Updating {{drugbox}} (no changed fields - added verified revid - updated 'DrugBank_Ref', 'ChEBI_Ref') per Chem/Drugbox validation (report errors or [[u |
populated new fields in {{drugbox}} and reordered per bot approval. Report errors and suggestions to User_talk:BogBot |
||
| Line 1: | Line 1: | ||
{{ |
{{Drugbox |
||
| verifiedrevid = 443584620 |
| verifiedrevid = 443584620 |
||
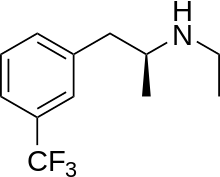
| IUPAC_name = (''S'')-''N''-Ethyl-1-[3-(trifluoromethyl)phenyl]-propan-2-amine |
| IUPAC_name = (''S'')-''N''-Ethyl-1-[3-(trifluoromethyl)phenyl]-propan-2-amine |
||
| image = Dexfenfluramine.svg |
| image = Dexfenfluramine.svg |
||
| ⚫ | |||
<!--Clinical data--> |
|||
| ⚫ | |||
| tradename = |
|||
| ⚫ | |||
| MedlinePlus = a682088 |
|||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
<!--Pharmacokinetic data--> |
|||
| ⚫ | |||
| ⚫ | |||
<!--Identifiers--> |
|||
| CASNo_Ref = {{cascite|correct|CAS}} |
| CASNo_Ref = {{cascite|correct|CAS}} |
||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| CAS_number = 3239-44-9 |
| CAS_number = 3239-44-9 |
||
| ATC_prefix = A08 |
| ATC_prefix = A08 |
||
| ATC_suffix = AA04 |
| ATC_suffix = AA04 |
||
| ATC_supplemental = |
|||
| ⚫ | |||
| ⚫ | |||
| PubChem = 66265 |
| PubChem = 66265 |
||
| DrugBank_Ref = {{drugbankcite|correct|drugbank}} |
| DrugBank_Ref = {{drugbankcite|correct|drugbank}} |
||
| DrugBank = APRD00648 |
| DrugBank = APRD00648 |
||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| KEGG_Ref = {{keggcite|correct|kegg}} |
| KEGG_Ref = {{keggcite|correct|kegg}} |
||
| KEGG = D07805 |
| KEGG = D07805 |
||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
<!--Chemical data--> |
|||
| ⚫ | |||
| molecular_weight = 231.257 g/mol |
| molecular_weight = 231.257 g/mol |
||
| ⚫ | |||
| bioavailability = |
|||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| metabolism = |
|||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| pregnancy_category = |
|||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| routes_of_administration = |
|||
}} |
}} |
||
<!-- Deleted image removed: [[Image:ReduxAd.png|thumb|300px|left|A direct-to-physician advertisement for Redux.]] --> |
<!-- Deleted image removed: [[Image:ReduxAd.png|thumb|300px|left|A direct-to-physician advertisement for Redux.]] --> |
||
Revision as of 21:30, 30 August 2011
 | |
| Clinical data | |
|---|---|
| MedlinePlus | a682088 |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 36% |
| Elimination half-life | 17-20 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C12H16F3N |
| Molar mass | 231.257 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Dexfenfluramine, marketed as dexfenfluramine hydrochloride under the name Redux, is a serotoninergic anorectic drug: it reduces appetite by increasing the amount of extracellular serotonin in the brain. It is the d-enantiomer of fenfluramine and is structually similar to amphetamine, but lacks any psychologically stimulating effects.
Dexfenfluramine was for some years in the mid-1990s approved by the United States Food and Drug Administration for the purposes of weight loss. However, following multiple concerns about the cardiovascular side-effects of the drug, such approval was withdrawn and it was retired from the market in 1997. After it was removed in the US, dexfenfluramine was also pulled out in other global markets. It was later superseded by sibutramine, which although initially considered a safer alternative to both dexfenfluramine and fenfluramine[citation needed], was removed from the US market as well in 2010[2].
The drug was manufactured by Interneuron Pharmaceuticals, a company co-founded by Richard Wurtman, aimed at marketing discoveries by Massachusetts Institute of Technology scientists.[3] In the case of Redux, Interneuron's manufacture was under licence to Wyeth-Ayerst Laboratories.[4] Although at the time of its release, some optimism prevailed that it might herald a new approach[citation needed], there remained some reservations amongst neurologists, twenty-two of whom petitioned the FDA to delay approval[citation needed]. Their concern was based on the work of George A. Ricaurte whose techniques and conclusions were later questioned.[citation needed]
In actuality, most conversant with the discussion felt that dexfenfuramine's only advantage was patent protection for its licensed companies. Racemic fenfluramine was far cheaper.
See also
References
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 Oct 2023.
- ^ http://online.wsj.com/article/BT-CO-20101008-710904.html.
{{cite news}}: Missing or empty|title=(help) [dead link] - ^ Lemonick, Michael D; Dowell, William; Nash, J. Madeleine; Ramirez, Ainissa; Reid, Brian; Ressner, Jeffrey (Sep 23, 1996), . (ed.), "The New Miracle Drug?", Time, retrieved 03 October 2010
{{citation}}:|chapter=ignored (help);|editor=has numeric name (help); Check date values in:|accessdate=(help); External link in|chapterurl=|chapterurl=ignored (|chapter-url=suggested) (help) - ^ Lemonick, Michael D; Nash, J. Madeleine; Park, Alice; Thompson, Dick (Sep 29, 1997), . (ed.), "The Mood Molecule", Time, retrieved 04 October 2010
{{citation}}:|chapter=ignored (help);|editor=has numeric name (help); Check date values in:|accessdate=(help); External link in|chapterurl=|chapterurl=ignored (|chapter-url=suggested) (help)
External links
- FDA Announces Withdrawal Fenfluramine and Dexfenfluramine (Fen-Phen)
- Drug description
- Dexfenfluramine hydrochloride
- Questions and Answers about Withdrawal of Fenfluramine (Pondimin) and Dexfenfluramine (Redux)
- Frontline: Dangerous prescriptions - Interview with Leo Lutwak, M.D. in which he discuses the side effects of fenfluramine, its successor Redux, and the Fen-Phen combination
