m WP:CHECKWIKI error fix for #61. Punctuation goes before References. Do general fixes if a problem exists. - using AWB (9421) |
aminopolycarboxylic acid links |
||
| Line 85: | Line 85: | ||
}} |
}} |
||
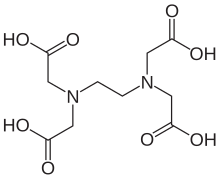
'''Ethylenediaminetetraacetic acid''', widely abbreviated as '''EDTA''' (for other names, see Table), is |
'''Ethylenediaminetetraacetic acid''', widely abbreviated as '''EDTA''' (for other names, see Table), is an [[aminopolycarboxylic acid]] and a colourless, water-soluble solid. Its [[conjugate base]] is named '''ethylenediaminetetraacetate'''. It is widely used to dissolve [[limescale]]. Its usefulness arises because of its role as a hexadentate ("six-toothed") [[ligand]] and [[chelating agent]], i.e. its ability to "sequester" [[metal]] [[ion]]s such as Ca<sup>2+</sup> and Fe<sup>3+</sup>. After being bound by EDTA, metal ions remain in solution but exhibit diminished reactivity. EDTA is produced as several salts, notably disodium EDTA and calcium disodium EDTA. |
||
==Synthesis== |
==Synthesis== |
||
| Line 98: | Line 98: | ||
==Coordination chemistry principles== |
==Coordination chemistry principles== |
||
[[Image:Metal-EDTA.svg|thumb|left|150px|Metal-EDTA [[chelate]]]] |
[[Image:Metal-EDTA.svg|thumb|left|150px|Metal-EDTA [[chelate]]]] |
||
In [[coordination chemistry]], EDTA<sup>4-</sup> is a member of the [[ |
In [[coordination chemistry]], EDTA<sup>4-</sup> is a member of the [[aminopolycarboxylic acid] family of ligands. EDTA<sup>4-</sup> usually binds to a metal cation through its two amines and four carboxylates. Many of the resulting [[complex (chemistry)|coordination compound]]s adopt [[octahedral geometry]]. Although of little consequence for its applications, these octahedral complexes are [[Chirality (chemistry)|chiral]]. The anion [Co(EDTA)]<sup>−</sup> has been resolved into [[enantiomer]]s.<ref>Kirchner, S. Barium (Ethylenediaminetetracetato) Cobalt(III) 4-Hydrate" Inorganic Syntheses, 1957, Volume 5, pages 186-188. {{DOI|10.1002/9780470132364.ch52}}</ref> Many complexes of EDTA<sup>4-</sup> adopt more complex structures due to (i) the formation of an additional bond to water, i.e. seven-coordinate complexes, or (ii) the displacement of one carboxylate arm by water. Ferric complex of EDTA is seven-coordinate.<ref>J. M. López-Alcalá, M. C. Puerta-Vizcaíno, F. González-Vílchez, E. N. Duesler and R. E. Tapscott "A redetermination of sodium aqua[ethylenediaminetetraacetato(4-)]ferrate(III) dihydrate, Na[Fe(C<sub>10</sub>H<sub>12</sub>N<sup>2</sup>O<sup>8</sup>)(H<sup>2</sup>O)].2H<sub>2</sub>O" Acta Cryst. (1984). C40, 939-941. {{DOI|10.1107/S0108270184006338}}</ref> Early work on the development of EDTA was undertaken by [[Gerold Schwarzenbach]] in the 1940s.<ref>[http://www.chm.bris.ac.uk/motm/edta/edtah.htm Edta - Motm]</ref> EDTA forms especially strong complexes with Mn(II), Cu(II), Fe(III), Pb (II) and Co(III).<ref>{{cite book | last = Holleman | first = A. F. | coauthors = Wiberg, E. | title = Inorganic Chemistry | publisher = Academic Press | location = San Diego | year = 2001 | doi = | isbn = 0-12-352651-5}}</ref> |
||
Several features of EDTA's complexes are relevant to its applications. First, because of its high [[denticity]], this ligand has a high affinity for metal cations: |
Several features of EDTA's complexes are relevant to its applications. First, because of its high [[denticity]], this ligand has a high affinity for metal cations: |
||
Revision as of 00:32, 26 August 2013
| |
 Di-sodium EDTA
| |
| Names | |
|---|---|
| Systematic IUPAC name
2-({2-[Bis(carboxymethyl)amino]ethyl}(carboxymethyl)amino)acetic acid | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| Abbreviations | EDTA, H4EDTA |
| 1716295 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.000.409 |
| EC Number |
|
| 144943 | |
| KEGG | |
| MeSH | Edetic+Acid |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 3077 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H16N2O8 | |
| Molar mass | 292.244 g·mol−1 |
| Appearance | Colourless crystals |
| Density | 860 mg mL−1 (at 20 °C) |
| log P | −0.836 |
| Acidity (pKa) | 1.782 |
| Basicity (pKb) | 12.215 |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
−1.7654–−1.7580 MJ mol−1 |
Std enthalpy of
combustion (ΔcH⦵298) |
−4.4617–−4.4545 MJ mol−1 |
| Pharmacology | |
| |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H319 | |
| P305+P351+P338 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
2.580 g kg−1 (oral, rat) |
| Related compounds | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ethylenediaminetetraacetic acid, widely abbreviated as EDTA (for other names, see Table), is an aminopolycarboxylic acid and a colourless, water-soluble solid. Its conjugate base is named ethylenediaminetetraacetate. It is widely used to dissolve limescale. Its usefulness arises because of its role as a hexadentate ("six-toothed") ligand and chelating agent, i.e. its ability to "sequester" metal ions such as Ca2+ and Fe3+. After being bound by EDTA, metal ions remain in solution but exhibit diminished reactivity. EDTA is produced as several salts, notably disodium EDTA and calcium disodium EDTA.
Synthesis
The compound was first described in 1935 by Ferdinand Munz, who prepared the compound from ethylenediamine and chloroacetic acid.[1] Today, EDTA is mainly synthesised from ethylenediamine (1,2-diaminoethane), formaldehyde, and sodium cyanide.[2] This route yields the sodium salt, which can be converted in a subsequent step into the acid forms:
- H2NCH2CH2NH2 + 4 CH2O + 4 NaCN + 4 H2O → (NaO2CCH2)2NCH2CH2N(CH2CO2Na)2 + 4 NH3
- (NaO2CCH2)2NCH2CH2N(CH2CO2Na)2 + 4 HCl → (HO2CCH2)2NCH2CH2N(CH2CO2H)2 + 4 NaCl
In this way, about 80M kilograms are produced each year. Impurities cogenerated by this route include glycine and nitrilotriacetic acid; they arise from reactions of the ammonia coproduct.[3]
Nomenclature
To describe EDTA and its various protonated forms, chemists distinguish between EDTA4−, the conjugate base that is the ligand, and H4EDTA, the precursor to that ligand. At very low pH (very acidic conditions) the fully protonated H6EDTA2+ form predominates, whereas at very high pH or very basic condition, the fully deprotonated Y4− form is prevalent. In this article, the term EDTA is used to mean H4-xEDTAx-, whereas in its complexes EDTA4- stands for the tetra-deprotonated ligand.
Coordination chemistry principles

In coordination chemistry, EDTA4- is a member of the [[aminopolycarboxylic acid] family of ligands. EDTA4- usually binds to a metal cation through its two amines and four carboxylates. Many of the resulting coordination compounds adopt octahedral geometry. Although of little consequence for its applications, these octahedral complexes are chiral. The anion [Co(EDTA)]− has been resolved into enantiomers.[4] Many complexes of EDTA4- adopt more complex structures due to (i) the formation of an additional bond to water, i.e. seven-coordinate complexes, or (ii) the displacement of one carboxylate arm by water. Ferric complex of EDTA is seven-coordinate.[5] Early work on the development of EDTA was undertaken by Gerold Schwarzenbach in the 1940s.[6] EDTA forms especially strong complexes with Mn(II), Cu(II), Fe(III), Pb (II) and Co(III).[7]
Several features of EDTA's complexes are relevant to its applications. First, because of its high denticity, this ligand has a high affinity for metal cations:
- [Fe(H2O)6]3+ + H4EDTA [Fe(EDTA)]− + 6 H2O + 4 H+ (Keq = 1025.1)
Written in this way, the equilibrium quotient shows that metal ions compete with protons for binding to EDTA. Because metal ions are extensively enveloped by EDTA, their catalytic properties are often suppressed. Finally, since complexes of EDTA4- are anionic, they tend to be highly soluble in water. For this reason, EDTA is able to dissolve deposits of metal oxides and carbonates.
Uses
Industry
In industry, EDTA is mainly used to sequester metal ions in aqueous solution. In the textile industry, it prevents metal ion impurities from modifying colours of dyed products. In the pulp and paper industry, EDTA inhibits the ability of metal ions, especially Mn2+, from catalyzing the disproportionation of hydrogen peroxide, which is used in "chlorine-free bleaching." In a similar manner, EDTA is added to some food as a preservative or stabilizer to prevent catalytic oxidative decoloration, which is catalyzed by metal ions.[8] In soft drinks containing ascorbic acid and sodium benzoate, EDTA mitigates formation of benzene (a carcinogen).[9]
The reduction of water hardness in laundry applications and the dissolution of scale in boilers both rely on EDTA and related complexants to bind Ca2+, Mg2+, as well as other metal ions. Once bound to EDTA, these metal centers tend not to form precipitates or to interfere with the action of the soaps and detergents. For similar reasons, cleaning solutions often contain EDTA.
The solubilization of ferric ions, at or below near neutral pH can be accomplished using EDTA. This property is useful in agriculture including hydroponics. However, given the pH dependence of ligand formation, EDTA is not helpful for improving Fe solubility in above neutral soils.[10] Otherwise, at near-neutral pH and above, iron(III) forms insoluble salts, which are less bioavailable to susceptible plant species. Aqueous [Fe(edta)]- is used for removing ("scrubbing") hydrogen sulfide from gas streams. This conversion is achieved by oxidizing the hydrogen sulfur to elemental sulfur, which is non-volatile:
- 2 [Fe(edta)]- + H2S → 2 [Fe(edta)]2− + S + 2 H+
In this application, the ferric center is reduced to its ferrous derivative, which can then be reoxidized by air. In similar manner, nitrogen oxides are removed from gas streams using [Fe(edta)]2-. The oxidizing properties of [Fe(edta)]- are also exploited in photography, where it is used to solubilize silver particles.[3]
EDTA was used in the separation of the lanthanide metals by ion-exchange chromatography. Perfected by F.H. Spedding et al. in 1954, the method relies on the steady increase in stability constant of the lanthanide EDTA complexes with atomic number. Using sulfonated polystyrene beads and copper(II) as a retaining ion, EDTA causes the lanthanides to migrate down the column of resin while separating into bands of pure lanthanide. The lanthanides elute in order of decreasing atomic number. Due to the expense of this method, relative to counter-current solvent extraction, ion-exchange is now used only to obtain the highest purities of lanthanide (typically greater than 4N, 99.99%).[citation needed]
Medicine
EDTA is used to bind metal ions in the practice of chelation therapy, e.g., for treating mercury and lead poisoning.[11] It is used in a similar manner to remove excess iron from the body. This therapy is used to treat the complication of repeated blood transfusions, as would be applied to treat thalassaemia. The U.S. FDA approved the use of EDTA for lead poisoning[12] on July 16, 1953, under the brand name of Versenate,[13] which was licensed to the pharmaceutical company Riker. Alternative medical practitioners believe EDTA acts as a powerful antioxidant to prevent free radicals from injuring blood vessel walls, therefore reducing atherosclerosis.[14] The U.S. FDA has not approved it for the treatment of atherosclerosis.[15]
Dentists and endodontists use EDTA solutions to remove inorganic debris (smear layer) and lubricate the canals in endodontics. This procedure helps prepare root canals for obturation. Furthermore, EDTA solutions with the addition of a surfactant loosen up calcifications inside a root canal and allow instrumentation (canals shaping) and facilitate apical advancement of a file in a tight/calcified root canal towards the apex. It serves as a preservative (usually to enhance the action of another preservative such as benzalkonium chloride or thiomersal) in ocular preparations and eyedrops.[16] In evaluating kidney function, the complex [Cr(edta)]- is administered intravenously and its filtration into the urine is monitored. This method is useful for evaluating glomerular filtration rate.[17]
EDTA is used extensively in the analysis of blood. It is an anticoagulant for blood samples for CBC/FBEs.
Laboratory studies also suggest that EDTA chelation may prevent collection of platelets on the lining of the vessel [such as arteries] (which can otherwise lead to formation of blood clots, which itself is associated with atheromatous plaque formation or rupture, and thereby ultimately disrupts blood flow). These ideas have so far been proven ineffective;[18] however, a major clinical study of the effects of EDTA on coronary arteries is currently (2008) proceeding.[19] EDTA played a role in the O.J. Simpson trial when the defense alleged that one of the blood samples collected from Simpson's estate was found to contain traces of the compound.[20]
EDTA is a slime dispersant, and has been found to be highly effective in reducing bacterial growth during implantation of intraocular lenses (IOLs).[21]
Cosmetics
In shampoos, cleaners and other personal care products EDTA salts are added as a sequestering agent to improve their stability in air.[22]
Laboratory applications
In the laboratory, EDTA is widely used for scavenging metal ions: In biochemistry and molecular biology, ion depletion is commonly used to deactivate metal-dependent enzymes, either as an assay for their reactivity or to suppress damage to DNA or proteins.[23] In analytical chemistry, EDTA is used in complexometric titrations and analysis of water hardness or as a masking agent to sequester metal ions that would interfere with the analyses. EDTA finds many specialized uses in the biomedical laboratories, such as in veterinary ophthalmology as an anticollagenase to prevent the worsening of corneal ulcers in animals. In tissue culture EDTA is used as a chelating agent that binds to calcium and prevents joining of cadherins between cells, preventing clumping of cells grown in liquid suspension, or detaching adherent cells for passaging. In histopathology, EDTA can be used as a decalcifying agent making it possible to cut sections using a microtome once the tissue sample is demineralised. EDTA is also known to inhibit a range of metallopeptidases, the method of inhibition occurs via the chelation of the metal ion required for catalytic activity.[24] EDTA can also be used to test for bioavailability of heavy metals in sediments.
Toxicity and environmental considerations
EDTA is in such widespread use that questions have been raised whether it is a persistent organic pollutant. Research indicates that under many conditions, EDTA is fully biodegradable. However, when simulating certain non-optimal degradation conditions (high pH), less than 1% of the EDTA was degraded instead to ethylenediaminetriacetic acid, which can then cyclize to 3-ketopiperazine-N,N-diacetate, a cumulative, persistent, organic chemical with unknown effects on the environment.[25] An alternative chelating agent with fewer environmental pollution implications is EDDS.
EDTA exhibits low acute toxicity with LD50 (rat) of 2.0 – 2.2 g/kg.[3] It has been found to be both cytotoxic and weakly genotoxic in laboratory animals. Oral exposures have been noted to cause reproductive and developmental effects.[22] The same study by Lanigan[22] also found that both dermal exposure to EDTA in most cosmetic formulations and inhalation exposure to EDTA in aerosolized cosmetic formulations would produce exposure levels below those seen to be toxic in oral dosing studies.
Methods of detection and analysis
The most sensitive method of detecting and measuring EDTA in biological samples is selected-reaction-monitoring capillary-electrophoresis mass-spectrometry (abbreviation SRM-CE/MS), which has a detection limit of 7.3 ng/mL in human plasma and a quantitation limit of 15 ng/mL.[26] This method works with sample volumes as small as ~7-8 nL.[26]
EDTA has also been measured in non-alcoholic beverages using high performance liquid chromatography (HPLC) at a level of 2.0 μg/mL.[27][28]
See also
References
- ^ F. Münz "Polyamino carboxylic acids to I. G. Farbenindustrie, DE 718 981, 1935; US 2 130 505, 1938.
- ^ Synthesis of EDTA
- ^ a b c J. Roger Hart "Ethylenediaminetetraacetic Acid and Related Chelating Agents" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005.doi:10.1002/14356007.a10_095
- ^ Kirchner, S. Barium (Ethylenediaminetetracetato) Cobalt(III) 4-Hydrate" Inorganic Syntheses, 1957, Volume 5, pages 186-188. doi:10.1002/9780470132364.ch52
- ^ J. M. López-Alcalá, M. C. Puerta-Vizcaíno, F. González-Vílchez, E. N. Duesler and R. E. Tapscott "A redetermination of sodium aqua[ethylenediaminetetraacetato(4-)]ferrate(III) dihydrate, Na[Fe(C10H12N2O8)(H2O)].2H2O" Acta Cryst. (1984). C40, 939-941. doi:10.1107/S0108270184006338
- ^ Edta - Motm
- ^ Holleman, A. F. (2001). Inorganic Chemistry. San Diego: Academic Press. ISBN 0-12-352651-5.
{{cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Furia T (1964). "EDTA in Foods – A technical review". Food Technology. 18 (12): 1874–1882.
- ^ US Food and Drug Administration: Center for Food Safety and Applied Nutrition Questions and Answers on the Occurrence of Benzene in Soft Drinks and Other Beverages
- ^ Norvell and Lindsay, “Reactions of EDTA Complexes of Fe, Zn, Mn, and Cu with Soils1.”
- ^ Ruth DeBusk; et al. (2002). "Ethylenediaminetetraacetic acid (EDTA)". Retrieved 2007-07-25.
{{cite web}}: Explicit use of et al. in:|author=(help) - ^ "Calcium Disodium Versenate (Edetate Calcium Disodium) Injection [Graceway Pharmaceuticals, Llc]". Dailymed.nlm.nih.gov. Retrieved 2013-01-01.
- ^ "Drugs@FDA: FDA Approved Drug Products". Accessdata.fda.gov. Retrieved 2013-01-01.
- ^ "Home > Medical Reference > Complementary Medicine > EDTA overview". University of Maryland Medical Center. Retrieved 16 December 2009.
- ^ "Postmarket Drug Safety Information for Patients and Providers > Questions and Answers on Edetate Disodium (marketed as Endrate and generic products)". Retrieved 16 December 2010.
- ^ See "les conservateurs en opthalmologie" Doctors Patrice Vo Tan & Yves lachkar, Librarie Médicale Théa.
- ^ Shirley, D.G., Walter, S.J. and Noormohamed, F.H. (2002). "Natriuretic effect of caffeine: assessment of segmental sodium reabsorption in humans". Clinical Science. 103 (5): 461–466. doi:10.1042/CS20020055. PMID 12401118.
{{cite journal}}:|access-date=requires|url=(help)CS1 maint: multiple names: authors list (link) - ^ Green, Saul (December 14, 2002). "EDTA Chelation Therapy for Atherosclerosis And Degenerative Diseases: Implausibility and Paradoxical Oxidant Effects". Quackwatch. Retrieved 16 December 2009.
{{cite web}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ "Trial to Assess Chelation Therapy (TACT) - Full Text View". ClinicalTrials.gov. Retrieved 2013-01-01.
- ^ Margolock, David (July 26, 1995). "F.B.I. Disputes Simpson Defense on Tainted Blood". The New York Times. pp. A12. Retrieved 16 December 2009.
- ^ "Impact of slime dispersants and anti-adhesives on in vitro biofilm formation of Staphylococcus epidermidis on intraocular lenses and on antibiotic activities". Jac.oxfordjournals.org. Retrieved 2013-01-01.
- ^ a b c Lanigan RS and Yamarik TA (2002). "Final report on the safety assessment of EDTA, calcium disodium EDTA, diammonium EDTA, dipotassium EDTA, disodium EDTA, TEA-EDTA, tetrasodium EDTA, tripotassium EDTA, trisodium EDTA, HEDTA, and trisodium HEDTA". Int J Toxicol. 21 Suppl 2: 95–142. doi:10.1080/10915810290096522. PMID 12396676.
{{cite journal}}:|access-date=requires|url=(help) - ^ Dominguez, K; Ward, WS (2009). "A novel nuclease activity that is activated by Ca(2+) chelated to EGTA". Systems Biology in Reproductive Medicine. 55 (5–6): 193–99. doi:10.3109/19396360903234052.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Auld D.S "Removal and replacement of metal ions in metallopeptidases " Methods Enzymol (1995) 248, 228-242.
- ^ Zhiwen Yuan, Jeanne M. VanBriesen "The Formation of Intermediates in EDTA and NTA Biodegradation" Environmental Engineering Science 2006, volume 23, pp. 533-544. doi:10.1089/ees.2006.23.533
- ^ a b Robin L. Sheppard, and Jack Henion (1997). "Determining EDTA in Blood" (– Scholar search). Analytical Chemistry. 69 (15): 477A–480A. doi:10.1021/ac971726p. PMID 9253241. Retrieved 2007-07-25.
{{cite journal}}: External link in|format= - ^ S. Loyaux-Lawniczak, J. Douch, and P. Behra (1999). "Optimisation of the analytical detection of EDTA by HPLC in natural waters". Fresenius' J. Anal. Chem. 364 (8): 727–731. doi:10.1007/s002160051422. Retrieved 2007-07-25.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Carolina E. Cagnasso, Laura B. López, Viviana G.
Rodríguez and Mirta E. Valencia (2006). "Development and validation of a method for the determination of EDTA in non-alcoholic drinks by HPLC". Journal of Food Composition and Analysis. 20 (3–4): 248. doi:10.1016/j.jfca.2006.05.008.
{{cite journal}}:|access-date=requires|url=(help); Unknown parameter|month=ignored (help); line feed character in|author=at position 49 (help)CS1 maint: multiple names: authors list (link)
External links
- The MEROPS online database for peptidases and their inhibitors: EDTA
- Lanigan RS, Yamarik TA (2002). "Final report on the safety assessment of EDTA, calcium disodium EDTA, diammonium EDTA, dipotassium EDTA, disodium EDTA, TEA-EDTA, tetrasodium EDTA, tripotassium EDTA, trisodium EDTA, HEDTA, and trisodium HEDTA". Int. J. Toxicol. 21 Suppl 2: 95–142. doi:10.1080/10915810290096522. PMID 12396676.
- EDTA: Molecule of the Month
- EDTA Determination of Total Water Hardness
- EDTA: the chelating agent under environmental scrutiny, Química Nova, Nov.-Dec., 2003 (text version)
- EDTA: the chelating agent under environmental scrutiny, Química Nova, Nov.-Dec., 2003 (PDF version)


