89.173.17.50 (talk) No edit summary |
KMnO4 and OsO4 might be fine for academia, but not in commodity world |
||
| Line 1: | Line 1: | ||
[[Image:Ethylene glycol.svg|thumb|right|[[Ethylene glycol]], a simple diol]] |
[[Image:Ethylene glycol.svg|thumb|right|[[Ethylene glycol]], a simple diol]] |
||
[[File:Resorcinol.svg|thumb|right|[[Resorcinol]]]] |
[[File:Resorcinol.svg|thumb|right|[[Resorcinol]]]] |
||
A '''diol''' is a [[chemical compound]] containing two [[hydroxyl group]]s (—OH groups).<ref>{{JerryMarch}}</ref> This pairing of functional groups is pervasive and many subcategories have been identified. The most common industrial diol is [[ethylene glycol]]. Examples of diols in which the hydroxyl functional groups are more widely separated include 1,4-butanediol HO—(CH<sub>2</sub>)<sub>4</sub>—OH and [[bisphenol A]], and [[1,3-Propanediol|propylene-1,3-diol]] or beta propylene glycol, HO-CH<sub>2</sub>-CH<sub>2</sub>-CH<sub>2</sub>-OH. |
A '''diol''' is a [[chemical compound]] containing two [[hydroxyl group]]s (—OH groups).<ref>{{JerryMarch}}</ref> This pairing of functional groups is pervasive and many subcategories have been identified. The most common industrial diol is [[ethylene glycol]]. Examples of diols in which the hydroxyl functional groups are more widely separated include 1,4-butanediol HO—(CH<sub>2</sub>)<sub>4</sub>—OH and [[bisphenol A]], and [[1,3-Propanediol|propylene-1,3-diol]] or beta propylene glycol, HO-CH<sub>2</sub>-CH<sub>2</sub>-CH<sub>2</sub>-OH. |
||
== Classification == |
== Classification == |
||
===Geminal diols=== |
===Geminal diols=== |
||
A [[geminal diol]] has two hydroxyl groups bonded to the same atom. Examples include [[methanediol]] H<sub>2</sub>C(OH)<sub>2</sub>, the hydrated form of [[formaldehyde]]. Another example is (F<sub>3</sub>C)<sub>2</sub>C(OH)<sub>2</sub>, the hydrated form of [[hexafluoroacetone]]. |
A [[geminal diol]] has two hydroxyl groups bonded to the same atom. Examples include [[methanediol]] H<sub>2</sub>C(OH)<sub>2</sub>, the hydrated form of [[formaldehyde]]. Another example is (F<sub>3</sub>C)<sub>2</sub>C(OH)<sub>2</sub>, the hydrated form of [[hexafluoroacetone]]. |
||
| Line 11: | Line 10: | ||
In a vicinal diol, the two hydroxyl groups occupy [[vicinal (chemistry)|vicinal]] positions, that is, they are attached to adjacent atoms. These compounds are called glycols. Examples include 1,2-ethanediol or ethylene glycol HO—(CH<sub>2</sub>)<sub>2</sub>—OH, a common ingredient of [[antifreeze]] products. Another example is propane-1,2-diol or alpha propylene glycol, HO—CH<sub>2</sub>—CH(OH)—CH<sub>3</sub> used in the food and medicine industry as well as a relatively non-poisonous antifreeze product. |
In a vicinal diol, the two hydroxyl groups occupy [[vicinal (chemistry)|vicinal]] positions, that is, they are attached to adjacent atoms. These compounds are called glycols. Examples include 1,2-ethanediol or ethylene glycol HO—(CH<sub>2</sub>)<sub>2</sub>—OH, a common ingredient of [[antifreeze]] products. Another example is propane-1,2-diol or alpha propylene glycol, HO—CH<sub>2</sub>—CH(OH)—CH<sub>3</sub> used in the food and medicine industry as well as a relatively non-poisonous antifreeze product. |
||
===Other economically important diols=== |
|||
===Bis(phenol)s=== |
|||
[[Bisphenol A]] is an important compound that contains two phenol groups. |
[[Bisphenol A]] is an important compound that contains two phenol groups. It is a building block in the production of [[polycarbonate]] plastics. [[1,4-Butanediiol]] and [[1,5-Pentanediol|1,5-pentanediol]] are important precursors to [[polyurethane]]s. |
||
===Examples of aliphatic diols=== |
|||
{| class="wikitable" |
|||
|- |
|||
! Linearity of the diol !! Hydroxyls on adjacent carbons (vicinal diols) !! Hydroxyls on non-adjacent carbons |
|||
|- |
|||
| Linear || [[Ethylene glycol]] || [[1,3-Propanediol]], [[1,4-Butanediol]], [[1,5-Pentanediol]], [[1,8-octanediol]], |
|||
|- |
|||
| Branched || [[Propylene glycol|1,2-Propanediol]], [[1,2-Butanediol]], [[2,3-Butanediol]] || [[1,3-Butanediol]], [[1,2-pentanediol]], [[Etohexadiol]], [[p-Menthane-3,8-diol]], [[2-Methyl-2,4-pentanediol]] |
|||
|} |
|||
==Synthesis== |
==Synthesis== |
||
Because diols are a common [[functional group]] arrangement, numerous methods of preparation have been developed. |
Because diols are a common [[functional group]] arrangement, numerous methods of preparation have been developed.<ref name=Ull>Peter Werle, Marcus Morawietz, Stefan Lundmark, Kent Sörensen, Esko Karvinen and Juha Lehtonen "Alcohols, Polyhydric" Ullmann's Encyclopedia of Industrial Chemistry, 2008, Wiley-VCH, Weinheim. {{DOI|10.1002/14356007.a01_305.pub2}}</ref> |
||
===Vicinal diols=== |
|||
* Vicinal diols can be produced from the [[oxidation]] of [[alkene]]s, usually with dilute [[acidic]] [[potassium permanganate]], also known as potassium manganate(VII). Using alkaline potassium manganate(VII) produces a colour change from clear deep purple to clear green; acidic potassium manganate(VII) turns clear colourless. |
|||
| ⚫ | For commodity chemicals, the main route to vicinal diols is the hydrolysis of epoxides. The epoxides are prepared by oxidation with [[hydrogen peroxide]] of the alkene. An example in the synthesis of ''trans-cyclohexanediol''.<ref>trans-cyclohexanediol [[Organic Syntheses]], Coll. Vol. 3, p.217 (1955); Vol. 28, p.35 ('''1948''') http://www.orgsynth.org/orgsyn/pdfs/CV3P0217.pdf.</ref> or by [[microreactor]]:<ref>''Advantages of Synthesizing trans-1,2-Cyclohexanediol in a Continuous Flow Microreactor over a Standard Glass Apparatus'' Andreas Hartung, Mark A. Keane, and Arno Kraft [[J. Org. Chem.]] '''2007''', 72, 10235-10238 {{DOI|10.1021/jo701758p}}</ref> |
||
* [[Osmium tetroxide]] can similarly be used to oxidize alkenes to vicinal diols. |
|||
| ⚫ | |||
:[[Image:CyclohexanediolSynthesis.svg|400px|cyclohexanediol synthesis]] |
:[[Image:CyclohexanediolSynthesis.svg|400px|cyclohexanediol synthesis]] |
||
* A chemical reaction called [[Sharpless asymmetric dihydroxylation]] can be used to produce [[chirality (chemistry)|chiral]] diols from alkenes using an osmate [[reagent]] and a chiral [[catalyst]]. |
|||
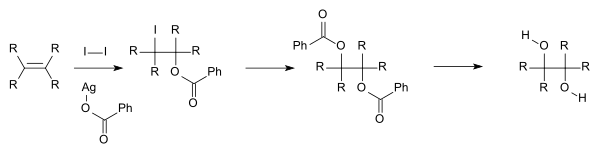
For academic research and pharmaceutical areas, vicinal diols are often produced from the [[oxidation]] of [[alkene]]s, usually with dilute [[acidic]] [[potassium permanganate]]. Using alkaline potassium manganate(VII) produces a colour change from clear deep purple to clear green; acidic potassium manganate(VII) turns clear colourless. [[Osmium tetroxide]] can similarly be used to oxidize alkenes to vicinal diols. The chemical reaction called [[Sharpless asymmetric dihydroxylation]] can be used to produce [[chirality (chemistry)|chiral]] diols from alkenes using an osmate [[reagent]] and a chiral [[catalyst]]. Another method is the [[Woodward cis-hydroxylation]] (cis diol) and the related [[Prévost reaction]] (anti diol), depicted below, which both use iodine and the silver salt of a carboxylic acid. |
|||
:[[Image:PrevostReaction.svg|600px|Prevost reaction]] |
:[[Image:PrevostReaction.svg|600px|Prevost reaction]] |
||
* In the [[Prins reaction]] 1,3-diols can be formed in a reaction between an [[alkene]] and [[formaldehyde]]. |
|||
===1,3-Diols=== |
|||
* Geminal diols can be formed by the hydration of ketones. |
|||
1,3-Diols are prepared industrially by aldol condensation of ketones with [[formaldehyde]]. The resulting carbonyl is reduced using the [[Cannizzaro reaction]] or catalytic hydrogenation: |
|||
| ⚫ | |||
: RC(O)CH<sub>3</sub> + CH<sub>2</sub>O → RC(O)CH<sub>2</sub>CH<sub>2</sub>OH |
|||
: RC(O)CH<sub>2</sub>CH<sub>2</sub>OH + H<sub>2</sub> → RCH(OH)CH<sub>2</sub>CH<sub>2</sub>OH + |
|||
2,2-disubstituted propane-1,3-diols are prepared in this way. Examples include 2-methyl-2-propyl-1,3-propanediol and [[neopentyl glycol]]. |
|||
1,3-Diols can be prepared by hydration of α,β-unsaturated ketones and aldehydes. The resulting keto-alcohol is [[hydrogenated. Another route involves the [[hydroformylation]] of epoxides followed by hydrogenation of the aldehyde. This method has been used for 1,3-propanediol from [[ethylene oxide]]. |
|||
| ⚫ | More specialized routes to 1,3-diols involves the reaction between an [[alkene]] and [[formaldehyde]], the [[Prins reaction]]. 1,3-diols can be produced [[diastereoselectivity|diastereoselectively]] from the corresponding β-hydroxy [[ketone]]s using the [[Evans–Saksena reduction|Evans–Saksena]], [[Narasaka–Prasad reduction|Narasaka–Prasad]] or [[Evans–Tishchenko reaction|Evans–Tishchenko]] reduction protocols. |
||
===1,4-, 1,5-, and longer diols=== |
|||
Diols where the alcohol groups are separated by several carbon centers are generally prepared by hydrogenation of diesters. |
|||
==Reactions== |
==Reactions== |
||
From the industrial perspective, the dominant reactions of the diols is in the production of [[polyurethane]]s and [[alkyd resin]]s.<ref name=Ull/> |
|||
===General diols=== |
===General diols=== |
||
Revision as of 14:49, 7 November 2013


A diol is a chemical compound containing two hydroxyl groups (—OH groups).[1] This pairing of functional groups is pervasive and many subcategories have been identified. The most common industrial diol is ethylene glycol. Examples of diols in which the hydroxyl functional groups are more widely separated include 1,4-butanediol HO—(CH2)4—OH and bisphenol A, and propylene-1,3-diol or beta propylene glycol, HO-CH2-CH2-CH2-OH.
Classification
Geminal diols
A geminal diol has two hydroxyl groups bonded to the same atom. Examples include methanediol H2C(OH)2, the hydrated form of formaldehyde. Another example is (F3C)2C(OH)2, the hydrated form of hexafluoroacetone.
Vicinal diols
In a vicinal diol, the two hydroxyl groups occupy vicinal positions, that is, they are attached to adjacent atoms. These compounds are called glycols. Examples include 1,2-ethanediol or ethylene glycol HO—(CH2)2—OH, a common ingredient of antifreeze products. Another example is propane-1,2-diol or alpha propylene glycol, HO—CH2—CH(OH)—CH3 used in the food and medicine industry as well as a relatively non-poisonous antifreeze product.
Other economically important diols
Bisphenol A is an important compound that contains two phenol groups. It is a building block in the production of polycarbonate plastics. 1,4-Butanediiol and 1,5-pentanediol are important precursors to polyurethanes.
Synthesis
Because diols are a common functional group arrangement, numerous methods of preparation have been developed.[2]
Vicinal diols
For commodity chemicals, the main route to vicinal diols is the hydrolysis of epoxides. The epoxides are prepared by oxidation with hydrogen peroxide of the alkene. An example in the synthesis of trans-cyclohexanediol.[3] or by microreactor:[4]
For academic research and pharmaceutical areas, vicinal diols are often produced from the oxidation of alkenes, usually with dilute acidic potassium permanganate. Using alkaline potassium manganate(VII) produces a colour change from clear deep purple to clear green; acidic potassium manganate(VII) turns clear colourless. Osmium tetroxide can similarly be used to oxidize alkenes to vicinal diols. The chemical reaction called Sharpless asymmetric dihydroxylation can be used to produce chiral diols from alkenes using an osmate reagent and a chiral catalyst. Another method is the Woodward cis-hydroxylation (cis diol) and the related Prévost reaction (anti diol), depicted below, which both use iodine and the silver salt of a carboxylic acid.
1,3-Diols
1,3-Diols are prepared industrially by aldol condensation of ketones with formaldehyde. The resulting carbonyl is reduced using the Cannizzaro reaction or catalytic hydrogenation:
- RC(O)CH3 + CH2O → RC(O)CH2CH2OH
- RC(O)CH2CH2OH + H2 → RCH(OH)CH2CH2OH +
2,2-disubstituted propane-1,3-diols are prepared in this way. Examples include 2-methyl-2-propyl-1,3-propanediol and neopentyl glycol.
1,3-Diols can be prepared by hydration of α,β-unsaturated ketones and aldehydes. The resulting keto-alcohol is [[hydrogenated. Another route involves the hydroformylation of epoxides followed by hydrogenation of the aldehyde. This method has been used for 1,3-propanediol from ethylene oxide.
More specialized routes to 1,3-diols involves the reaction between an alkene and formaldehyde, the Prins reaction. 1,3-diols can be produced diastereoselectively from the corresponding β-hydroxy ketones using the Evans–Saksena, Narasaka–Prasad or Evans–Tishchenko reduction protocols.
1,4-, 1,5-, and longer diols
Diols where the alcohol groups are separated by several carbon centers are generally prepared by hydrogenation of diesters.
Reactions
From the industrial perspective, the dominant reactions of the diols is in the production of polyurethanes and alkyd resins.[2]
General diols
Diols react as alcohols, by esterification and ether formation.
Diols such as ethylene glycol are used as co-monomers in polymerization reactions forming polymers including some polyesters and polyurethanes. A different monomer with two identical functional groups, such as a dioyl dichloride or dioic acid is required to continue the process of polymerization through repeated esterification processes.
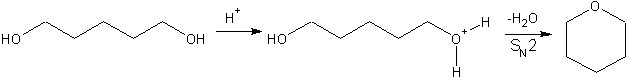
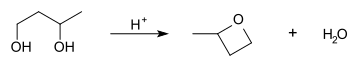
A diol can be converted to cyclic ether by using an acid catalyst, this is diol cyclization. Firstly, it involves protonation of the hydroxyl group. Then, followed by intramolecular nucleophilic substitution, the second hydroxyl group attacks the electron deficient carbon. Provided that there are enough carbon atoms that the angle strain is not too much, a cyclic ether can be formed.
Diols can also be converted to lactones employing the Fétizon oxidation reaction.
Vicinal diols
In glycol cleavage, the C-C bond in a vicinal diol is cleaved with formation of ketone or aldehyde functional groups. See Diol oxidation.
Geminal diols
In general, organic geminal diols readily dehydrate to form a carbonyl group. For example, carbonic acid ((HO)2C=O) is unstable and has a tendency to convert to carbon dioxide (CO2) and water (H2O). Nevertheless, in rare situations the chemical equilibrium is in favor of the geminal diol. For example, when formaldehyde (H2C=O) is dissolved in water the geminal diol (H2C(OH)2), methanediol, is favored. Other examples are the cyclic geminal diols decahydroxycyclopentane (C5(OH)10) and dodecahydroxycyclohexane (C6(OH)12), which are stable, whereas the corresponding oxocarbons (C5O5 and C6O6) do not seem to be.
See also
- Alcohols, chemical compounds with one hydroxyl group
- Triols, chemical compounds with three hydroxyl group
- Polyols, chemical compounds with multiple hydroxyl groups
- Ethylene glycol
- Glycol Nucleic Acid(GNA)
References
- ^ March, Jerry (1985), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, 3rd edition, New York: Wiley, ISBN 9780471854722, OCLC 642506595
- ^ a b Peter Werle, Marcus Morawietz, Stefan Lundmark, Kent Sörensen, Esko Karvinen and Juha Lehtonen "Alcohols, Polyhydric" Ullmann's Encyclopedia of Industrial Chemistry, 2008, Wiley-VCH, Weinheim. doi:10.1002/14356007.a01_305.pub2
- ^ trans-cyclohexanediol Organic Syntheses, Coll. Vol. 3, p.217 (1955); Vol. 28, p.35 (1948) http://www.orgsynth.org/orgsyn/pdfs/CV3P0217.pdf.
- ^ Advantages of Synthesizing trans-1,2-Cyclohexanediol in a Continuous Flow Microreactor over a Standard Glass Apparatus Andreas Hartung, Mark A. Keane, and Arno Kraft J. Org. Chem. 2007, 72, 10235-10238 doi:10.1021/jo701758p