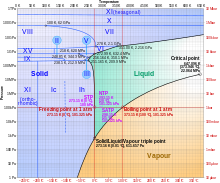
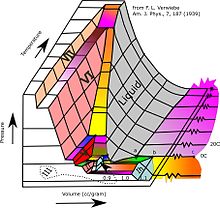

Currently, 19 solid phases of water (both crystalline and amorphous) have been observed at various densities, along with hypothetical proposed phases of ice that have not been observed.[2]
Theory
Most liquids under increased pressure freeze at higher temperatures because the pressure helps to hold the molecules together. However, the strong hydrogen bonds in water make it different: for some pressures higher than 1 atm (0.10 MPa), water freezes at a temperature below 0 °C, as shown in the phase diagram below. The melting of ice under high pressures is thought to contribute to the movement of glaciers.[3]
Ice, water, and water vapour can coexist at the triple point, which is exactly 273.16 K (0.01 °C) at a pressure of 611.657 Pa.[4][5] The kelvin was defined as 1/273.16 of the difference between this triple point and absolute zero,[6] though this definition changed in May 2019.[7] Unlike most other solids, ice is difficult to superheat. In an experiment, ice at −3 °C was superheated to about 17 °C for about 250 picoseconds.[8]
Subjected to higher pressures and varying temperatures, ice can form in nineteen separate known crystalline phases. With care, at least fifteen of these phases (one of the known exceptions being ice X) can be recovered at ambient pressure and low temperature in metastable form.[9][10] The types are differentiated by their crystalline structure, proton ordering,[11] and density. There are also two metastable phases of ice under pressure, both fully hydrogen-disordered; these are IV and XII. Ice XII was discovered in 1996. In 2006, XIII and XIV were discovered.[12] Ices XI, XIII, and XIV are hydrogen-ordered forms of ices Ih, V, and XII respectively. In 2009, ice XV was found at extremely high pressures and −143 °C.[13] At even higher pressures, ice is predicted to become a metal; this has been variously estimated to occur at 1.55 TPa[14] or 5.62 TPa.[15]
Non-crystalline ice
As well as crystalline forms, solid water can exist in amorphous states as amorphous solid water (ASW) of varying densities. Water in the interstellar medium is dominated by amorphous ice, making it likely the most common form of water in the universe. Low-density ASW (LDA), also known as hyperquenched glassy water, may be responsible for noctilucent clouds on Earth and is usually formed by deposition of water vapor in cold or vacuum conditions. High-density ASW (HDA) is formed by compression of ordinary ice Ih or LDA at GPa pressures. Very-high-density ASW (VHDA) is HDA slightly warmed to 160 K under 1–2 GPa pressures.
In outer space, hexagonal crystalline ice (the predominant form found on Earth) is extremely rare. Amorphous ice is more common; however, hexagonal crystalline ice can be formed by volcanic action.[16]
Ice from a theorized superionic water may possess two crystalline structures. At pressures in excess of 500,000 bars (7,300,000 psi) such superionic ice would take on a body-centered cubic structure. However, at pressures in excess of 1,000,000 bars (15,000,000 psi) the structure may shift to a more stable face-centered cubic lattice. It is speculated that superionic ice could compose the interior of ice giants such as Uranus and Neptune.[17]
Known phases
| Phase | Characteristics |
|---|---|
| Amorphous ice | Amorphous ice is ice lacking crystal structure. Amorphous ice exists in four forms: low-density (LDA) formed at atmospheric pressure, or below, medium-density (MDA), high-density (HDA) and very-high-density amorphous ice (VHDA), forming at higher pressures. LDA forms by extremely quick cooling of liquid water ("hyperquenched glassy water", HGW), by depositing water vapour on very cold substrates ("amorphous solid water", ASW) or by heating high density forms of ice at ambient pressure ("LDA"). Recently, a medium-density amorphous form ("MDA") has been shown to exist, created by ball-milling ice Ih at low temperatures.[18] |
| Ice Ih | Normal hexagonal crystalline ice. Virtually all ice in the biosphere is ice Ih, with the exception only of a small amount of ice Ic. |
| Ice Ic | A metastable cubic crystalline variant of ice. The oxygen atoms are arranged in a diamond structure. It is produced at temperatures between 130 and 220 K, and can exist up to 240 K,[19][20] when it transforms into ice Ih. It may occasionally be present in the upper atmosphere.[21] More recently, it has been shown that many samples which were described as cubic ice were actually stacking disordered ice with trigonal symmetry.[22] The first samples of ice I with cubic symmetry (i.e. cubic ice) were only reported in 2020.[23] |
| Ice II | A rhombohedral crystalline form with highly ordered structure. Formed from ice Ih by compressing it at temperature of 190–210 K. When heated, it undergoes transformation to ice III. |
| Ice III | A tetragonal crystalline ice, formed by cooling water down to 250 K at 300 MPa. Least dense of the high-pressure phases. Denser than water. |
| Ice IV | A metastable rhombohedral phase. It can be formed by heating high-density amorphous ice slowly at a pressure of 810 MPa. It does not form easily without a nucleating agent.[24] |
| Ice V | A monoclinic crystalline phase. Formed by cooling water to 253 K at 500 MPa. Most complicated structure of all the phases.[25] |
| Ice VI | A tetragonal crystalline phase. Formed by cooling water to 270 K at 1.1 GPa. Exhibits Debye relaxation.[26] |
| Ice VII | A cubic phase. The hydrogen atoms' positions are disordered. Exhibits Debye relaxation. The hydrogen bonds form two interpenetrating lattices. |
| Ice VIIt | Forms at around 5 GPa, when Ice VII becomes tetragonal.[27] |
| Ice VIII | A more ordered version of ice VII, where the hydrogen atoms assume fixed positions. It is formed from ice VII, by cooling it below 5 °C (278 K) at 2.1 GPa. |
| Ice IX | A tetragonal phase. Formed gradually from ice III by cooling it from 208 K to 165 K, stable below 140 K and pressures between 200 MPa and 400 MPa. It has density of 1.16 g/cm3, slightly higher than ordinary ice. |
| Ice X | Proton-ordered symmetric ice. Forms at pressures around 70 GPa,[28] or perhaps as low as 30 GPa.[27] |
| Ice XI | An orthorhombic, low-temperature equilibrium form of hexagonal ice. It is ferroelectric. Ice XI is considered the most stable configuration of ice Ih.[29] |
| Ice XII | A tetragonal, metastable, dense crystalline phase. It is observed in the phase space of ice V and ice VI. It can be prepared by heating high-density amorphous ice from 77 K to about 183 K at 810 MPa. It has a density of 1.3 g·cm−3 at 127 K (i.e., approximately 1.3 times denser than water). |
| Ice XIII | A monoclinic crystalline phase. Formed by cooling water to below 130 K at 500 MPa. The proton-ordered form of ice V.[30] |
| Ice XIV | An orthorhombic crystalline phase. Formed below 118 K at 1.2 GPa. The proton-ordered form of ice XII.[30] |
| Ice XV | A proton-ordered form of ice VI formed by cooling water to around 80–108 K at 1.1 GPa. |
| Ice XVI | The least dense crystalline form of water, topologically equivalent to the empty structure of sII clathrate hydrates. |
| Square ice | Square ice crystals form at room temperature when squeezed between two layers of graphene. The material was a new crystalline phase of ice when it was first reported in 2014.[31][32] The research derived from the earlier discovery that water vapor and liquid water could pass through laminated sheets of graphene oxide, unlike smaller molecules such as helium. The effect is thought to be driven by the van der Waals force, which may involve more than 10,000 atmospheres of pressure.[31] |
| Ice XVII | A porous hexagonal crystalline phase with helical channels, with density near that of ice XVI.[33][34][35] Formed by placing hydrogen-filled ice in a vacuum and increasing the temperature until the hydrogen molecules escape.[33] |
| Ice XVIII | A form of water also known as superionic water or superionic ice in which oxygen ions develop a crystalline structure while hydrogen ions move freely. |
| Ice XIX | Another phase related to ice VI formed by cooling water to around 100 K at approximately 2 GPa.[2] |
References
- ^ David, Carl (8 August 2016). "Verwiebe's '3-D' Ice phase diagram reworked". Chemistry Education Materials.
- ^ a b Metcalfe, Tom (9 March 2021). "Exotic crystals of 'ice 19' discovered". Live Science.
- ^ "The Life of a Glacier". National Snow and Data Ice Center. Archived from the original on 15 December 2014.
- ^ Wagner, Wolfgang; Saul, A.; Pruss, A. (May 1994). "International Equations for the Pressure Along the Melting and Along the Sublimation Curve of Ordinary Water Substance". Journal of Physical and Chemical Reference Data. 23 (3): 515–527. Bibcode:1994JPCRD..23..515W. doi:10.1063/1.555947.
- ^ Murphy, D. M. (2005). "Review of the vapour pressures of ice and supercooled water for atmospheric applications". Quarterly Journal of the Royal Meteorological Society. 131 (608): 1539–1565. Bibcode:2005QJRMS.131.1539M. doi:10.1256/qj.04.94. S2CID 122365938.
- ^ "SI base units". Bureau International des Poids et Mesures. Archived from the original on 16 July 2012. Retrieved 31 August 2012.
- ^ "Information for users about the proposed revision of the SI" (PDF). Bureau International des Poids et Mesures. Archived from the original (PDF) on 21 January 2018. Retrieved 6 January 2019.
- ^ Iglev, H.; Schmeisser, M.; Simeonidis, K.; Thaller, A.; Laubereau, A. (2006). "Ultrafast superheating and melting of bulk ice". Nature. 439 (7073): 183–186. Bibcode:2006Natur.439..183I. doi:10.1038/nature04415. PMID 16407948. S2CID 4404036.
- ^ La Placa, S. J.; Hamilton, W. C.; Kamb, B.; Prakash, A. (1972). "On a nearly proton ordered structure for ice IX". Journal of Chemical Physics. 58 (2): 567–580. Bibcode:1973JChPh..58..567L. doi:10.1063/1.1679238.
- ^ Klotz, S.; Besson, J. M.; Hamel, G.; Nelmes, R. J.; Loveday, J. S.; Marshall, W. G. (1999). "Metastable ice VII at low temperature and ambient pressure". Nature. 398 (6729): 681–684. Bibcode:1999Natur.398..681K. doi:10.1038/19480. S2CID 4382067.
- ^ Dutch, Stephen. "Ice Structure". University of Wisconsin Green Bay. Archived from the original on 16 October 2016. Retrieved 12 July 2017.
- ^ Salzmann, Christoph G.; Radaelli, Paolo G.; Hallbrucker, Andreas; Mayer, Erwin; Finney, John L. (24 March 2006). "The Preparation and Structures of Hydrogen Ordered Phases of Ice". Science. 311 (5768): 1758–1761. Bibcode:2006Sci...311.1758S. doi:10.1126/science.1123896. PMID 16556840. S2CID 44522271.
- ^ Sanders, Laura (11 September 2009). "A Very Special Snowball". Science News. Archived from the original on 14 September 2009. Retrieved 11 September 2009.
- ^ Militzer, Burkhard; Wilson, Hugh F. (2 November 2010). "New Phases of Water Ice Predicted at Megabar Pressures". Physical Review Letters. 105 (19): 195701. arXiv:1009.4722. Bibcode:2010PhRvL.105s5701M. doi:10.1103/PhysRevLett.105.195701. PMID 21231184. S2CID 15761164.
- ^ MacMahon, J. M. (1970). "Ground-State Structures of Ice at High-Pressures". Physical Review B. 84 (22): 220104. arXiv:1106.1941. Bibcode:2011PhRvB..84v0104M. doi:10.1103/PhysRevB.84.220104. S2CID 117870442.
- ^ Chang, Kenneth (9 December 2004). "Astronomers Contemplate Icy Volcanoes in Far Places". The New York Times. Archived from the original on 9 May 2015. Retrieved 30 July 2012.
- ^ Zyga, Lisa (25 April 2013). "New phase of water could dominate the interiors of Uranus and Neptune". Phys.org.
- ^ Rosu-Finsen, Alexander; Davies, Michael B.; Amon, Alfred; Wu, Han; Sella, Andrea; Michaelides, Angelos; Salzmann, Christoph G. (2023-02-03). "Medium-density amorphous ice". Science. 379 (6631): 474–478. Bibcode:2023Sci...379..474R. doi:10.1126/science.abq2105. ISSN 0036-8075. PMID 36730416. S2CID 256504172.
- ^ Murray, Benjamin J.; Bertram, Allan K. (2006). "Formation and stability of cubic ice in water droplets". Physical Chemistry Chemical Physics. 8 (1): 186–192. Bibcode:2006PCCP....8..186M. doi:10.1039/b513480c. hdl:2429/33770. PMID 16482260.
- ^ Murray, Benjamin J. (2008). "The Enhanced formation of cubic ice in aqueous organic acid droplets". Environmental Research Letters. 3 (2): 025008. Bibcode:2008ERL.....3b5008M. doi:10.1088/1748-9326/3/2/025008.
- ^ Murray, Benjamin J.; Knopf, Daniel A.; Bertram, Allan K. (2005). "The formation of cubic ice under conditions relevant to Earth's atmosphere". Nature. 434 (7030): 202–205. Bibcode:2005Natur.434..202M. doi:10.1038/nature03403. PMID 15758996. S2CID 4427815.
- ^ Malkin, Tamsin L.; Murray, Benjamin J.; Salzmann, Christoph G.; Molinero, Valeria; Pickering, Steven J.; Whale, Thomas F. (2015). "Stacking disorder in ice I". Physical Chemistry Chemical Physics. 17 (1): 60–76. doi:10.1039/c4cp02893g. PMID 25380218.
- ^ Salzmann, Christoph G.; Murray, Benjamin J. (June 2020). "Ice goes fully cubic". Nature Materials. 19 (6): 586–587. Bibcode:2020NatMa..19..586S. doi:10.1038/s41563-020-0696-6. PMID 32461682. S2CID 218913209.
- ^ Chaplin, Martin (10 April 2012). "Ice-four (Ice IV)". Water Structure and Science. London South Bank University. Archived from the original on 12 August 2011. Retrieved 27 May 2022.
- ^ Chaplin, Martin (10 April 2012). "Ice-five (Ice V)". Water Structure and Science. London South Bank University. Archived from the original on 12 October 2003. Retrieved 30 July 2012.
- ^ Chaplin, Martin (10 April 2012). "Ice-six (Ice VI)". Water Structure and Science. London South Bank University. Archived from the original on 23 September 2012. Retrieved 30 July 2012.
- ^ a b Grande, Zachary M.; et al. (2022). "Pressure-driven symmetry transitions in dense H2O ice". APS Physics. 105 (10): 104109. Bibcode:2022PhRvB.105j4109G. doi:10.1103/PhysRevB.105.104109. S2CID 247530544.
- ^ Chaplin, Martin (10 April 2012). "Ice-seven (Ice VII)". Water Structure and Science. London South Bank University. Archived from the original on 2 November 2011. Retrieved 30 July 2012.
- ^ Chaplin, Martin (17 February 2017). "Ice-eleven (ice XI)". Water Structure and Science. London South Bank University. Archived from the original on 23 March 2017. Retrieved 11 March 2017.
- ^ a b Chaplin, Martin (10 April 2012). "Ice-twelve (Ice XII)". Water Structure and Science. London South Bank University. Archived from the original on 2 November 2011. Retrieved 30 July 2012.
- ^ a b "Sandwiching water between graphene makes square ice crystals at room temperature". ZME Science. 2015-03-27. Retrieved 2018-05-02.
- ^ Algara-Siller, G.; Lehtinen, O.; Wang, F. C.; Nair, R. R.; Kaiser, U.; Wu, H. A.; Geim, A. K.; Grigorieva, I. V. (2015-03-26). "Square ice in graphene nanocapillaries". Nature. 519 (7544): 443–445. arXiv:1412.7498. Bibcode:2015Natur.519..443A. doi:10.1038/nature14295. PMID 25810206. S2CID 4462633.
- ^ a b del Rosso, Leonardo; Celli, Milva; Ulivi, Lorenzo (7 November 2016). "New porous water ice metastable at atmospheric pressure obtained by emptying a hydrogen-filled ice". Nature Communications. 7 (1): 13394. arXiv:1607.07617. Bibcode:2016NatCo...713394D. doi:10.1038/ncomms13394. PMC 5103070. PMID 27819265.
- ^ Chaplin, Martin. "Ice-seventeen (Ice XVII)". Archived from the original on 11 September 2022. Retrieved 12 September 2022.
{{cite web}}: CS1 maint: bot: original URL status unknown (link)[self-published source?] - ^ Liu, Yuan; Huang, Yingying; Zhu, Chongqin; Li, Hui; Zhao, Jijun; Wang, Lu; Ojamäe, Lars; Francisco, Joseph S.; Zeng, Xiao Cheng (25 June 2019). "An ultralow-density porous ice with the largest internal cavity identified in the water phase diagram". Proceedings of the National Academy of Sciences. 116 (26): 12684–12691. Bibcode:2019PNAS..11612684L. doi:10.1073/pnas.1900739116. PMC 6600908. PMID 31182582.
