m missed file |
populated new fields in {{drugbox}} and reordered per bot approval. Report errors and suggestions to User_talk:BogBot |
||
| Line 1: | Line 1: | ||
{{ |
{{Drugbox |
||
| Verifiedfields = changed |
| Verifiedfields = changed |
||
| Watchedfields = changed |
| Watchedfields = changed |
||
| Line 5: | Line 5: | ||
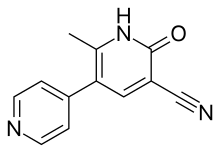
| IUPAC_name = 2-methyl-6-oxo-1,6-dihydro-3,4'-bipyridine-5-carbonitrile |
| IUPAC_name = 2-methyl-6-oxo-1,6-dihydro-3,4'-bipyridine-5-carbonitrile |
||
| image = Milrinone.svg |
| image = Milrinone.svg |
||
<!--Clinical data--> |
|||
| tradename = |
|||
| Drugs.com = {{drugs.com|monograph|milrinone-lactate}} |
|||
| MedlinePlus = a601020 |
|||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
<!--Pharmacokinetic data--> |
|||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
<!--Identifiers--> |
|||
| CASNo_Ref = {{cascite|correct|CAS}} |
| CASNo_Ref = {{cascite|correct|CAS}} |
||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
||
| ChemSpiderID = 4052 |
| ChemSpiderID = 4052 |
||
| UNII_Ref = {{fdacite|correct|FDA}} |
| UNII_Ref = {{fdacite|correct|FDA}} |
||
| UNII = JU9YAX04C7 |
| UNII = JU9YAX04C7 |
||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ChEMBL_Ref = {{ebicite|correct|EBI}} |
| ChEMBL_Ref = {{ebicite|correct|EBI}} |
||
| ChEMBL = 189 |
| ChEMBL = 189 |
||
<!--Chemical data--> |
|||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| StdInChI_Ref = {{stdinchicite|correct|chemspider}} |
| StdInChI_Ref = {{stdinchicite|correct|chemspider}} |
||
| StdInChI = 1S/C12H9N3O/c1-8-11(9-2-4-14-5-3-9)6-10(7-13)12(16)15-8/h2-6H,1H3,(H,15,16) |
| StdInChI = 1S/C12H9N3O/c1-8-11(9-2-4-14-5-3-9)6-10(7-13)12(16)15-8/h2-6H,1H3,(H,15,16) |
||
| StdInChIKey_Ref = {{stdinchicite|correct|chemspider}} |
| StdInChIKey_Ref = {{stdinchicite|correct|chemspider}} |
||
| StdInChIKey = PZRHRDRVRGEVNW-UHFFFAOYSA-N |
| StdInChIKey = PZRHRDRVRGEVNW-UHFFFAOYSA-N |
||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| melting_point = 315 |
| melting_point = 315 |
||
| boiling_point = 449 |
| boiling_point = 449 |
||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
}} |
}} |
||
'''Milrinone''' (Primacor) is a [[phosphodiesterase 3 inhibitor]]. It potentiates the effect of [[cyclic adenosine monophosphate]] (cAMP). |
'''Milrinone''' (Primacor) is a [[phosphodiesterase 3 inhibitor]]. It potentiates the effect of [[cyclic adenosine monophosphate]] (cAMP). |
||
Revision as of 21:52, 30 August 2011
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601020 |
| Routes of administration | IV only |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 100% (as IV bolus, infusion) |
| Protein binding | 70 to 80% |
| Metabolism | Hepatic (12%) |
| Elimination half-life | 2.3 hours (mean, in CHF) |
| Excretion | Urine (85% as unchanged drug) within 24 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.071.709 |
| Chemical and physical data | |
| Formula | C12H9N3O |
| Molar mass | 211.219 g/mol g·mol−1 |
| 3D model (JSmol) | |
| Density | 1.344 g/cm3 |
| Melting point | 315 °C (599 °F) |
| Boiling point | 449 °C (840 °F) |
| |
| |
| | |
Milrinone (Primacor) is a phosphodiesterase 3 inhibitor. It potentiates the effect of cyclic adenosine monophosphate (cAMP).
Milrinone also enhances relaxation of the left ventricle by increasing Ca2+-ATPase activity on the cardiac sarcoplasmic reticulum. This increases calcium ion uptake.
It has positive inotropic, vasodilating and minimal chronotropic effects. It is used in the management of heart failure only when conventional treatment with vasodilators and diuretics has proven insufficient. This is due to the potentially fatal adverse effects of milrinone, including ventricular arrhythmias.
Whereas beneficial hemodynamic effects are shown (at least short-term), several studies have shown no or a negative effect on mortality rates of hospitalized patients receiving milrinone.[1]
One negative side to the use of milrinone is the short half-life (1 to 2 hours). This can result in a prolonged weaning and possible adverse outcomes from stopping this medication rapidly.
Synthesis
Singh, B.; 1983, U.S. patent 4,413,127.
References
External links

