) using battery-powered heating-systems... Currently, there is also insufficient evidence on the potential effects of second-hand emissions produced by HTPs.
Cite error: The named reference "WHO2018" was defined multiple times with different content (see the help page). - ^ Cite error: The named reference
charwas invoked but never defined (see the help page). - ^ a b c Caputi, TL (24 August 2016). "Heat-not-burn tobacco products are about to reach their boiling point". Tobacco Control. 26 (5): 609–610. doi:10.1136/tobaccocontrol-2016-053264. PMID 27558827.
- ^ a b Cite error: The named reference
constituentswas invoked but never defined (see the help page). - ^ a b Bentley, Guy (15 March 2017). "Heat-Not-Burn Tobacco: The Next Wave Of A Harm-Reduction Revolution". Forbes. Cite error: The named reference "Bentley2017" was defined multiple times with different content (see the help page).
- ^ a b "Further development of the partial guidelines for implementation of Articles 9 and 10 of the WHO FCTC" (PDF). World Health Organization. 12 July 2016. pp. 5–6.
As previously discussed, products that heat rather than burn are claimed to be less harmful than traditional cigarettes, although these claims of risk reduction are based on industry-funded studies. Independent studies should be conducted to investigate these claims. Convincing evidence has yet to be provided for the claims of risk reduction and health benefits of products that heat rather than burn tobacco [37]. Some scientists consider these heated tobacco products to be just as harmful as conventional cigarettes[38].
- ^ a b c Lopez, Alexa A.; Hiler, Marzena; Maloney, Sarah; Eissenberg, Thomas; Breland, Alison B. (2016). "Expanding clinical laboratory tobacco product evaluation methods to loose-leaf tobacco vaporizers". Drug and Alcohol Dependence. 169: 33–40. doi:10.1016/j.drugalcdep.2016.10.005. ISSN 0376-8716. PMC 5140724. PMID 27768968.
- ^ a b Chin, Neo Chai (2 March 2017). "Heated tobacco products just as bad as cigarettes: Amy Khor". Today (Singapore newspaper). Mediacorp.
- ^ "Alternatieve tabaksproducten: harm reduction?" (PDF). Netherlands National Institute for Public Health and the Environment. 2016. pp. 1–66.
- ^ Lindson-Hawley, Nicola; Hartmann-Boyce, Jamie; Fanshawe, Thomas R.; Begh, Rachna; Farley, Amanda; Lancaster, Tim (2016). "Interventions to reduce harm from continued tobacco use". Cochrane Database of Systematic Reviews. 10: CD005231. doi:10.1002/14651858.CD005231.pub3. PMID 27734465.
- ^ a b McKelvey, Karma; Popova, Lucy; Kim, Minji; Lempert, Lauren Kass; Chaffee, Benjamin W.; Vijayaraghavan, Maya; Ling, Pamela; Halpern-Felsher, Bonnie (2018). "IQOS labelling will mislead consumers". Tobacco Control. 27 (Suppl 1): s48–s54. doi:10.1136/tobaccocontrol-2018-054333. PMID 30158208.
- ^ Moazed, Farzad; Chun, Lauren; Matthay, Michael A.; Calfee, Carolyn S.; Gotts, Jeffrey (2018). "Assessment of industry data on pulmonary and immunosuppressive effects of IQOS". Tobacco Control. 27 (Suppl 1): s20–s25. doi:10.1136/tobaccocontrol-2018-054296. PMID 30158203.
- ^ a b c "ASH reaction to new Philip Morris IQOS 'heat not burn' product". Action on Smoking and Health. 30 November 2016.
- ^ Conquero, Belén V. (20 January 2017). "¿Es posible conseguir un tabaco menos nocivo?" (in European Spanish). Larazon.es.
- ^ Pieper, Elke; Mallock, Nadja; Henkler-Stephani, Frank; Luch, Andreas (2018). "Tabakerhitzer als neues Produkt der Tabakindustrie: Gesundheitliche Risiken". Bundesgesundheitsblatt - Gesundheitsforschung - Gesundheitsschutz. 61 (11): 1422–1428. doi:10.1007/s00103-018-2823-y. PMID 30284624.
- ^ a b Liu, Xiaoqiu; Lugo, Alessandra; Spizzichino, Lorenzo; Tabuchi, Takahiro; Gorini, Giuseppe; Gallus, Silvano (2018). "Heat-Not-Burn Tobacco Products Are Getting Hot in Italy". Journal of Epidemiology. 28 (5): 274–275. doi:10.2188/jea.JE20180040. PMC 5911679. PMID 29657258.
These trends may be of concern, since we have previously shown that nearly half of Italian IQOS users (45%) and over half of the people interested in IQOS (51%) are never smokers. Therefore, such a product may represent, at least in Italy, a gateway for nicotine addiction among never smokers rather than a harm reduction substitution for current smokers
- ^ Liu, Xiaoqiu; Lugo, Alessandra; Spizzichino, Lorenzo; Tabuchi, Takahiro; Pacifici, Roberta; Gallus, Silvano (26 January 2018). "Heat-not-burn tobacco products: concerns from the Italian experience". Tobacco Control: tobaccocontrol-2017-054054. doi:10.1136/tobaccocontrol-2017-054054.
- ^ a b Li, Gerard; Saad, Sonia; Oliver, Brian G.; Chen, Hui (1 August 2018). "Heat or Burn? Impacts of Intrauterine Tobacco Smoke and E-Cigarette Vapor Exposure on the Offspring's Health Outcome". Toxics. 6 (3): 43. doi:10.3390/toxics6030043. ISSN 2305-6304. PMC 6160993. PMID 30071638.
There is insufficient evidence on the efficacy of heat-not-burn tobacco on smoking cessation. Neither is there information on the potential impact of maternal inhalation of heat-not-burn tobacco smoke during pregnancy on fetal outcomes, all of which require urgent attention.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Bruin, Jennifer E.; Gerstein, Hertzel C.; Holloway, Alison C. (2 April 2010). "Long-Term Consequences of Fetal and Neonatal Nicotine Exposure: A Critical Review". Toxicological Sciences. 116 (2): 364–374. doi:10.1093/toxsci/kfq103. ISSN 1096-6080. PMC 2905398. PMID 20363831.
Overall, the evidence provided in this review overwhelmingly indicates that nicotine should no longer be considered the safe component of cigarette smoke. In fact, many of the adverse postnatal health outcomes associated with maternal smoking during pregnancy may be attributable, at least in part, to nicotine alone.
- ^ England, Lucinda J.; Kim, Shin Y.; Tomar, Scott L.; Ray, Cecily S.; Gupta, Prakash C.; Eissenberg, Thomas; Cnattingius, Sven; Bernert, John T.; Tita, Alan Thevenet N.; Winn, Deborah M.; Djordjevic, Mirjana V.; Lambe, Mats; Stamilio, David; Chipato, Tsungai; Tolosa, Jorge E. (31 December 2010). "Non-cigarette tobacco use among women and adverse pregnancy outcomes". Acta Obstetricia et Gynecologica Scandinavica. 89 (4): 454–464. doi:10.3109/00016341003605719. ISSN 1600-0412. PMC 5881107. PMID 20225987.
The use of any products containing nicotine likely will have adverse effects of fetal neurological development.
- ^ Schraufnagel DE, Blasi F, Drummond MB, Lam DC, Latif E, Rosen MJ, Sansores R, Van Zyl-Smit R (September 2014). "Electronic cigarettes. A position statement of the forum of international respiratory societies" (PDF). American Journal of Respiratory and Critical Care Medicine. 190 (6): 611–8. doi:10.1164/rccm.201407-1198PP. PMID 25006874.
- ^ a b c d e f g h i j k l m n o p q r s Cite error: The named reference
PieperMallock2018was invoked but never defined (see the help page). - ^ Forster, Mark; Liu, Chuan; Duke, Martin G; McAdam, Kevin G; Proctor, Christopher J (2015). "An experimental method to study emissions from heated tobacco between 100-200°C". Chemistry Central Journal. 9 (1). doi:10.1186/s13065-015-0096-1. ISSN 1752-153X. PMC 4418098. PMID 25941536.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ a b c d e f g h i j St.Helen, Gideon; Jacob III, Peyton; Nardone, Natalie; Benowitz, Neal L (2018). "IQOS: examination of Philip Morris International's claim of reduced exposure". Tobacco Control. 27 (Suppl 1): s30–s36. doi:10.1136/tobaccocontrol-2018-054321. ISSN 0964-4563. PMID 30158205.
 This article incorporates text by Gideon St.Helen, Peyton Jacob III, Natalie Nardone, and Neal L Benowitz available under the CC BY 4.0 license.
This article incorporates text by Gideon St.Helen, Peyton Jacob III, Natalie Nardone, and Neal L Benowitz available under the CC BY 4.0 license.
- ^ Davis, Barbara; Williams, Monique; Talbot, Prue (20 February 2018). "iQOS: evidence of pyrolysis and release of a toxicant from plastic". Tobacco Control: tobaccocontrol–2017–054104. doi:10.1136/tobaccocontrol-2017-054104 (inactive 25 October 2018). ISSN 0964-4563. PMID 29535257.
{{cite journal}}: CS1 maint: DOI inactive as of October 2018 (link) - ^ Katz, Mitchell H. (July 2017). "No Smoke—Just Cancer-Causing Chemicals". JAMA Internal Medicine. 177 (7): 1052. doi:10.1001/jamainternmed.2017.1425. ISSN 2168-6106. PMID 28531245.
- ^ a b c Caputi, TL (24 August 2016). "Heat-not-burn tobacco products are about to reach their boiling point". Tobacco Control. 26 (5): 609–610. doi:10.1136/tobaccocontrol-2016-053264. PMID 27558827.
- ^ a b c Cite error: The named reference
LiSaad2018was invoked but never defined (see the help page). - ^ a b c McNeill 2018, p. 201.
- ^ MHNZ 2017, p. 4.
- ^ a b c d e f g h i j k l m n o Bialous, Stella A; Glantz, Stanton A (2018). "Heated tobacco products: another tobacco industry global strategy to slow progress in tobacco control". Tobacco Control. 27 (Suppl 1): s111–s117. doi:10.1136/tobaccocontrol-2018-054340. ISSN 0964-4563. PMC 6202178. PMID 30209207.
 This article incorporates text by Stella A Bialous and Stanton A Glantz available under the CC BY 4.0 license.
This article incorporates text by Stella A Bialous and Stanton A Glantz available under the CC BY 4.0 license.
- ^ Mcgill, Douglas C (19 November 1988). "'Smokeless' Cigarette's Hapless Start". The New York Times. ISSN 0362-4331.
- ^ Haig, Matt (2003). Brand Failures: The Truth about the 100 Biggest Branding Mistakes of All Time. Kogan Page Publishers. ISBN 978-0-7494-4433-4.
- ^ Parker-Pope, Tara (10 February 2001). ""Safer" Cigarettes: A History". PBS.
- ^ Hilts, Philip J. (27 November 1994). "Little Smoke, Little Tar, but Full Dose of Nicotine". The New York Times. ISSN 0362-4331.
- ^ a b O'Connell, Dominic (30 November 2016). "Philip Morris could stop making conventional cigarettes". BBC News.
- ^ a b Anderson, S J; Ling, P M (2008). ""And they told two friends...and so on": RJ Reynolds' viral marketing of Eclipse and its potential to mislead the public". Tobacco Control. 17 (4): 222–229. doi:10.1136/tc.2007.024273. ISSN 0964-4563. PMC 2845302. PMID 18332064.
- ^ Haig, Matt (2005). Brand Failures: The Truth about the 100 Biggest Branding Mistakes of All Time. Kogan Page Publishers. pp. 51–. ISBN 978-0-7494-4433-4.
- ^ Fisher, Daniel (29 May 2014). "Is This The Cigarette Of The Future, And Will The FDA Let You Buy It?". Forbes.
- ^ "New heat-not-burn brand from RAI". Tobacco Journal International. 5 January 2015.
- ^ a b Craver, Richard (28 July 2015). "Reynolds ends Revo test market in Wisconsin". Winston-Salem Journal.
- ^ a b c d e f g h i Harlay, Jérôme (9 November 2016). "What you need to know about Heat-not-Burn (HNB) cigarettes". VapingPost.
- ^ a b Rossel, Stefanie (1 June 2016). "All eyes on iQOS". Tobacco Reporter.
- ^ a b c d e f g h i j Elias, Jesse; Dutra, Lauren M; St. Helen, Gideon; Ling, Pamela M (2018). "Revolution or redux? Assessing IQOS through a precursor product". Tobacco Control. 27 (Suppl 1): s102–s110. doi:10.1136/tobaccocontrol-2018-054327. ISSN 0964-4563. PMID 30305324.
- ^ Pollack, Juddan (27 October 1997). "Philip Morris tries smokeless Accord: tobacco marketer, cautious about brand, doing 'consumer research'". Ad Age.
- ^ "Anti-smoking body attacks smokeless cigarette device". Tobacco Journal International. 11 December 2007.
- ^ Houston, Cameron (27 June 2007). "Revealed: tobacco giant's secret new weapon in the age of smoking bans". The Age.
- ^ Cooper, Ted (1 February 2014). "Why Philip Morris International's New Heated Products Will Do Better Than Its Last Attempt". The Motley Fool.
- ^ Lubin, Gus (25 June 2012). "Philip Morris Is Releasing A Bunch Of Crazy New Cigarettes". Business Insider.
- ^ MacGuill, Shane (23 January 2014). "Has Philip Morris Learned from Heat-not-Burn Tobacco's Past?". Euromonitor International.
- ^ Davies, Rob; Monaghan, Angela (30 November 2016). "Philip Morris's vision of cigarette-free future met with scepticism". The Guardian.
- ^ McNeill 2018, p. 208.
- ^ a b McNeill 2018, p. 23.
- ^ McNeill 2018, p. 219.
- ^ a b Tuinstra, Taco (16 December 2014). "Certified organic e-liquids being launched by VTM". Tobacco Reporter.
- ^ a b O'Connell, Thomas (9 July 2013). "Method for preparing tobacco extract for electronic smoking devices".
- ^ a b Staal, Yvonne CM; van de Nobelen, Suzanne; Havermans, Anne; Talhout, Reinskje (2018). "New Tobacco and Tobacco-Related Products: Early Detection of Product Development, Marketing Strategies, and Consumer Interest". JMIR Public Health and Surveillance. 4 (2): e55. doi:10.2196/publichealth.7359. ISSN 2369-2960. PMID 29807884.
{{cite journal}}: CS1 maint: unflagged free DOI (link) This article incorporates text by Yvonne CM Staal, Suzanne van de Nobelen, Anne Havermans, and Reinskje Talhout available under the CC BY 4.0 license.
This article incorporates text by Yvonne CM Staal, Suzanne van de Nobelen, Anne Havermans, and Reinskje Talhout available under the CC BY 4.0 license.
- ^ "BAT finds strong Japan demand for its Glo smokeless tobacco device". The Japan Times. Reuters. 22 March 2017.
- ^ a b c Caruana, Diane (25 October 2017). "BAT to launch its HnB device in Russia". VapingPost.
- ^ a b c d e Caplinger, Dan (31 May 2017). "Here's Why the Worst Might Be Yet to Come for Philip Morris International". The Motley Fool. Cite error: The named reference "Caplinger2017" was defined multiple times with different content (see the help page).
- ^ "Innovation Drives BAT's $47 Billion Bid -- WSJ". ADVFN. 24 October 2016.
- ^ a b MHNZ 2017, p. 5.
- ^ a b News Desk (24 October 2016). "World's second largest tobacco company tells people to quit smoking". The Express Tribune.
- ^ Felberbaum, Michael (26 June 2014). "Philip Morris Int'l to Sell Marlboro HeatSticks". Salon (website). Associated Press.
- ^ a b c Gillette, Felix; Kaplan, Jennifer; Chambers, Sam (8 March 2017). "Big Tobacco Has Caught Startup Fever". Bloomberg News.
- ^ Caplinger, Dan (23 November 2015). "5 Things Every Philip Morris Investor Should Know". The Motley Fool.
- ^ Nathan, Ralph (12 October 2016). "Why Philip Morris's iQOS Sales in Japan Are Promising". Market Realist.
- ^ "Philip Morris looks beyond cigarettes with alternative products". Reuters. 30 November 2016.
- ^ a b Kim, Minji (2017). "Philip Morris International introduces new heat-not-burn product, IQOS, in South Korea". Tobacco Control: tobaccocontrol-2017-053965. doi:10.1136/tobaccocontrol-2017-053965. ISSN 0964-4563. PMID 29170165.
- ^ Clarke, Toni (25 January 2018). "U.S. panel deals blow to Philip Morris tobacco device". Reuters.
- ^ Mulier, Thomas; Thesing, Gabi (26 June 2014). "Philip Morris Sees $700 Million Boost From iQOS Smoking Device". Bloomberg News.
- ^ a b Kaplan, Jennifer (19 April 2018). "Philip Morris Plunges the Most in a Decade on Slump in Cigarettes". Bloomberg News.
- ^ Mathers, Annalise; Schwartz, Robert; O'Connor, Shawn; Fung, Michael; Diemert, Lori (2018). "Marketing IQOS in a dark market". Tobacco Control: tobaccocontrol-2017-054216. doi:10.1136/tobaccocontrol-2017-054216. ISSN 0964-4563. PMID 29724866.
- ^ a b c McKelvey, Karma; Popova, Lucy; Kim, Minji; Chaffee, Benjamin W; Vijayaraghavan, Maya; Ling, Pamela; Halpern-Felsher, Bonnie (November 2018). "Heated tobacco products likely appeal to adolescents and young adults". Tobacco Control. 27 (Suppl 1): s41–s47. doi:10.1136/tobaccocontrol-2018-054596. ISSN 0964-4563. PMID 30352843. Cite error: The named reference "McKelveyPopova2018" was defined multiple times with different content (see the help page).
- ^ "Tobacco company charged over importing prohibited product". The New Zealand Herald. 18 May 2017.
- ^ a b Drope, Jeffrey; Cahn, Zachary; Kennedy, Rosemary; Liber, Alex C.; Stoklosa, Michal; Henson, Rosemarie; Douglas, Clifford E.; Drope, Jacqui (2017). "Key issues surrounding the health impacts of electronic nicotine delivery systems (ENDS) and other sources of nicotine". CA: A Cancer Journal for Clinicians. 67 (6): 449–471. doi:10.3322/caac.21413. ISSN 0007-9235. PMID 28961314.
- ^ "Philip Morris's Cigarette Alternative Could Hit U.S. in 2017". Bloomberg News. 5 October 2016.
- ^ a b Hendlin, Yogi Hale; Elias, Jesse; Ling, Pamela M. (2017). "The Pharmaceuticalization of the Tobacco Industry". Annals of Internal Medicine. 167 (4): 278. doi:10.7326/M17-0759. ISSN 0003-4819. PMC 5568794. PMID 28715843.
- ^ "Philip Morris to invest 300 million euros in Greece for smoke-free product". Reuters. 22 March 2017.
- ^ Hyo-sik, Lee (17 May 2017). "Philip Morris unveils smoke-free cigarette in Korea". The Korea Times.
- ^ a b Lasseter, Tom; Wilson, Duff; Wilson, Thomas; Bansal, Paritosh (15 May 2018). "Philip Morris device knows a lot about your smoking habit". Reuters.
- ^ a b c d e f g h i j k l m n o p q r s Auer, Reto; Concha-Lozano, Nicolas; Jacot-Sadowski, Isabelle; Cornuz, Jacques; Berthet, Aurélie (1 July 2017). "Heat-Not-Burn Tobacco Cigarettes: Smoke by Any Other Name". JAMA Internal Medicine. 177 (7): 1050–1052. doi:10.1001/jamainternmed.2017.1419. ISSN 2168-6106. PMC 5543320. PMID 28531246.
- ^ Duprey, Rich (9 December 2017). "Will 2018 Be Philip Morris International Inc's Best Year Yet?". Billings Gazette.
- ^ a b Tai, Mariko (31 August 2015). "Philip Morris rolls out iQOS smokeless smokes". Nikkei Asian Review.
- ^ a b Cite error: The named reference
JenssenWalley2017was invoked but never defined (see the help page). - ^ Conquero, Belén V. (20 January 2017). "¿Es posible conseguir un tabaco menos nocivo?" [Is it possible to get a less noxious tobacco?]. La Razón (in Spanish).
- ^ "Addictive nicotine and harmful substances also present in heated tobacco". Netherlands National Institute for Public Health and the Environment. 15 May 2018.
- ^ a b Katz, MH; Redberg, RF (6 November 2017). "Science Requires Open Discourse". JAMA internal medicine. 178 (1). doi:10.1001/jamainternmed.2017.5763. PMID 29114738.
- ^ Kiefer, Bertrand (21 June 2017). "Tabac : nouveau produit, vieilles methods" [Tobacco: New product, Old methods]. Revue Médicale Suisse (in French): 1312.
- ^ a b c William Wan (14 August 2017). "Big tobacco's new cigarette is sleek, smokeless — but is it actually healthier?". The Washington Post.
- ^ "Questions over risks of Philip Morris's smoke-free tobacco device IQOS". CBC. 24 January 2018.
- ^ a b c d e f "FDA Panel Gives Qualified Support To Claims For". National Public Radio. 25 January 2018.
- ^ a b "Modified Risk Tobacco Product Applications (MRTPAs) MR0000059-MR0000061 Philip Morris Products S.A." (PDF). Food and Drug Administration. 24–25 January 2018. pp. 12–15.
- ^ Lasseter, Tom; Bansal, Paritosh; Wilson, Thomas; Miyazaki, Ami; Wilson, Duff; Kalra, Aditya (20 December 2017). "Scientists describe problems in Philip Morris e-cigarette experiments". Reuters.
- ^ a b Trefis Team (30 December 2016). "FDA Approval For iQOS To Be A Game Changer For Altria". Forbes.
- ^ speccomm (26 May 2017). "FDA starts iQOS review". Tobacco Reporter.
- ^ Chambers, Sam (26 January 2018). "Big Tobacco spending billions to develop products that could move industry beyond cigarettes — but regulators are skeptical". Chicago Tribune. Archived from the original on 21 June 2018.
- ^ "Meeting of the Tobacco Products Scientific Advisory Committee January 24-25, 2018" (PDF). Food and Drug Administration. 24–25 January 2018.
- ^ Kaplan, Sheila; Thomas, Katie (11 February 2018). "F.D.A. Chief Goes Against the Administration Stereotype". The New York Times.
- ^ Yui, Monami (28 August 2016). "Big Tobacco Wants to Turn Japan's Smokers Into Vapers". Bloomberg News.
- ^ Mulier, Thomas; Chambers, Sam; Liefgreen, Liefgreen (24 March 2016). "Marlboro Kicks Some Ash". Bloomberg News.
- ^ Varlet, Vincent; Concha-Lozano, Nicolas; Berthet, Aurélie; Plateel, Grégory; Favrat, Bernard; De Cesare, Mariangela; Lauer, Estelle; Augsburger, Marc; Thomas, Aurélien; Giroud, Christian (2016). "Drug vaping applied to cannabis: Is "Cannavaping" a therapeutic alternative to marijuana?". Scientific Reports. 6 (1). doi:10.1038/srep25599. ISSN 2045-2322. PMC 4881394. PMID 27228348.
- ^ Vu, An T.; Taylor, Kenneth M.; Holman, Matthew R.; Ding, Yan S.; Hearn, Bryan; Watson, Clifford H. (2015). "Polycyclic Aromatic Hydrocarbons in the Mainstream Smoke of Popular U.S. Cigarettes". Chemical Research in Toxicology. 28 (8): 1616–1626. doi:10.1021/acs.chemrestox.5b00190. ISSN 0893-228X. PMC 4540633. PMID 26158771.
- ^ "The iSmoke OneHitter, iSmoke "3-in-1" and iSmoke Oven join portfolio". Convenience Store News. 2017.
- ^ a b "Consumer-Centric Vaping". Convenience Store Decisions. Harbor Communications. 4 March 2015.
- ^ a b c d e "The comparison Of The Heat Not Burn Products IUOC and IQOS". AB Newswire. 6 September 2017.
- ^ "KT&G to release heat-not-burn tobacco amid iQOS growing popularity". The Korea Herald. 8 June 2017.
- ^ a b "KT&G launches sales of new tobacco-heating device". Yonhap News Agency. 11 November 2017.
- ^ Yakowicz, Will (10 June 2015). "This Silicon Valley Company Just Raised $47 Million to Smoke Cigarette Makers". Inc.
- ^ Dawson, Freddie (31 July 2015). "Pax Labs Looking At International Expansion". Forbes.
- ^ Biggs, John (17 June 2012). "Smoke Up: An Interview With The Creator Of The Ultracool Pax Vaporizer". TechCrunch.
- ^ Lavrinc, Damon (1 July 2013). "Review: Ploom Model Two". Wired.
- ^ Lawler, Ryan (11 March 2015). "The Pax 2 Improves Upon One Of The Best Vaporizers On The Market". TechCrunch.
- ^ a b Stenovec, Tim (12 March 2016). "How two guys from Stanford built the 'iPhone of vaporizers'". Business Insider.
- ^ Rossel, Stefanie (1 July 2016). "Blending nature and technology". Tobacco Reporter.
- ^ Tuinstra, Taco (26 January 2016). "JT announces launch of next-generation Ploom". Tobacco Reporter.
- ^ "JT to expand Ploom TECH sales". Tobacco Reporter. 10 October 2016.
- ^ Uranaka, Taiga; Sarkar, Himani (29 May 2017). "Japan Tobacco plans to quadruple smokeless tobacco output capacity by 2018: CEO". Channel NewsAsia. Reuters.
- ^ a b c Monkeyshines (1 April 2015). "High Buy: V2 Pro Series 7". High Times.
- ^ a b Kahn, Steven (11 March 2016). "V2 Pro Series 3 Vaporizer Review – Updated for 2017". Bloomberg News.
- ^ a b Silver, Curtis (19 May 2017). "V2 Pro Series 3X Is The Highly Versatile Vape You've Been Looking For". Forbes.
- ^ Craft, Scott (16 June 2015). "V2 Pro Series 7 Review: Premium Vapor Quality From A Surprisingly Affordable Vaporizer". iDigitalTimes.
- ^ McNeill 2018, p. 22, 218.
- ^ Unger, Michael; Unger, Darian W. (2018). "E-cigarettes/electronic nicotine delivery systems: a word of caution on health and new product development". Journal of Thoracic Disease. 10 (S22): S2588–S2592. doi:10.21037/jtd.2018.07.99. ISSN 2072-1439. PMC 6178300. PMID 30345095.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Cite error: The named reference
KotzKastaun2018was invoked but never defined (see the help page). - ^ a b Rapaport, Lisa (26 May 2017). "'Heat-not-burn' cigarettes still release cancer-causing chemicals". Reuters.
- ^ Liber, Alex C. (2018). "Heated tobacco products and combusted cigarettes: comparing global prices and taxes". Tobacco Control: tobaccocontrol-2018-054602. doi:10.1136/tobaccocontrol-2018-054602. ISSN 0964-4563. PMID 30381439.
- ^ Fisher, Daniel (16 June 2014). "Philip Morris International Bets Big On The Future Of Smoking". Forbes.
- ^ a b c d e f g h Liu, Xiaoqiu; Lugo, Alessandra; Spizzichino, Lorenzo; Tabuchi, Takahiro; Gorini, Giuseppe; Gallus, Silvano (2018). "Heat-Not-Burn Tobacco Products Are Getting Hot in Italy". Journal of Epidemiology. 28 (5): 274–275. doi:10.2188/jea.JE20180040. ISSN 0917-5040. PMC 5911679. PMID 29657258.
 This article incorporates text by Xiaoqiu Liu, Alessandra Lugo, Lorenzo Spizzichino, Takahiro Tabuchi, Giuseppe Gorini, and Silvano Gallus available under the CC BY 4.0 license.
This article incorporates text by Xiaoqiu Liu, Alessandra Lugo, Lorenzo Spizzichino, Takahiro Tabuchi, Giuseppe Gorini, and Silvano Gallus available under the CC BY 4.0 license.
- ^ Siegel-Itzkovich, Judy (2 March 2017). "Orgs slam Litzman for allowing sale of iQOS heated, smokeless cigarettes". The Jerusalem Post.
- ^ Ronny Linder-Ganz (16 January 2018). "In Blow to Philip Morris, Israel to Tax iQOS E-cigarettes Like Ordinary Cigarettes". Haaretz.
- ^ Judy Siegel-Itzkovich (3 April 2018). "Justice Ministry says iQOS product will be treated as ordinary tobacco". The Jerusalem Post.
- ^ Tabuchi, Takahiro; Gallus, Silvano; Shinozaki, Tomohiro; Nakaya, Tomoki; Kunugita, Naoki; Colwell, Brian (2018). "Heat-not-burn tobacco product use in Japan: its prevalence, predictors and perceived symptoms from exposure to secondhand heat-not-burn tobacco aerosol". Tobacco Control. 27 (e1): e25–e33. doi:10.1136/tobaccocontrol-2017-053947. ISSN 0964-4563. PMC 6073918. PMID 29248896.
- ^ Tabuchi, Takahiro; Kiyohara, Kosuke; Hoshino, Takahiro; Bekki, Kanae; Inaba, Yohei; Kunugita, Naoki (2016). "Awareness and use of electronic cigarettes and heat-not-burn tobacco products in Japan". Addiction. 111 (4): 706–713. doi:10.1111/add.13231. ISSN 0965-2140. PMID 26566956.
- ^ "Tax hike on heat-not-burn tobacco products under consideration as LDP begins review of tax reforms". The Japan Times. 8 September 2017.
- ^ Jae-hyuk, Park (26 May 2017). "IQOS available in Seoul Saturday". The Korea Times.
- ^ a b Trefis Team (13 September 2017). "Why Is Korea Easier To Conquer For iQOS Than Europe?". NASDAQ.
- ^ "Singapore Enhances Tobacco Control Measures". Ministry of Health (Singapore). 28 July 2016.
- ^ Elder, Vaughan (13 January 2017). "Legality of tobacco product in question". Otago Daily Times.
- ^ Caruana, Diane (3 February 2017). "iQos heatsticks declared illegal in NZ". VapingPost.
- ^ a b "New Zealand's legal action against IQOS postponed, consultation with Big Tobacco follows". 130 (1465). New Zealand Medical Journal. 10 November 2017.
{{cite journal}}: Cite journal requires|journal=(help) - ^ "iQOS may not be as harm-free as claimed, study finds". The New Zealand Herald. 14 March 2018.
- ^ Zaharia, Marius (27 March 2018). "New Zealand court gives Philip Morris nod to sell heated tobacco product". Reuters.
- ^ MHNZ 2017, p. ii.