Content deleted Content added
Updating {{drugbox}} (changes to verified and watched fields - updated 'DrugBank_Ref', 'ChEBI_Ref') per Chem/Drugbox validation (report errors or [[use |
populated new fields in {{drugbox}} and reordered per bot approval. Report errors and suggestions to User_talk:BogBot |
||
| Line 1: | Line 1: | ||
{{ |
{{Drugbox |
||
| Verifiedfields = changed |
| Verifiedfields = changed |
||
| Watchedfields = changed |
| Watchedfields = changed |
||
| Line 5: | Line 5: | ||
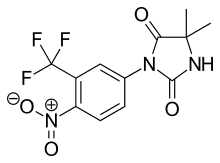
| IUPAC_name = 5,5-dimethyl-3-[4-nitro-3-(trifluoromethyl)phenyl]<br>imidazolidine-2,4-dione |
| IUPAC_name = 5,5-dimethyl-3-[4-nitro-3-(trifluoromethyl)phenyl]<br>imidazolidine-2,4-dione |
||
| image = Nilutamide.svg |
| image = Nilutamide.svg |
||
<!--Clinical data--> |
|||
| tradename = Nilandron |
|||
| Drugs.com = {{drugs.com|monograph|nilutamide}} |
|||
| MedlinePlus = a697044 |
|||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
<!--Pharmacokinetic data--> |
|||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
<!--Identifiers--> |
|||
| CASNo_Ref = {{cascite|correct|CAS}} |
| CASNo_Ref = {{cascite|correct|CAS}} |
||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
||
| ChemSpiderID = 4337 |
| ChemSpiderID = 4337 |
||
| UNII_Ref = {{fdacite|correct|FDA}} |
| UNII_Ref = {{fdacite|correct|FDA}} |
||
| UNII = 51G6I8B902 |
| UNII = 51G6I8B902 |
||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ChEMBL_Ref = {{ebicite|correct|EBI}} |
| ChEMBL_Ref = {{ebicite|correct|EBI}} |
||
| ChEMBL = 1274 |
| ChEMBL = 1274 |
||
<!--Chemical data--> |
|||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| StdInChI_Ref = {{stdinchicite|correct|chemspider}} |
| StdInChI_Ref = {{stdinchicite|correct|chemspider}} |
||
| StdInChI = 1S/C12H10F3N3O4/c1-11(2)9(19)17(10(20)16-11)6-3-4-8(18(21)22)7(5-6)12(13,14)15/h3-5H,1-2H3,(H,16,20) |
| StdInChI = 1S/C12H10F3N3O4/c1-11(2)9(19)17(10(20)16-11)6-3-4-8(18(21)22)7(5-6)12(13,14)15/h3-5H,1-2H3,(H,16,20) |
||
| StdInChIKey_Ref = {{stdinchicite|correct|chemspider}} |
| StdInChIKey_Ref = {{stdinchicite|correct|chemspider}} |
||
| StdInChIKey = XWXYUMMDTVBTOU-UHFFFAOYSA-N |
| StdInChIKey = XWXYUMMDTVBTOU-UHFFFAOYSA-N |
||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
| ⚫ | |||
}} |
}} |
||
'''Nilutamide''' is an [[antiandrogen]] medication used in the treatment of advanced stage [[prostate cancer]]. Nilutamide blocks the [[androgen receptor]], preventing its interaction with [[testosterone]]. Because most prostate cancer cells rely on the stimulation of the androgen receptor for growth and survival, nilutamide can prolong life in men with prostate cancer. Nilutamide is marketed under the name Nilandron in the United States and under the name Anandron in Canada. |
'''Nilutamide''' is an [[antiandrogen]] medication used in the treatment of advanced stage [[prostate cancer]]. Nilutamide blocks the [[androgen receptor]], preventing its interaction with [[testosterone]]. Because most prostate cancer cells rely on the stimulation of the androgen receptor for growth and survival, nilutamide can prolong life in men with prostate cancer. Nilutamide is marketed under the name Nilandron in the United States and under the name Anandron in Canada. |
||
Revision as of 04:41, 30 August 2011
 | |
| Clinical data | |
|---|---|
| Trade names | Nilandron |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a697044 |
| Routes of administration | oral |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 38.0 to 59.1 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.153.268 |
| Chemical and physical data | |
| Formula | C12H10F3N3O4 |
| Molar mass | 317.221 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Nilutamide is an antiandrogen medication used in the treatment of advanced stage prostate cancer. Nilutamide blocks the androgen receptor, preventing its interaction with testosterone. Because most prostate cancer cells rely on the stimulation of the androgen receptor for growth and survival, nilutamide can prolong life in men with prostate cancer. Nilutamide is marketed under the name Nilandron in the United States and under the name Anandron in Canada.
References
- Kassouf W, Tanguay S, Aprikian AG. Nilutamide as second line hormone therapy for prostate cancer after androgen ablation fails. J. Urol. 2003 May;169(5):1742-4. PMID 12686822
- Moguilewsky M, Bertagna C, Hucher M. (1987) Pharmacological and clinical studies of the antiandrogen Anandron (nilutamide). J Steroid Biochem. (4-6):871-5.
- Hsieh AC, Ryan CJ. (2008) Novel concepts in androgen receptor blockade. Cancer J. 14(1):11-14.
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 Oct 2023.
