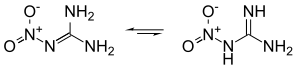131.170.90.3 (talk) No edit summary |
not made from guano or guanidine, nice story but probable myth |
||
| Line 52: | Line 52: | ||
}} |
}} |
||
'''Nitroguanidine''' is |
'''Nitroguanidine''' is an [[organic compound]] with the formula [[(NH<sub>2</sub>)<sub>2</sub>CN(H)ONO<sub>2</sub>. It is a colorless, crystalline solid. It melts at 232 °C and decomposes at 250 °C. It is not flammable and is an low sensitivity explosive; however, its [[detonation velocity]] is high. It is used as a propellant (air bags), fertilizer, and for other purposes. |
||
==Manufacture== |
==Manufacture== |
||
Nitroguanidine is produced commercially by a two step processs starting with the hydrolysis of [[calcium cyanamide]] in the presence of [[ammonium nitrate]]. Via the intermediacy of biguanidine, this step affords the salt guanidinium nitrate. In the second step, the nitrate salt is treated with sulfuric acid, a process that dehydrates the salt and forms the [[nitrate ester]].<ref name=Ullmann>Thomas Güthner, Bernd Mertschenk and Bernd Schulz "Guanidine and Derivatives" in Ullmann's Encyclopedia of Industrial Chemistry, 2006, Wiley-VCH, Weinheim. {{DOI|10.1002/14356007.a12_545.pub2}}</ref> |
|||
Nitroguanidine is manufactured from [[guanine]], a naturally-occurring substance typically found in the excrement of bats and birds ([[guano]]). Guanine is extracted from the droppings and is then oxidized to form [[guanidine]].<ref>{{cite journal| first=A| last=Strecker| title=Untersuchungen über die chemischen Beziehungen zwischen Guanin, Xanthin, Theobromin, Caffein und Kreatinin| journal=Annalen der Chemie und Pharmacie| volume=118| pages= 151–177| year=1861| doi=10.1002/jlac.18611180203| issue=2}}</ref> Guanidine is then nitrated to form nitroguanidine. The efficient process used to perform industrial-scale synthesis is considered proprietary by the manufacturers and is not yet available for public release. A laboratory synthesis involves the fusion of [[sulfamic acid]] with [[urea]], followed by nitration of the formed guanidine sulfate.<ref>{{cite web |
|||
:[[C(NH<sub>2</sub>)<sub>3</sub>]NO<sub>3</sub> → [[(NH<sub>2</sub>)<sub>2</sub>CN(H)ONO<sub>2</sub> + H<sub>2</sub>O |
|||
| url=http://www.sciencemadness.org/talk/viewthread.php?tid=8911 |
|||
| title=Nitroguanidine: from sulphamic acid and urea| author = Axt | work= Sciencemadness Discussion Board |
|||
| date=August 1, 2007}}</ref> |
|||
Nitroguanidine can be generated by treatment of urea with ammonium nitrate. Owing to problems of reliability and safety, this process has not been commerciallized despite its attractive economic features. |
|||
==Uses== |
==Uses== |
||
===Explosives=== |
===Explosives=== |
||
Revision as of 18:25, 7 September 2013
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
1-Nitroguanidine
| |||
| Other names
Picrite
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.008.313 | ||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| CH4N4O2 | |||
| Molar mass | 104.07 g/mol | ||
| Appearance | Colorless crystalline solid | ||
| Melting point | 232 °C (450 °F; 505 K) | ||
| Boiling point | 250 °C (decomp.) | ||
| Explosive data | |||
| RE factor | 1.00 | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Explosive | ||
| Related compounds | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Nitroguanidine is an organic compound with the formula [[(NH2)2CN(H)ONO2. It is a colorless, crystalline solid. It melts at 232 °C and decomposes at 250 °C. It is not flammable and is an low sensitivity explosive; however, its detonation velocity is high. It is used as a propellant (air bags), fertilizer, and for other purposes.
Manufacture
Nitroguanidine is produced commercially by a two step processs starting with the hydrolysis of calcium cyanamide in the presence of ammonium nitrate. Via the intermediacy of biguanidine, this step affords the salt guanidinium nitrate. In the second step, the nitrate salt is treated with sulfuric acid, a process that dehydrates the salt and forms the nitrate ester.[1]
- [[C(NH2)3]NO3 → [[(NH2)2CN(H)ONO2 + H2O
Nitroguanidine can be generated by treatment of urea with ammonium nitrate. Owing to problems of reliability and safety, this process has not been commerciallized despite its attractive economic features.
Uses
Explosives
Nitroguanidine is used as an explosive propellant, notably in triple-base smokeless powder. The nitroguanidine reduces the propellant's flash and flame temperature without sacrificing chamber pressure. These are typically used in large bore guns where barrel erosion and flash are particularly important to avoid.
Pesticides
Nitroguanidine derivatives are used as insecticides, having a comparable effect to nicotine. Derivatives include clothianidin, dinotefuran, imidacloprid, and thiamethoxam.
Biochemistry
The nitrosoylated derivative nitrosoguanidine is often used to mutagenize bacterial cells for biochemical studies.
Structure
Nitroguanidine can exist in distinct tautomeric forms, as a nitroimine (left) or a nitroamine (right). In solution and in the solid state, the nitroimine form predominates (resonance stabilized).[2][3]
References
- ^ Thomas Güthner, Bernd Mertschenk and Bernd Schulz "Guanidine and Derivatives" in Ullmann's Encyclopedia of Industrial Chemistry, 2006, Wiley-VCH, Weinheim. doi:10.1002/14356007.a12_545.pub2
- ^ Bulusu, S.; Dudley, R. L.; Autera, J. R. (1987). "Structure of nitroguanidine: nitroamine or nitroimine? New NMR evidence from nitrogen-15 labeled sample and nitrogen-15 spin coupling constants". Magnetic Resonance in Chemistry. 25 (3): 234–238. doi:10.1002/mrc.1260250311.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Murmann, R. K.; Glaser, Rainer; Barnes, Charles L. (2005). "Structures of nitroso- and nitroguanidine x - ray crystallography and computational analysis". Journal of Chemical Crystallography. 35 (4): 317–325. doi:10.1007/s10870-005-3252-y.
{{cite journal}}: CS1 maint: multiple names: authors list (link)